Scalable Manufacturing of Fulvestrant Intermediates via Stable Isothiourea Coupling Technology
Scalable Manufacturing of Fulvestrant Intermediates via Stable Isothiourea Coupling Technology
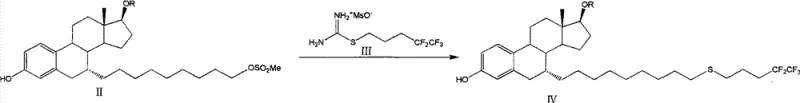
The pharmaceutical industry continuously seeks robust synthetic routes for complex oncology agents like fulvestrant, a potent estrogen receptor antagonist used in the treatment of advanced breast cancer. A pivotal advancement in this domain is detailed in patent CN103788164A, which discloses a novel preparation method focusing on the efficient construction of the critical thioether linkage at the 7-alpha position of the steroid nucleus. This technology represents a significant departure from conventional methodologies by employing a stable isothiourea mesylate reagent, specifically S-(4,4,5,5,5-pentafluoroamyl)isothiourea mesylate, to introduce the fluorinated side chain. The strategic shift from volatile, odorous thiols to a crystalline, shelf-stable salt addresses long-standing challenges in process safety and reproducibility. For R&D directors and process chemists, this approach offers a compelling solution to the difficulties associated with handling reactive sulfur species, while simultaneously enhancing the overall yield and purity of the target intermediate. The method's compatibility with common industrial solvents and bases further underscores its potential for seamless integration into existing manufacturing infrastructures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fulvestrant and its analogues has relied heavily on the nucleophilic substitution of a steroid leaving group with a pre-formed sodium mercaptide derived from the fluorinated side chain. This traditional pathway, while chemically feasible, presents substantial operational hazards and inefficiencies at a commercial scale. Sodium mercaptides are notoriously unstable and possess an extremely offensive odor, necessitating specialized containment equipment and rigorous safety protocols that drive up capital expenditure. Furthermore, these thiolate species are highly susceptible to air oxidation, leading to the formation of disulfide impurities that are difficult to separate from the desired product, thereby compromising the impurity profile of the API. The requirement for strictly anhydrous and anaerobic conditions often limits the choice of solvents and complicates the workup procedure, resulting in lower throughput and increased waste generation. For procurement managers, the reliance on such hazardous reagents translates to higher costs for safety compliance and waste disposal, alongside potential supply chain disruptions due to the limited availability of high-quality thiol precursors.
The Novel Approach
In contrast, the methodology outlined in CN103788164A introduces a paradigm shift by utilizing S-(4,4,5,5,5-pentafluoroamyl)isothiourea mesylate as the sulfur transfer agent. This reagent is a stable, non-volatile solid that eliminates the handling risks associated with free thiols and their metal salts. The reaction proceeds smoothly under relatively mild conditions, typically involving heating in solvents such as acetonitrile or ethanol in the presence of a mild base like potassium carbonate or triethylamine. This innovation drastically simplifies the reaction setup, removing the need for inert atmosphere gloveboxes or specialized scrubbing systems for noxious gases. The stability of the isothiourea salt ensures consistent reactivity batch-to-batch, which is critical for maintaining quality control in GMP manufacturing. Moreover, the byproduct of this coupling reaction is urea or a related soluble species, which can be easily removed via aqueous extraction, streamlining the isolation of the crude product. This transition from a hazardous liquid reagent to a benign solid reagent exemplifies a green chemistry improvement that aligns with modern regulatory expectations for sustainable pharmaceutical manufacturing.
Mechanistic Insights into Isothiourea-Mediated Nucleophilic Substitution
The core of this synthetic strategy lies in the nucleophilic displacement of the mesylate leaving group on the steroid intermediate (Formula II) by the sulfur atom of the isothiourea reagent (Formula III). Mechanistically, the reaction likely proceeds via an SN2 pathway, where the sulfur atom, activated by the adjacent imine functionality of the isothiourea moiety, attacks the electrophilic carbon at the 7-position of the steroid skeleton. The presence of the mesylate counterion on the isothiourea reagent enhances its solubility in polar organic solvents, facilitating effective collision frequency between the reactants. The use of bases such as potassium carbonate serves to neutralize the methanesulfonic acid generated during the displacement, driving the equilibrium towards product formation without promoting elimination side reactions that could degrade the sensitive steroid double bond system. This careful balance of reactivity ensures that the stereochemical integrity at the 7-alpha position is maintained, which is crucial for the biological activity of the final drug substance.
From an impurity control perspective, this mechanism offers distinct advantages over thiol-based routes. In traditional methods, the high nucleophilicity of free thiolates can lead to over-alkylation or attack on other electrophilic sites within the molecule. The isothiourea reagent acts as a "masked" thiol equivalent, moderating the nucleophilicity to favor the specific substitution at the primary or secondary mesylate site. Additionally, the oxidative instability of free thiols is completely bypassed, virtually eliminating the formation of symmetrical disulfides (R-S-S-R) which are common and persistent impurities in thioether syntheses. The subsequent hydrolysis of the isothiourea adduct (if formed as an intermediate salt) or the direct formation of the thioether ensures a cleaner reaction profile. This mechanistic elegance translates directly to reduced downstream processing requirements, as fewer chromatographic purifications are needed to meet stringent pharmacopeial standards for genotoxic impurities and residual solvents.
