Advanced Manufacturing of Linezolid Intermediates via Novel Imine Protection Strategy
Introduction to Next-Generation Linezolid Manufacturing
The global demand for effective antibacterial agents continues to rise in response to increasing antimicrobial resistance, placing significant pressure on pharmaceutical supply chains to deliver high-quality active pharmaceutical ingredients (APIs) efficiently. Patent CN101415694A introduces a groundbreaking methodology for the preparation of linezolid, a critical oxazolidinone antibiotic, addressing key bottlenecks in traditional synthetic routes. This innovative process utilizes a highly concentrated three-step sequence that significantly reduces solvent consumption and cycle times compared to legacy methods. By leveraging a unique imine protection strategy, the technology enables the production of linezolid with exceptional purity and yield, making it an attractive option for manufacturers seeking to optimize their production capabilities. The strategic implementation of this route offers a robust pathway for reliable pharmaceutical intermediate supplier networks to enhance their portfolio offerings.
Furthermore, the environmental footprint of this synthesis is markedly reduced through the minimization of large solvent volumes typically required in multi-step organic syntheses. The process begins with the formation of a protected imine intermediate, which serves as a versatile precursor for the subsequent cyclization. This approach not only streamlines the workflow but also mitigates the generation of hazardous waste, aligning with modern green chemistry principles. For procurement managers and supply chain heads, understanding the technical nuances of such patents is crucial for evaluating long-term sourcing strategies and ensuring continuity of supply for essential medications. The detailed disclosure within the patent provides a clear roadmap for scaling this chemistry from laboratory benchtop to industrial reactor vessels.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for oxazolidinone antibiotics often suffer from inherent inefficiencies that drive up manufacturing costs and complicate supply chain logistics. Many established methods rely on the use of expensive chiral catalysts or resolving agents to achieve the necessary stereochemistry, which can introduce significant variability and cost volatility into the production process. Additionally, conventional pathways frequently involve multiple protection and deprotection steps that require harsh reaction conditions, leading to lower overall yields and the generation of complex impurity profiles that are difficult to purge. The reliance on cryogenic temperatures or specialized reagents in older methodologies further restricts the ability to scale these processes effectively in standard manufacturing facilities. These factors collectively contribute to extended lead times and higher unit costs, posing challenges for maintaining competitive pricing in the generic pharmaceutical market.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent utilizes a streamlined strategy that bypasses many of these historical hurdles through the clever application of imine chemistry. The core innovation lies in the reaction of (S)-1-chloro-3-(benzylideneamino)propan-2-ol with a fluorophenyl carbamate to directly construct the oxazolidinone ring system. This method operates under relatively mild conditions, typically ranging from room temperature to approximately 65°C, which reduces energy consumption and equipment stress. The use of a benzylidene imine group acts as a temporary mask for the amine, allowing the cyclization to proceed cleanly without interference, and is subsequently removed via simple hydrolysis. This efficiency translates directly into cost reduction in API manufacturing by minimizing unit operations and maximizing throughput. The process is designed to be highly concentrated, drastically cutting down on the volume of solvents like DMF or dichloromethane required, which simplifies downstream processing and waste management.
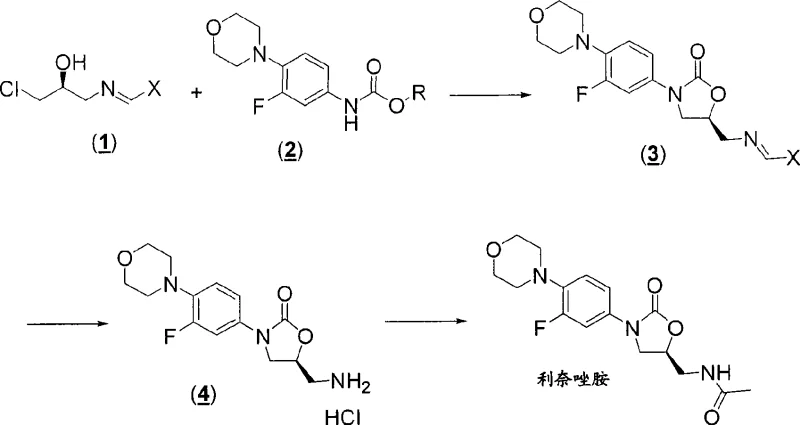
Mechanistic Insights into Base-Catalyzed Cyclization
The chemical elegance of this synthesis is rooted in the precise mechanistic control exerted during the cyclization step, which is critical for ensuring high-purity linezolid. The reaction employs a strong base, preferably lithium tert-butoxide, to deprotonate the carbamate nitrogen, generating a nucleophilic species that attacks the chloro-substituted carbon of the chiral imine intermediate. This intramolecular substitution proceeds with inversion of configuration at the chiral center, but since the starting material is derived from (S)-epichlorohydrin, the final stereochemistry is perfectly aligned with the therapeutic requirements of linezolid. The choice of solvent plays a pivotal role here; aprotic non-nucleophilic solvents such as MTBE or dichloromethane are preferred to stabilize the transition state and prevent side reactions. The robustness of this mechanism allows for a wide operating window, providing manufacturers with the flexibility to optimize reaction times between 2 to 24 hours depending on batch size and thermal dynamics.
