Scalable Production of High-Purity 1,2-Diols Using Recyclable Selenium Polymer Catalysts
The chemical synthesis of 1,2-diols represents a critical pathway in the production of high-value pharmaceutical intermediates and fine chemicals, yet traditional methodologies often suffer from poor atom economy and excessive waste generation. Patent CN102942451A introduces a transformative approach by utilizing selenium-containing high polymers as heterogeneous catalysts for the direct oxidation of olefins using hydrogen peroxide. This technology addresses the longstanding challenge of catalyst recovery associated with small-molecule selenium catalysts, offering a robust solution for industrial-scale manufacturing. By shifting from homogeneous to heterogeneous catalysis, the process enables simple filtration for catalyst separation, drastically simplifying the downstream processing workflow. The innovation lies not only in the chemical transformation but also in the engineering of the catalyst support, which ensures stability and reusability over multiple reaction cycles. For R&D directors and process chemists, this patent provides a blueprint for developing cleaner, more sustainable synthetic routes that align with modern green chemistry principles. The ability to operate under mild conditions, specifically between 15°C and 80°C, further enhances the safety profile and energy efficiency of the manufacturing process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1,2-diols from olefins has relied heavily on stoichiometric oxidants such as m-chloroperbenzoic acid (MCPBA) or homogeneous selenium catalysts like diphenyl diselenide. While effective on a laboratory scale, these methods present severe drawbacks when translated to commercial production environments. The use of MCPBA generates substantial quantities of m-chlorobenzoic acid as a by-product, resulting in poor atom economy and creating significant waste disposal burdens for chemical plants. Furthermore, homogeneous selenium catalysts, although efficient, are notoriously difficult to separate from the reaction mixture, often requiring complex chromatographic purification or extensive washing procedures that increase operational costs. The residual selenium content in the final product is also a major concern for pharmaceutical applications, necessitating rigorous and expensive metal scavenging steps to meet regulatory purity standards. These factors collectively contribute to high production costs and extended lead times, making conventional routes less attractive for large-scale supply chains.
The Novel Approach
The novel methodology described in the patent overcomes these hurdles by immobilizing selenium species onto a polystyrene backbone, creating a insoluble, heterogeneous catalyst system. 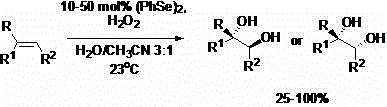 As illustrated in the general reaction scheme, the transformation utilizes hydrogen peroxide as a clean oxidant, producing water as the only by-product alongside the desired diol. The key breakthrough is the physical state of the catalyst; because the selenium species are covalently bound or strongly adsorbed onto the polymer matrix, they remain solid throughout the reaction in liquid solvents like acetonitrile. This allows for the catalyst to be recovered simply by filtration at the end of the reaction, eliminating the need for complex extraction or distillation steps to remove the catalyst. The patent demonstrates that this system maintains high catalytic activity, achieving yields comparable to or exceeding homogeneous systems, while offering the distinct logistical advantage of catalyst recycling. This shift fundamentally changes the economic model of the synthesis, turning a consumable catalyst expense into a reusable capital asset.
As illustrated in the general reaction scheme, the transformation utilizes hydrogen peroxide as a clean oxidant, producing water as the only by-product alongside the desired diol. The key breakthrough is the physical state of the catalyst; because the selenium species are covalently bound or strongly adsorbed onto the polymer matrix, they remain solid throughout the reaction in liquid solvents like acetonitrile. This allows for the catalyst to be recovered simply by filtration at the end of the reaction, eliminating the need for complex extraction or distillation steps to remove the catalyst. The patent demonstrates that this system maintains high catalytic activity, achieving yields comparable to or exceeding homogeneous systems, while offering the distinct logistical advantage of catalyst recycling. This shift fundamentally changes the economic model of the synthesis, turning a consumable catalyst expense into a reusable capital asset.
Mechanistic Insights into Selenium-Catalyzed Olefin Oxidation
The mechanistic pathway for this transformation involves the activation of hydrogen peroxide by the selenium center on the polymer support to form a reactive selenoxide or selenone intermediate. This electrophilic selenium species then interacts with the electron-rich double bond of the olefin substrate, facilitating the transfer of oxygen atoms to form the diol functionality. The polymer support plays a crucial role not just in recovery, but potentially in modulating the electronic environment of the selenium active site, which can influence selectivity and reaction rates. Detailed studies within the patent indicate that the nature of the selenium substituent on the polymer significantly impacts performance, with polystyrene-supported selenous acid (Cat A) exhibiting superior activity compared to other variants like selenides or selenates. 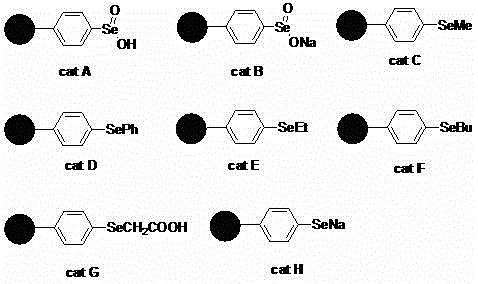 The structural diversity shown in the catalyst library allows for fine-tuning of the catalytic properties to suit specific substrate profiles. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters such as temperature and solvent polarity to maximize throughput. The heterogeneous nature of the reaction also minimizes side reactions often seen in homogeneous phases, leading to a cleaner impurity profile in the crude product.
The structural diversity shown in the catalyst library allows for fine-tuning of the catalytic properties to suit specific substrate profiles. Understanding this mechanism is vital for R&D teams aiming to optimize reaction parameters such as temperature and solvent polarity to maximize throughput. The heterogeneous nature of the reaction also minimizes side reactions often seen in homogeneous phases, leading to a cleaner impurity profile in the crude product.
