Scalable Asymmetric Hydrogenation of Imines for High-Purity Chiral Amine Production
Scalable Asymmetric Hydrogenation of Imines for High-Purity Chiral Amine Production
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies for the synthesis of chiral amines, which serve as critical building blocks for a vast array of bioactive molecules. Patent CN102951980A discloses a groundbreaking method for the asymmetric catalytic hydrogenation of imines, utilizing a sophisticated iridium-based catalyst system to produce chiral amines with high efficiency and selectivity. This technology represents a significant leap forward in synthetic organic chemistry, particularly for the manufacturing of pesticide intermediates such as S-metolachlor, where stereochemical purity is paramount for biological activity. By employing a complex formed from an iridium precursor and a specialized ferrocene-based chiral ligand in the presence of iodide additives and acid, this process achieves remarkable turnover numbers and enantiomeric excess values that meet rigorous industrial standards. The innovation lies not only in the catalyst design but also in the operational simplicity, allowing for reactions to proceed under relatively mild conditions while maintaining high conversion rates. For global supply chains, this patent offers a pathway to more reliable agrochemical intermediate supplier networks by simplifying the production of complex chiral structures that were previously difficult to access economically.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral amines has been plagued by significant technical and economic hurdles that hinder efficient large-scale manufacturing. Conventional approaches often rely on the resolution of racemic mixtures, a process that is inherently inefficient as it theoretically discards fifty percent of the material, leading to substantial waste and increased raw material costs. Furthermore, the separation of enantiomers frequently requires expensive chiral resolving agents or complex chromatographic techniques, which are difficult to scale and often result in lower overall yields. In the context of agrochemical production, where margins can be tight and volumes high, these inefficiencies translate directly into higher costs and longer lead times for high-purity chiral amines. Additionally, older catalytic systems sometimes suffer from poor stability or require harsh reaction conditions that are incompatible with sensitive functional groups, limiting the substrate scope and necessitating additional protection and deprotection steps. The reliance on biological enzyme splitting for chiral alcohol precursors, as noted in prior art, introduces further complexity, requiring extensive screening of lytic enzymes and time-consuming optimization that delays time-to-market for new products.
The Novel Approach
In stark contrast to these legacy methods, the technology described in patent CN102951980A introduces a streamlined catalytic hydrogenation route that directly converts prochiral imines into chiral amines with high stereoselectivity. This novel approach utilizes an iridium complex coordinated with a unique chiral ligand, {(R)-1-[(S)-2-di-phenyl-phosphino-ferrocene]}-propyl-di-(3,5-dimethyl-phenyl)phosphine, which creates a highly specific chiral environment around the metal center. The inclusion of iodide sources, such as tetrabutylammonium iodide or elemental iodine, alongside an organic acid activator, significantly enhances the catalytic activity and stability of the system. This allows for the direct hydrogenation of imines derived from readily available amines and ketones, such as 2-methyl-6-ethyl-aniline and methoxyacetone, bypassing the need for resolution steps entirely. 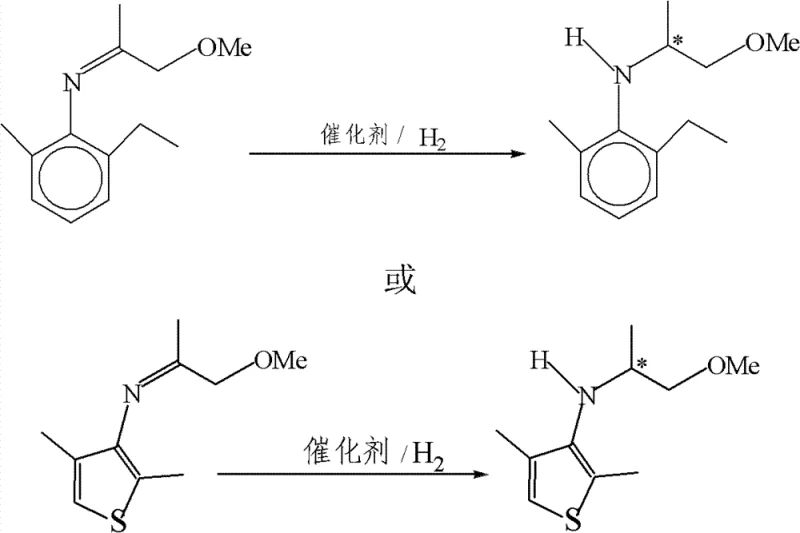
