Advanced Synthesis of Prucalopride Intermediates for Commercial Scale-Up
Advanced Synthesis of Prucalopride Intermediates for Commercial Scale-Up
The pharmaceutical landscape for gastrointestinal motility disorders has been significantly shaped by the development of selective 5-HT4 receptor agonists, with prucalopride succinate standing out as a premier therapeutic agent. As detailed in patent CN103570699A, published in early 2014, a transformative methodology has been established for the preparation of prucalopride and its key intermediates. This intellectual property outlines a robust synthetic pathway that addresses historical inefficiencies in producing this critical benzofuran derivative. For R&D directors and procurement specialists alike, understanding the nuances of this patent is essential for securing a reliable prucalopride intermediate supplier capable of delivering high-purity materials. The disclosed method leverages a strategic nitro-group protection strategy, utilizing 4-nitro-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid as a stable starting material, which fundamentally alters the economic and technical feasibility of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN103570699A, the industry relied on synthetic routes that were fraught with chemical inefficiencies and operational hazards. One conventional pathway, often cited in earlier literature such as Chinese patent CN1071332C, involved the direct condensation of 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid with a piperidine amine derivative. 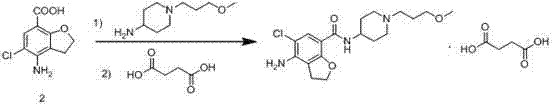 . The fundamental flaw in this approach lies in the reactivity of the free amino group on the benzofuran ring; during the amidation process, this nucleophilic center competes with the intended carboxylic acid activation, leading to polymerization, oligomerization, and complex impurity profiles that are notoriously difficult to purge. Furthermore, alternative routes attempting to construct the side chain via alkylation of a pre-formed benzofuran core often encountered severe steric hindrance and required harsh conditions that degraded the sensitive heterocyclic scaffold. These legacy methods resulted in low overall yields, excessive solvent consumption for purification, and inconsistent batch-to-batch quality, creating significant bottlenecks for any procurement manager seeking cost reduction in pharmaceutical intermediates manufacturing.
. The fundamental flaw in this approach lies in the reactivity of the free amino group on the benzofuran ring; during the amidation process, this nucleophilic center competes with the intended carboxylic acid activation, leading to polymerization, oligomerization, and complex impurity profiles that are notoriously difficult to purge. Furthermore, alternative routes attempting to construct the side chain via alkylation of a pre-formed benzofuran core often encountered severe steric hindrance and required harsh conditions that degraded the sensitive heterocyclic scaffold. These legacy methods resulted in low overall yields, excessive solvent consumption for purification, and inconsistent batch-to-batch quality, creating significant bottlenecks for any procurement manager seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The breakthrough presented in the patent data introduces a paradigm shift by reversing the order of functional group manipulation, specifically by employing a nitro-group surrogate for the final amino functionality. Instead of struggling with a reactive amino-acid, the novel process utilizes 4-nitro-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid, a chemically robust building block that withstands vigorous coupling conditions without degradation. 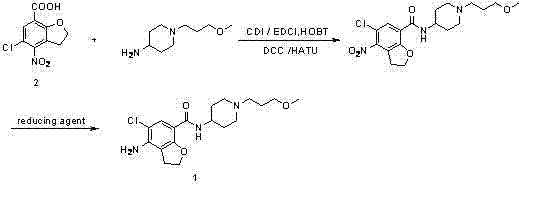 . This strategic modification allows for a clean, high-yielding amide bond formation using standard coupling agents like CDI or EDCI, completely bypassing the side reactions associated with the free amine. Once the amide backbone is securely established, the nitro group is selectively reduced to the amine in a dedicated second step. This decoupling of the amidation and reduction steps not only simplifies the purification workflow but also dramatically improves the mass balance of the overall process, offering a clear pathway for the commercial scale-up of complex benzofuran derivatives.
. This strategic modification allows for a clean, high-yielding amide bond formation using standard coupling agents like CDI or EDCI, completely bypassing the side reactions associated with the free amine. Once the amide backbone is securely established, the nitro group is selectively reduced to the amine in a dedicated second step. This decoupling of the amidation and reduction steps not only simplifies the purification workflow but also dramatically improves the mass balance of the overall process, offering a clear pathway for the commercial scale-up of complex benzofuran derivatives.
Mechanistic Insights into CDI-Mediated Amidation and Nitro Reduction
The core of this synthetic success lies in the precise mechanistic control exerted during the coupling phase. The patent highlights the use of carbonyldiimidazole (CDI) as a superior activating agent compared to traditional phosphonium or uronium salts. In the first step, CDI reacts with the carboxylic acid of the nitro-benzofuran to form a highly reactive acyl-imidazole intermediate. This species is electrophilic enough to react rapidly with the sterically hindered 1-(3-methoxypropyl)-4-piperidinamine, yet stable enough to minimize racemization or hydrolysis side products. The reaction proceeds optimally in polar aprotic solvents like tetrahydrofuran (THF) or DMF at elevated temperatures ranging from 50°C to 90°C. The choice of THF is particularly advantageous for industrial applications due to its ease of removal and recycling. Following the coupling, the isolation of the nitro-intermediate is achieved simply by aqueous quenching, where the product precipitates as a high-purity solid, often exceeding 95% yield, thereby eliminating the need for column chromatography.