Impurity control is inherently improved in this system due to the absence of soluble selenium species that could otherwise contaminate the product stream. In homogeneous catalysis, trace amounts of selenium can persist through workup, requiring additional purification stages that reduce overall yield. With the polymer-supported system, the physical separation via filtration ensures that the bulk of the catalyst remains in the solid phase, significantly reducing the burden on downstream purification units. This results in a final product with higher purity specifications, which is critical for pharmaceutical intermediates where strict limits on heavy metals and organometallic residues are enforced. The ability to recycle the catalyst up to 10 times without significant loss of activity further confirms the stability of the selenium-polymer bond under oxidative conditions. This robustness ensures consistent product quality batch after batch, a key requirement for validating a commercial manufacturing process.
How to Synthesize 1,2-Cyclohexanediol Efficiently
The synthesis of 1,2-cyclohexanediol serves as a prime example of the efficacy of this technology, demonstrating high yields and operational simplicity. The process begins by suspending the polystyrene-supported selenous acid catalyst in acetonitrile along with the cyclohexene substrate, ensuring thorough mixing before the introduction of the oxidant. Hydrogen peroxide is then added carefully to manage the exotherm, with the reaction proceeding at mild temperatures around 30°C to optimize selectivity and safety. Following the reaction period, the solid catalyst is removed by standard filtration, and the filtrate is subjected to distillation to recover the solvent and isolate the pure diol product. This streamlined workflow eliminates the need for aqueous workups or column chromatography, making it highly suitable for kilogram-to-ton scale production.
- Charge a reactor with the olefin substrate, polystyrene-supported selenous acid catalyst (Cat A), and acetonitrile solvent, then stir to form a suspension.
- Slowly add aqueous hydrogen peroxide (27.5% concentration) while maintaining the reaction temperature between 15°C and 80°C, ideally around 30°C.
- Upon completion, filter the reaction mixture to recover the solid catalyst, then distill the filtrate to recover solvent and isolate the pure 1,2-diol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this selenium-polymer catalytic technology offers substantial strategic benefits regarding cost structure and operational reliability. The primary economic driver is the drastic reduction in catalyst consumption costs achieved through the ability to recover and reuse the heterogeneous catalyst multiple times. Unlike homogeneous catalysts which are consumed or lost in every batch, the polymer-supported variant acts as a durable tool that amortizes its cost over many production cycles, leading to significant long-term savings. Additionally, the simplification of the workup procedure reduces the consumption of auxiliary materials such as extraction solvents and purification media, further lowering the variable cost per kilogram of product. The use of hydrogen peroxide as a terminal oxidant is also economically favorable compared to expensive stoichiometric oxidants like MCPBA, contributing to a more competitive cost of goods sold.
- Cost Reduction in Manufacturing: The elimination of complex purification steps required to remove homogeneous catalyst residues directly translates to reduced processing time and lower utility costs. By avoiding the need for specialized metal scavengers or extensive chromatography, the manufacturing process becomes leaner and more cost-effective. The high atom economy of using hydrogen peroxide ensures that raw material costs are minimized, as the oxidant is converted primarily into water rather than heavy organic waste. These factors combine to create a manufacturing route that is significantly more economical than traditional methods, enhancing the margin potential for the final chemical product.
- Enhanced Supply Chain Reliability: The robustness of the catalyst and the simplicity of the reaction conditions contribute to a more reliable supply chain with fewer points of failure. The ability to operate at ambient pressure and moderate temperatures reduces the risk of safety incidents that could disrupt production schedules. Furthermore, the ease of catalyst recovery ensures that production is not dependent on the continuous sourcing of fresh catalyst for every batch, buffering the supply chain against raw material volatility. This stability is crucial for maintaining consistent delivery timelines to downstream pharmaceutical customers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The heterogeneous nature of the process facilitates easy scale-up from pilot plant to commercial production without the engineering challenges associated with handling toxic homogeneous selenium solutions. The reduction in chemical waste, particularly the avoidance of chlorinated by-products from MCPBA oxidation, simplifies environmental compliance and waste treatment protocols. This aligns with increasingly stringent global regulations on industrial emissions and effluent discharge, future-proofing the manufacturing facility against regulatory changes. The green chemistry profile of the process also enhances the corporate sustainability metrics, which is becoming a key differentiator in B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium-polymer catalytic technology in industrial settings. These insights are derived directly from the experimental data and process descriptions provided in the patent literature to ensure accuracy and relevance. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of using polymer-supported selenium catalysts over homogeneous variants?
A: Polymer-supported catalysts allow for simple filtration recovery, eliminating the need for complex purification steps to remove soluble selenium residues, thereby reducing production costs and environmental impact.
Q: Can the selenium polymer catalyst be reused multiple times without significant loss of activity?
A: Yes, the patent data indicates that the catalyst can be recycled and reused for up to 10 cycles while maintaining high product yields, demonstrating excellent stability and cost-efficiency.
Q: What is the optimal solvent system for this oxidation reaction?
A: Acetonitrile has been identified as the preferred solvent, providing superior reaction yields compared to ethanol, water, or acetic acid under the specified catalytic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,2-Diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the selenium-polymer system for producing high-purity 1,2-diols. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 1,2-cyclohexanediol or related intermediates meets the exacting standards required by the global pharmaceutical industry. We are committed to leveraging such green chemistry advancements to deliver superior value to our clients while maintaining the highest levels of quality and safety.
We invite you to collaborate with us to explore how this efficient synthesis route can optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in catalytic oxidation can become a cornerstone of your sourcing strategy for critical chemical intermediates.