How to Synthesize Fulvestrant Intermediate Efficiently
The implementation of this synthesis route requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process begins with the preparation of the key reagent, S-(4,4,5,5,5-pentafluoroamyl)isothiourea mesylate, which is synthesized by mesylating 4,4,5,5,5-pentafluoropentanol followed by reaction with thiourea. This solid reagent is then coupled with the steroid mesylate intermediate in a heated solvent system. The choice of base and solvent is critical; experimental data suggests that acetonitrile and ethanol provide optimal results, with potassium carbonate and triethylamine serving as effective promoters. Following the coupling, the reaction mixture undergoes a straightforward aqueous workup to remove inorganic salts and urea byproducts. The crude thioether intermediate can then be carried forward to deprotection and oxidation steps to yield the final active pharmaceutical ingredient. For a detailed, step-by-step breakdown of the standardized operating procedures and specific molar ratios, please refer to the technical guide below.
- Preparation of the Fluorinated Reagent: Convert 4,4,5,5,5-pentafluoropentanol into its mesylate derivative, followed by reaction with thiourea to generate the stable S-(4,4,5,5,5-pentafluoroamyl)isothiourea mesylate salt.
- Coupling Reaction: React the steroid mesylate intermediate (Formula II) with the isothiourea reagent in a polar solvent like acetonitrile or ethanol, using a base such as potassium carbonate or triethylamine at reflux temperatures.
- Workup and Purification: Perform aqueous extraction to remove inorganic salts, followed by solvent evaporation and optional column chromatography or crystallization to isolate the high-purity thioether intermediate (Formula IV).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this isothiourea-based methodology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of raw material handling and storage. Unlike volatile thiols that require specialized tankers and strict temperature controls, the isothiourea mesylate reagent is a stable solid that can be shipped and stored using standard logistics channels. This inherent stability reduces the risk of supply disruptions caused by reagent degradation during transit, ensuring a more reliable flow of materials into the production facility. Furthermore, the elimination of noxious odors and hazardous gas evolution significantly lowers the operational costs associated with environmental health and safety (EHS) compliance. Facilities do not need to invest in expensive scrubbing towers or negative pressure containment units, leading to substantial capital savings and reduced overheads.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the simplification of the workflow and the reduction in waste treatment costs. By avoiding the use of unstable sodium mercaptides, the process eliminates the need for complex quenching procedures and extensive purification steps to remove sulfur-based impurities. The high conversion rates observed in the patent examples, often exceeding 90%, mean that less raw material is wasted, directly improving the cost of goods sold (COGS). Additionally, the use of commodity solvents like ethanol and acetonitrile, which are readily available and inexpensive compared to specialized ethers or chlorinated solvents, further contributes to cost efficiency. The streamlined workup, involving simple extractions and evaporations, reduces energy consumption and labor hours per batch, resulting in a leaner and more profitable manufacturing operation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reagents involved. The isothiourea salt has a long shelf life and is not sensitive to ambient moisture or oxygen, unlike traditional thiol reagents which often require cold chain shipping and inert atmosphere storage. This flexibility allows procurement teams to source materials from a broader range of suppliers without compromising quality, mitigating the risk of single-source dependency. The scalability of the reaction, demonstrated by its successful execution in multi-liter batches in the patent examples, assures supply chain planners that the process can be ramped up to meet commercial demand without encountering unforeseen engineering bottlenecks. The consistency of the reaction outcome also reduces the likelihood of batch failures, ensuring a steady and predictable output of API intermediates.
- Scalability and Environmental Compliance: From an environmental standpoint, this route aligns well with green chemistry principles. The avoidance of heavy metal catalysts or toxic reagents simplifies the disposal of process waste, reducing the environmental footprint of the manufacturing site. The aqueous workup generates waste streams that are easier to treat biologically or chemically compared to those containing high loads of organic sulfides. The high atom economy of the coupling step ensures that the majority of the starting materials end up in the final product, minimizing the generation of byproduct waste. This environmental compatibility not only aids in meeting regulatory discharge limits but also enhances the corporate sustainability profile, which is increasingly important for partnerships with major pharmaceutical companies committed to ESG goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fulvestrant synthesis route. These insights are derived directly from the experimental data and claims presented in patent CN103788164A, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply chain. The answers highlight the specific advantages of the isothiourea chemistry over legacy methods, focusing on practical aspects such as reagent stability, solvent selection, and impurity management.
Q: Why is the isothiourea mesylate reagent preferred over traditional sodium mercaptides?
A: Traditional sodium mercaptides are often unstable, malodorous, and prone to oxidation, complicating storage and handling. The isothiourea mesylate salt described in CN103788164A is a stable, solid reagent that significantly improves operational safety and simplifies the supply chain logistics for large-scale production.
Q: What solvents are optimal for this coupling reaction?
A: The patent data indicates that polar aprotic and protic solvents such as acetonitrile, ethanol, and mixtures thereof provide superior conversion rates. Acetonitrile and ethanol are particularly favored for their ability to dissolve both the steroid substrate and the ionic reagent while facilitating easy removal during workup.
Q: How does this method impact the impurity profile of the final API?
A: By utilizing a stable sulfur donor rather than reactive free thiols, the formation of disulfide byproducts and oxidation impurities is minimized. This results in a cleaner crude reaction mixture, reducing the burden on downstream purification steps and ensuring higher overall purity for the final fulvestrant API.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fulvestrant Intermediate Supplier
The technological advancements detailed in CN103788164A underscore the complexity and sophistication required to manufacture high-value oncology intermediates like those for fulvestrant. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative patent literature into commercial reality. Our team of process chemists is adept at optimizing reaction conditions, scaling up sensitive transformations, and implementing rigorous quality control measures to ensure product consistency. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch meets the highest industry standards.
We invite you to collaborate with us to leverage this advanced synthesis route for your fulvestrant supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your operational efficiency. Contact us today to discuss how we can support your project with reliable, high-quality intermediates and drive your product development forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