Impurity control is another area where this mechanism excels, particularly regarding the management of enantiomeric purity. The patent highlights that the crystallization of the intermediate imine compounds can surprisingly improve enantiomeric excess, acting as a built-in purification step before the final cyclization. This is achieved by recrystallizing the imine from specific solvent mixtures, such as MTBE and heptane, which selectively precipitate the desired enantiomer while leaving impurities in the mother liquor. Such intrinsic quality control measures reduce the burden on final purification stages, ensuring that the resulting amine hydrochloride salt meets stringent purity specifications prior to acylation. Understanding these mechanistic details allows R&D directors to anticipate potential scale-up issues and implement proactive quality assurance protocols.
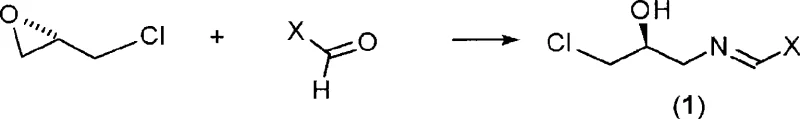
How to Synthesize Linezolid Efficiently
Implementing this synthesis requires careful attention to the preparation of the chiral starting materials and the control of reaction parameters during the cyclization phase. The process begins with the condensation of (S)-epichlorohydrin with a substituted benzaldehyde in the presence of ammonia to form the key imine intermediate. This step is crucial as it sets the stereochemical foundation for the entire molecule. Following isolation and optional recrystallization to enhance purity, the imine is coupled with the appropriate carbamate derivative under basic conditions. The detailed standardized synthesis steps see the guide below for specific molar ratios, temperature profiles, and workup procedures that ensure reproducible results.
- React (S)-epichlorohydrin with substituted benzaldehyde and ammonia to form the chiral imine intermediate (S)-1-chloro-3-(benzylideneamino)propan-2-ol.
- Couple the chiral imine intermediate with a fluorophenyl carbamate derivative using lithium tert-butoxide to form the protected oxazolidinone imine.
- Perform acidic hydrolysis to remove the imine protecting group followed by acylation to yield the final linezolid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented process offers tangible benefits that extend beyond mere technical feasibility. The streamlined nature of the three-step sequence inherently reduces the complexity of the manufacturing workflow, which correlates directly with lower operational expenditures. By eliminating the need for exotic reagents or complex multi-stage protection schemes, the process relies on commodity chemicals that are readily available in the global market, thereby enhancing supply chain reliability. The reduction in solvent usage not only lowers raw material costs but also decreases the logistical burden associated with solvent recovery and disposal. This efficiency makes the process highly attractive for commercial scale-up of complex pharmaceutical intermediates, allowing suppliers to offer more competitive pricing without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic advantages of this route are driven by the significant simplification of the synthetic pathway. By avoiding the use of expensive transition metal catalysts and reducing the number of isolation steps, the process minimizes both material and labor costs. The high concentration of the reaction mixture means that smaller reactor volumes can be used to produce the same amount of product, effectively increasing facility capacity. Furthermore, the ability to crystallize intermediates directly from the reaction mixture reduces the need for extensive chromatographic purification, which is often a major cost driver in fine chemical manufacturing. These factors combine to create a leaner, more cost-effective production model.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for pharmaceutical buyers, and this process addresses that by utilizing widely available starting materials like epichlorohydrin and various benzaldehydes. Unlike routes that depend on single-source specialty reagents, the inputs for this synthesis are produced by multiple vendors globally, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further securing the supply chain. This resilience ensures reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more quickly to market demands.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the process is designed with scalability in mind. The reduction in solvent volume directly translates to a smaller environmental footprint, facilitating compliance with increasingly strict waste disposal regulations. The crystallization steps described are amenable to large-scale filtration and drying equipment, ensuring that the transition from pilot plant to full commercial production is smooth. The generation of fewer by-products simplifies waste treatment protocols, lowering the overall cost of environmental compliance. This alignment with green chemistry principles enhances the sustainability profile of the supply chain, a key metric for modern corporate responsibility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this linezolid synthesis route. These answers are derived directly from the technical disclosures within the patent documentation to provide accurate guidance for industry professionals. Understanding these aspects is vital for making informed decisions about technology transfer and vendor qualification.
Q: What is the primary advantage of using an imine protecting group in this synthesis?
A: The imine group effectively protects the amine functionality during the cyclization step, preventing side reactions, while remaining labile enough to be easily removed via mild acidic hydrolysis in the final stages, simplifying purification.
Q: How is stereochemical purity maintained throughout the process?
A: Chirality is introduced at the very beginning using commercially available (S)-epichlorohydrin. The subsequent reactions proceed with retention of configuration, and the crystallization steps described in the patent further enhance enantiomeric excess to >99%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the patent explicitly details scalable crystallization procedures and the use of common industrial solvents like MTBE and dichloromethane, indicating a design optimized for commercial scale-up and robust supply chain integration.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linezolid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex molecules like linezolid can be manufactured with consistency and precision. We are committed to meeting stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch. By leveraging the insights from patents such as CN101415694A, we continuously optimize our processes to deliver superior value to our global partners.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of this novel route for your portfolio, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your requirements. Let us help you engineer a more resilient and cost-effective supply solution for your oxazolidinone antibiotic programs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
