Optimizing Fulvestrant Manufacturing: A Novel Steroid Synthesis Route for Global Supply Chains
Optimizing Fulvestrant Manufacturing: A Novel Steroid Synthesis Route for Global Supply Chains
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective pathways for the production of critical oncology therapeutics. A significant breakthrough in this domain is detailed in patent CN108264534B, which outlines a novel preparation method for Fulvestrant, a potent estrogen receptor antagonist widely used in breast cancer treatment. This intellectual property introduces a strategic departure from conventional synthetic methodologies by leveraging a Grignard substitution on a 7-carbonyl group, thereby circumventing the historically problematic reconstruction of the aromatic A-ring. By initiating the synthesis from a pre-functionalized steroid precursor (Compound 1), the process achieves superior selectivity at the 7-position side chain attachment. This technical advancement not only streamlines the reaction sequence but also mitigates the formation of difficult-to-separate impurities, presenting a compelling value proposition for reliable API intermediate supplier networks aiming to enhance their portfolio of high-purity steroid compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Fulvestrant has been plagued by inefficiencies inherent in early-generation routes. As illustrated in the prior art, traditional pathways often commence with Nandrolone as the starting material. This approach necessitates a series of aggressive transformations, including bromination, double bond formation, and the arduous construction of the aromatic ring. 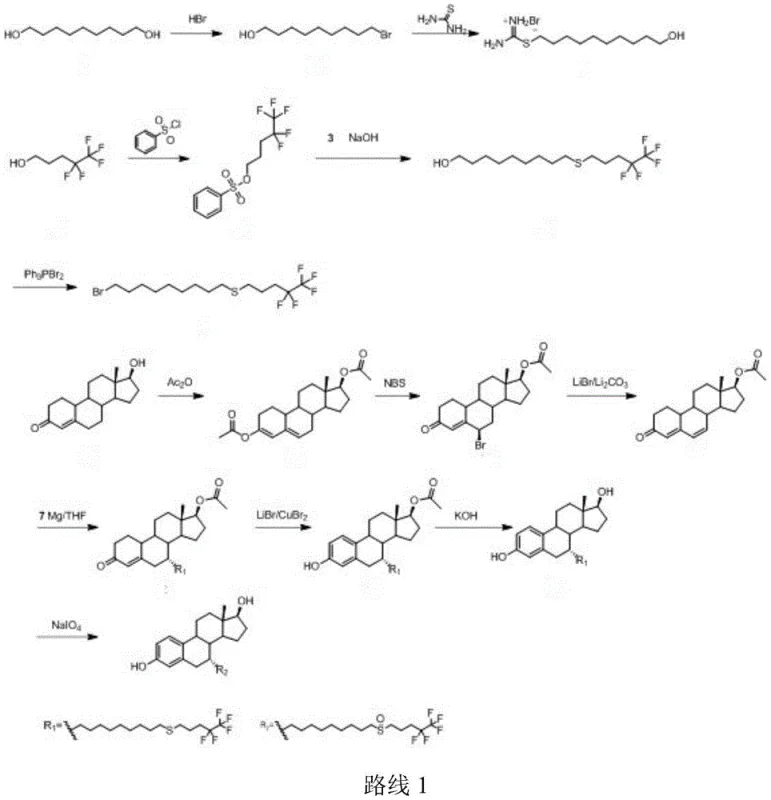 These conventional methods suffer from inherent instability during the bromination steps, leading to a proliferation of byproducts particularly during the 6-position and 7-position dehydrogenation phases. The resulting crude mixtures impose severe burdens on downstream purification processes, often requiring extensive chromatographic separation which drastically reduces overall yield. Furthermore, alternative routes reported in literature, such as those involving 6-position oxidation and carbonyl alpha-position substitution, have demonstrated molar yields as low as 20-30%. Such poor efficiency translates directly into exorbitant manufacturing costs and inconsistent supply continuity, creating significant bottlenecks for cost reduction in pharmaceutical intermediates manufacturing.