The subsequent reduction of the nitro group to the aniline derivative is equally critical for maintaining product integrity. While catalytic hydrogenation using Pd/C under pressure is a viable option, the patent details a highly effective chemical reduction protocol using hydrazine hydrate in the presence of activated carbon and ferric chloride. This system generates nascent hydrogen in situ, which selectively reduces the aromatic nitro group without affecting the newly formed amide bond or the chloro-substituent on the ring. This chemoselectivity is paramount; harsher reducing conditions could potentially dehalogenate the molecule or hydrolyze the amide linkage. By optimizing the stoichiometry of hydrazine and the loading of the iron catalyst, the process achieves conversion rates that rival hydrogenation but with significantly lower capital expenditure on high-pressure equipment. This mechanistic elegance ensures that the final prucalopride precursor meets stringent purity specifications required for downstream salt formation.
How to Synthesize Prucalopride Intermediate Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters outlined in the patent embodiments to ensure reproducibility and safety. The process begins with the activation of the nitro-acid in dry THF, followed by the controlled addition of the piperidine amine. Temperature control is vital during the exothermic coupling phase to prevent thermal runaway. Once the nitro-amide is isolated, the reduction step must be monitored closely via TLC or HPLC to prevent over-reduction or byproduct formation. The detailed standardized operating procedures for scaling this reaction from gram to kilogram quantities are provided below to assist technical teams in process validation.
- Dissolve 4-nitro-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid in THF and activate with CDI at 80°C before adding the piperidine amine.
- Isolate the nitro-intermediate solid by filtration after quenching with water.
- Reduce the nitro group to an amino group using hydrazine hydrate with FeCl3/C or Pd/C hydrogenation to obtain the final amine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain heads and procurement managers, the adoption of the methodology described in CN103570699A offers tangible strategic advantages beyond mere chemical curiosity. The primary benefit is the drastic simplification of the purification train. Because the nitro-intermediate precipitates cleanly from the reaction mixture, the reliance on expensive silica gel chromatography or complex crystallization sequences is virtually eliminated. This reduction in downstream processing directly translates to lower manufacturing costs and shorter cycle times. Furthermore, the starting materials, specifically the nitro-benzofuran acid and the methoxy-propyl-piperidine, are commercially available or easily synthesized from commodity chemicals, ensuring a stable and resilient supply chain that is not dependent on exotic or single-source reagents.
- Cost Reduction in Manufacturing: The implementation of this route eliminates the need for expensive protecting group strategies that were previously necessary to mask the reactive amino group during coupling. By utilizing the nitro group as a latent amine, the process removes at least two synthetic steps (protection and deprotection) from the traditional workflow. This step economy significantly reduces the consumption of reagents, solvents, and labor hours. Additionally, the high yields reported (up to 95% for coupling and 90% for reduction) mean that less raw material is wasted, directly improving the cost of goods sold (COGS) and allowing for more competitive pricing models in the global market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to higher batch success rates and reduced risk of production delays. Unlike processes requiring cryogenic temperatures or ultra-high vacuum, this synthesis operates at moderate temperatures (50-90°C) and atmospheric or low pressure. This operational flexibility allows manufacturers to utilize standard glass-lined or stainless steel reactors without specialized modifications. Consequently, lead times for high-purity pharmaceutical intermediates can be significantly shortened, as production slots are not bottlenecked by equipment availability or complex setup requirements, ensuring a continuous flow of material to downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the shift towards hydrazine-based reduction or mild hydrogenation reduces the generation of heavy metal waste associated with stoichiometric metal reductions (like iron/acetic acid slurries which produce massive iron oxide sludge). The solvents employed, primarily THF and ethanol, are well-understood and can be efficiently recovered and recycled through distillation, minimizing the facility's environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and permits for expansion, making the commercial scale-up of complex benzofuran derivatives a sustainable long-term investment for forward-thinking chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of prucalopride intermediates based on the patented technology. These insights are derived directly from the experimental data and claims within CN103570699A, providing clarity on process capabilities and quality assurance measures. Understanding these details is crucial for partners evaluating the feasibility of integrating this supply stream into their existing manufacturing networks.
Q: Why is the nitro-precursor route preferred over the amino-precursor route for prucalopride?
A: The amino-precursor route suffers from significant side reactions during the amidation step due to the nucleophilicity of the free amino group on the benzofuran ring. The nitro-precursor route avoids this by keeping the group inert until the final reduction step, resulting in much higher purity and yields (up to 95%).
Q: What are the optimal conditions for the reduction step in this synthesis?
A: While catalytic hydrogenation (Pd/C) is effective, the patent highlights a highly efficient chemical reduction using hydrazine hydrate with activated carbon and iron(III) chloride in ethanol or THF, achieving yields around 90% under mild conditions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like THF and ethanol, avoids extreme temperatures (50-90°C), and uses robust reagents like CDI and hydrazine, making it highly scalable and safe for multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prucalopride Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields observed in the lab are faithfully reproduced in the plant. We have invested heavily in state-of-the-art analytical infrastructure, including rigorous QC labs equipped with HPLC, GC-MS, and NMR, to guarantee that every batch of prucalopride intermediate meets stringent purity specifications and complies with international regulatory standards. Our commitment to quality ensures that your downstream API synthesis proceeds without interruption or impurity-related failures.
We invite you to collaborate with us to optimize your supply chain for this critical gastrointestinal medication. Whether you require custom synthesis adaptations or bulk procurement of the nitro-intermediate or the final amine, our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on transparent, data-driven insights into our manufacturing capabilities.