The versatility of this method is demonstrated by its ability to handle various substrates, including heterocyclic aromatic amines, yielding products with enantiomeric excess values sufficient for agricultural applications. By enabling the in-situ formation of imines from amines and ketones, the process further reduces operational steps and solvent usage, contributing to a greener and more cost-effective manufacturing protocol. This direct asymmetric synthesis strategy effectively eliminates the waste associated with racemic resolution, offering a sustainable alternative for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Iridium-Catalyzed Asymmetric Hydrogenation
The core of this technological advancement lies in the intricate interplay between the iridium metal center and the chiral ferrocene ligand, which dictates the stereochemical outcome of the hydrogenation reaction. The catalyst is generated in situ by reacting a dimeric iridium precursor, such as 1,5-cyclooctadiene iridium chloride, with the chiral phosphine ligand in the presence of an acid. This activation step likely generates a cationic iridium species that is highly electrophilic and capable of coordinating the imine substrate. The bulky 3,5-dimethyl-phenyl groups on the ligand create a steric pocket that forces the imine to approach the metal hydride species from a specific face, thereby controlling the formation of the new chiral center. The presence of iodide ions plays a crucial role in modulating the electronic properties of the catalyst, potentially stabilizing the active species and preventing catalyst deactivation pathways that are common in iridium chemistry. 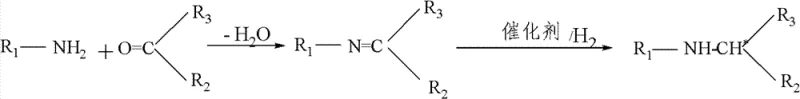
Furthermore, the acid component, typically acetic or formic acid, serves to protonate the imine nitrogen, increasing its electrophilicity and facilitating the hydride transfer step. This synergistic effect between the metal, ligand, iodide, and acid allows the reaction to proceed with high turnover frequencies, even at low catalyst loadings. The mechanism ensures that the hydrogen molecule is activated and transferred to the carbon-nitrogen double bond in a concerted manner that preserves the chiral information encoded in the ligand. Understanding these mechanistic nuances is vital for R&D directors aiming to optimize the process for new substrates, as slight modifications to the ligand structure or reaction additives could further enhance the enantioselectivity for specific target molecules. The robustness of this catalytic cycle underpins the method's ability to achieve substrate-to-catalyst ratios as high as 500,000, demonstrating exceptional efficiency.
How to Synthesize Chiral Amine Efficiently
The synthesis of chiral amines using this patented methodology involves a straightforward sequence of operations that can be easily adapted for both laboratory and pilot plant settings. The process begins with the preparation of the catalyst solution under an inert atmosphere to prevent oxidation of the sensitive phosphine ligands, followed by the charging of the reactor with the imine substrate and necessary additives. Detailed standard operating procedures regarding stoichiometry, pressure control, and workup protocols are essential for reproducibility and safety. The following guide outlines the generalized steps derived from the patent examples to assist technical teams in implementing this technology.
- Prepare the catalyst solution by mixing dimerization 1,5-cyclooctadiene iridium chloride with the chiral ligand {(R)-1-[(S)-2-di-phenyl-phosphino-ferrocene]}-propyl-di-(3,5-dimethyl-phenyl)phosphine in an organic acid solvent under nitrogen protection.
- Charge the autoclave with the imine substrate (or amine and ketone for in-situ formation), an iodide additive such as tetrabutylammonium iodide, and the prepared catalyst solution.