These conventional methods suffer from inherent instability during the bromination steps, leading to a proliferation of byproducts particularly during the 6-position and 7-position dehydrogenation phases. The resulting crude mixtures impose severe burdens on downstream purification processes, often requiring extensive chromatographic separation which drastically reduces overall yield. Furthermore, alternative routes reported in literature, such as those involving 6-position oxidation and carbonyl alpha-position substitution, have demonstrated molar yields as low as 20-30%. Such poor efficiency translates directly into exorbitant manufacturing costs and inconsistent supply continuity, creating significant bottlenecks for cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel process defined in CN108264534B adopts a convergent strategy that preserves the integrity of the steroid core while efficiently installing the requisite side chains. 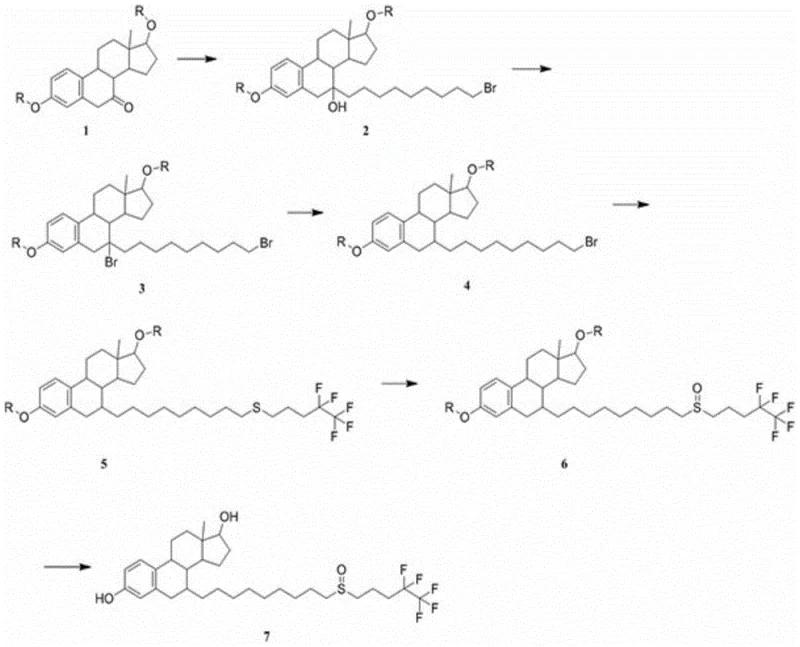 The innovation lies in the utilization of Compound 1, which already possesses the aromatic A-ring, thus eliminating the energy-intensive and low-yield aromatization steps entirely. The pathway proceeds through a highly controlled Grignard reaction with 1,9-dibromononane to install the long alkyl spacer, followed by selective functionalization to attach the fluorinated tail. This methodology ensures good selectivity and significantly improves the yield of the 7-position side chain coupling. By avoiding the reconstruction of the aromatic ring and utilizing common reagents, the process simplifies post-reaction treatment, making it far more operationally friendly and environmentally benign compared to the prior art. This represents a paradigm shift towards more sustainable and economically viable commercial scale-up of complex steroid intermediates.
The innovation lies in the utilization of Compound 1, which already possesses the aromatic A-ring, thus eliminating the energy-intensive and low-yield aromatization steps entirely. The pathway proceeds through a highly controlled Grignard reaction with 1,9-dibromononane to install the long alkyl spacer, followed by selective functionalization to attach the fluorinated tail. This methodology ensures good selectivity and significantly improves the yield of the 7-position side chain coupling. By avoiding the reconstruction of the aromatic ring and utilizing common reagents, the process simplifies post-reaction treatment, making it far more operationally friendly and environmentally benign compared to the prior art. This represents a paradigm shift towards more sustainable and economically viable commercial scale-up of complex steroid intermediates.
Mechanistic Insights into Grignard-Mediated Side Chain Installation
The cornerstone of this synthetic innovation is the precise execution of the Grignard substitution at the C7 position of the steroid nucleus. The mechanism involves the generation of an organomagnesium species from 1,9-dibromononane and magnesium powder in aprotic solvents such as tetrahydrofuran or dioxane. This reactive intermediate attacks the 7-carbonyl group of the protected steroid (Compound 1), forming a tertiary alcohol intermediate (Compound 2) with high regioselectivity. The choice of solvent and the stoichiometric control of magnesium are critical; the patent specifies a molar ratio of magnesium to Compound 1 between 0.6:1 and 0.9:1, ensuring complete conversion while minimizing the formation of Wurtz-type coupling byproducts. Following this, the terminal bromide of the side chain is activated for nucleophilic displacement. The subsequent steps involve a sophisticated sequence where the bromide is converted to a thiourea salt and then coupled with 4,4,5,5,5-pentafluoropentanol methanesulfonate under alkaline conditions. This thioether linkage is subsequently oxidized to the sulfoxide functionality using mild oxidants like hydrogen peroxide or m-chloroperoxybenzoic acid, a transformation that is crucial for the biological activity of the final Fulvestrant molecule.
Impurity control is rigorously addressed through the design of the reaction sequence, specifically the avoidance of harsh acidic or basic conditions that typically degrade the sensitive steroid skeleton. In conventional routes, the aromatization step often generates isomeric impurities that are structurally similar to the target, making them nearly impossible to remove without significant yield loss. In this novel route, the aromatic ring is present from the outset, protected as an ether or ester (R group), which remains stable throughout the Grignard and alkylation steps. The final deprotection step, utilizing mild hydrolysis with sodium hydroxide or dilute hydrochloric acid, cleanly reveals the phenolic hydroxyl group without affecting the newly formed sulfoxide or the fluorinated tail. This orthogonal protection strategy ensures that the final high-purity Fulvestrant meets stringent pharmacopeial standards with minimal need for repetitive recrystallization, thereby enhancing the overall process mass intensity (PMI) and reducing solvent waste.
How to Synthesize Fulvestrant Efficiently
The synthesis of Fulvestrant via this patented route offers a streamlined alternative to legacy processes, focusing on operational simplicity and high fidelity. The procedure begins with the preparation of the side-chain extended intermediate through a Grignard reaction, followed by a series of functional group interconversions that install the fluorinated sulfoxide tail. The detailed standardized synthetic steps, including specific reagent ratios, solvent choices, and workup procedures, are outlined below to guide process development teams in replicating this high-efficiency pathway.
- React the starting steroid ketone (Compound 1) with 1,9-dibromononane and magnesium powder to form the side-chain intermediate.
- Perform selective bromination and subsequent hydrogenation to refine the steroid core structure.
- Execute nucleophilic substitution with thiourea and fluorinated alcohol mesylate, followed by oxidation and deprotection to yield Fulvestrant.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic advantages beyond mere technical elegance. The primary value driver is the substantial simplification of the raw material supply chain. By eliminating the need for Nandrolone and the associated reagents required for aromatization, manufacturers can source more ubiquitous and cost-effective starting materials. The reliance on common reagents such as magnesium powder, thiourea, and standard alkyl halides reduces exposure to volatile pricing markets associated with specialized chiral catalysts or rare earth metals. Furthermore, the elimination of noble metal catalysis in key steps removes the necessity for expensive metal scavenging processes and the regulatory burden of validating residual metal levels in the final API, leading to significant cost reduction in manufacturing overheads.
- Cost Reduction in Manufacturing: The process architecture inherently lowers production costs by improving overall yield and reducing the number of purification cycles. Traditional routes often suffer from yields below 30% in critical oxidation steps, necessitating large batch sizes to achieve target output, which inflates utility and labor costs. In contrast, this new method's improved selectivity means less raw material is wasted on byproducts. Additionally, the use of standard solvents like ethyl acetate, methanol, and toluene allows for efficient solvent recovery and recycling loops, further driving down the variable cost per kilogram. The avoidance of complex chromatographic separations required for removing aromatization byproducts translates directly into reduced silica gel consumption and shorter cycle times.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions. The Grignard reaction and subsequent alkylation steps are well-understood unit operations that can be performed in standard glass-lined or stainless steel reactors without requiring specialized high-pressure or cryogenic equipment. This compatibility with existing infrastructure reduces the lead time for technology transfer and capacity expansion. Moreover, the stability of the intermediates, particularly the protected steroid cores, allows for potential campaign manufacturing strategies where key intermediates can be stockpiled, buffering against fluctuations in demand for the final API.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, the route is markedly superior. The avoidance of harsh bromination conditions used in Nandrolone-based routes minimizes the generation of corrosive waste streams and hazardous halogenated byproducts. The oxidation step utilizes hydrogen peroxide or periodates, which generate water or benign salts as byproducts, aligning with green chemistry principles. This reduced environmental footprint simplifies waste disposal compliance and lowers the carbon footprint of the manufacturing site, a critical metric for modern pharmaceutical supply chains aiming for sustainability certifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Fulvestrant synthesis route. These insights are derived directly from the experimental data and process descriptions within the patent, providing a clear understanding of the operational parameters and quality outcomes associated with this technology.
Q: How does this new route improve upon traditional Nandrolone-based synthesis?
A: Unlike traditional routes that require unstable bromination and complex aromatic ring construction from Nandrolone, this novel method starts with a pre-formed aromatic ring, significantly reducing byproduct formation and purification difficulty.
Q: What are the key cost drivers eliminated in this process?
A: The process eliminates the need for noble metal catalysis in certain steps and utilizes common, commercially available reagents like magnesium and thiourea, drastically simplifying the supply chain for raw materials.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the method employs standard unit operations such as reflux, extraction, and column chromatography with common solvents like THF and ethyl acetate, making it highly amenable to scale-up from pilot to multi-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fulvestrant Supplier
The technical potential of this Grignard-based synthesis route for Fulvestrant represents a significant opportunity for optimizing the global supply of this critical oncology drug. At NINGBO INNO PHARMCHEM, we possess the technical acumen and infrastructure to translate such innovative patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Fulvestrant intermediate meets the highest international standards.
We invite pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging this optimized synthetic route, we can help you reduce your cost of goods sold while securing a stable supply of high-quality intermediates. Please contact our technical procurement team to request specific COA data, route feasibility assessments, and a comprehensive quotation that reflects the efficiencies of this next-generation manufacturing process.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