- Replace nitrogen with hydrogen, pressurize to 10-100 atm, and stir at room temperature to 60°C until hydrogen absorption ceases, followed by vacuum distillation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this asymmetric hydrogenation technology presents a compelling value proposition centered on cost efficiency and supply reliability. The ability to operate with extremely high substrate-to-catalyst ratios means that the consumption of the expensive iridium metal and chiral ligand is minimized per kilogram of product, leading to significant cost reduction in agrochemical intermediate manufacturing. Unlike resolution processes that cap the maximum theoretical yield at fifty percent, this direct hydrogenation method offers near-quantitative conversion, effectively doubling the output from the same amount of starting materials. This dramatic improvement in atom economy translates directly into lower raw material costs and reduced waste disposal expenses, aligning with modern sustainability goals. Moreover, the use of commercially available starting materials like substituted anilines and simple ketones ensures a stable and diverse supply base, reducing the risk of bottlenecks associated with specialized chiral pool reagents.
- Cost Reduction in Manufacturing: The elimination of resolution steps and the high efficiency of the catalyst system drastically simplify the production workflow, removing the need for expensive chiral columns or resolving agents. By avoiding the discard of unwanted enantiomers, the process maximizes the utility of every gram of raw material purchased, resulting in substantial overall cost savings. The simplified downstream processing, often requiring only vacuum distillation to recover the product and solvent, further reduces energy consumption and operational overhead. These factors combine to create a highly competitive cost structure that allows manufacturers to offer high-purity chiral amine at more attractive price points without compromising quality.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system allows for flexible production scheduling, as the reaction can be completed within a reasonable timeframe ranging from a few hours to a day. The tolerance of the catalyst to various substrates means that the same catalytic platform can be used to produce a family of related intermediates, simplifying inventory management and equipment utilization. This flexibility ensures that supply chain heads can respond quickly to fluctuations in market demand for key agrochemical ingredients. Additionally, the potential for continuous operation mentioned in the patent opens the door for even greater throughput and consistency, further securing the supply chain against disruptions.
- Scalability and Environmental Compliance: The method is explicitly designed for industrial scalability, having been tested in autoclaves and shown to be suitable for loop reactors, which indicates a clear path from bench scale to multi-ton production. The reduction in chemical waste, particularly the avoidance of half the material being discarded as seen in resolution, significantly lowers the environmental footprint of the manufacturing process. This aligns with increasingly stringent global regulations on chemical waste and emissions, reducing the regulatory burden on production facilities. The use of common solvents and reagents also simplifies waste treatment protocols, making it easier to maintain compliance with environmental standards while expanding production capacity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing production lines.
Q: What is the enantiomeric excess (e.e.) achievable with this iridium catalyst system?
A: According to the patent data, the method can achieve e.e. values above 70%, with specific examples reaching up to 79% for key agrochemical intermediates like the precursor to S-metolachlor, which satisfies industrial requirements for pesticide synthesis.
Q: What are the optimal reaction conditions for this asymmetric hydrogenation?
A: The process operates effectively at temperatures ranging from room temperature to 60°C and hydrogen pressures between 10 to 100 atmospheres. The reaction typically completes within a few hours to 24 hours depending on the specific substrate and catalyst loading.
Q: Can this method be scaled for industrial production of chiral amines?
A: Yes, the patent explicitly highlights the method's suitability for large-scale preparation. It features a high substrate-to-catalyst ratio (s/c) of up to 500,000, simple operation, and the potential for continuous operation, making it highly practical for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN102951980A for the production of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with high-pressure hydrogenation reactors and rigorous QC labs capable of monitoring enantiomeric purity with precision, guaranteeing that every batch meets stringent purity specifications. We are committed to leveraging our technical expertise to help clients navigate the complexities of chiral synthesis, delivering consistent quality and reliability for critical supply chains.
We invite you to collaborate with us to optimize your supply chain for chiral amines and other specialty intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to our deep knowledge of catalytic hydrogenation and process optimization, allowing you to reduce lead time for high-purity chiral amines and secure a competitive advantage in the market. Please contact us to request specific COA data and route feasibility assessments for your target molecules, and let us demonstrate how our engineering capabilities can drive your project forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
