Advanced Tungsten-Catalyzed Isomerization for High-Purity Allyl Alcohol Manufacturing
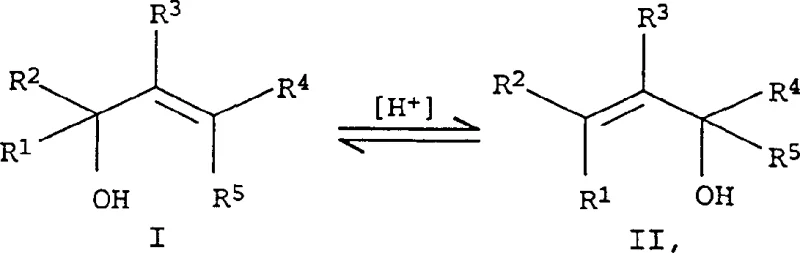
The chemical landscape of industrial organic synthesis is constantly evolving, driven by the relentless demand for higher purity intermediates and more efficient manufacturing pathways. Patent CN1171838C introduces a significant advancement in the isomerization of allyl alcohols, specifically targeting the conversion of primary or secondary precursors into valuable tertiary products. This technology leverages tungsten oxo(VI) complexes modified with specific amino alcohol ligands to achieve superior reaction kinetics and selectivity. For industries relying on terpene alcohols, such as the fragrance and vitamin sectors, this represents a pivotal shift away from older, less efficient catalytic systems. The ability to control the 1,3-migration of the hydroxyl group with minimal by-product formation addresses long-standing challenges in process chemistry. By optimizing the ligand environment around the tungsten center, manufacturers can now access higher conversions without compromising the integrity of the sensitive allylic structure. This report analyzes the technical merits and commercial implications of this patented methodology for global procurement and R&D strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isomerization of allyl alcohols was predominantly achieved through acid-catalyzed processes, which, while conceptually simple, suffer from severe practical drawbacks in an industrial setting. The presence of strong acids often promotes unwanted side reactions, including dehydration and cyclization, which drastically reduce the yield of the desired target molecule. These side pathways not only consume valuable raw materials but also generate complex impurity profiles that are difficult and costly to separate during downstream purification. Furthermore, earlier transition metal catalysts, such as those based on molybdenum or vanadium, often exhibited unsatisfactory reaction results or required harsh conditions that degraded product quality. Even initial tungsten-based systems, while showing promise, frequently struggled with low conversion rates when attempting to drive the equilibrium towards tertiary alcohols like linalool. The inability to efficiently manage the equilibrium position without excessive energy input or prolonged reaction times has been a persistent bottleneck. Consequently, manufacturers have faced inflated production costs and inconsistent supply quality due to these inherent chemical limitations.
The Novel Approach
The innovative method described in the patent overcomes these historical barriers by introducing amino alcohols as critical co-ligands within the tungsten oxo(VI) catalyst system. This modification fundamentally alters the electronic and steric environment of the active catalytic site, enabling a much more efficient isomerization pathway. Unlike previous methods that relied solely on simple nitrogenous bases, the incorporation of structures like triethanolamine or 1-amino-2-propanol significantly accelerates the rate of equilibrium establishment. This allows the reaction to proceed with higher activity even at moderate temperatures, reducing the thermal stress on the reactants and minimizing thermal degradation. The result is a process that maintains high selectivity for the desired tertiary allyl alcohol while achieving substantially higher conversion levels of the precursor. This breakthrough effectively decouples the trade-off between reaction speed and product purity, offering a robust solution for high-volume manufacturing. It provides a clear technical advantage for producing key fragrance ingredients and pharmaceutical intermediates with greater economic efficiency.
Mechanistic Insights into Tungsten-Catalyzed Isomerization
The core of this technological advancement lies in the precise coordination chemistry of the tungsten oxo(VI) complex when paired with specific amino alcohol ligands. The catalyst functions by facilitating the reversible 1,3-shift of the hydroxyl group along the carbon chain, a transformation that is thermodynamically controlled. The presence of the amino alcohol ligand, which contains both nitrogen and oxygen donor atoms, stabilizes the tungsten center and enhances its Lewis acidity in a controlled manner. This stabilization is crucial for activating the allylic substrate without triggering the decomposition pathways common in acid catalysis. The ligand structure allows for fine-tuning of the catalyst's properties, where the ratio of ligand to metal can be adjusted to balance reaction rate against selectivity. For instance, smaller amounts of ligand may favor faster rates, while larger amounts ensure higher selectivity, giving process chemists valuable control parameters. Understanding this mechanistic nuance is essential for scaling the process from laboratory benchtop to multi-ton commercial reactors.
Impurity control is another critical aspect where this catalytic system demonstrates superior performance compared to conventional alternatives. In traditional acid-catalyzed routes, the formation of ethers and dehydration products is a major concern that complicates purification and reduces overall mass balance. The tungsten-amino alcohol system significantly suppresses these side reactions, leading to a cleaner crude reaction mixture. This reduction in by-product formation means that less energy and fewer solvents are required for distillation and refining steps. The patent data indicates that low-boiling components, which are often dehydration products, are kept to a minimum, preserving the value of the feedstock. Additionally, the catalyst system allows for the removal of water generated during catalyst formation or reaction, which can otherwise inhibit the isomerization equilibrium. By managing water content through inert gas stripping or reactive distillation, the process maintains high efficiency throughout the batch cycle. This level of control over the reaction environment is vital for meeting the stringent purity specifications required by downstream applications in fine chemicals.
How to Synthesize Linalool Efficiently
Implementing this isomerization technology requires a systematic approach to catalyst preparation and reaction management to fully realize its potential benefits. The process begins with the generation of the active tungsten species, which can be prepared in situ or pre-formed before introduction to the reactor. Precise control over the molar ratios of the tungsten source, the nitrogenous base, and the amino alcohol ligand is essential to optimize the catalytic activity for the specific substrate being processed. Operators must ensure that the reaction environment is free from moisture and oxygen contaminants that could deactivate the sensitive metal complex. Once the catalyst is active, the precursor allyl alcohol is introduced, and the mixture is heated to the optimal temperature range defined by the patent parameters. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the tungsten oxo(VI) complex catalyst, optionally incorporating amino alcohol ligands such as triethanolamine or 1-amino-2-propanol to enhance activity.
- Mix the precursor allyl alcohol, such as geraniol or nerol, with the catalyst solution in a reaction vessel equipped for heating and inert gas flow.
- Heat the mixture to a temperature between 150°C and 250°C while maintaining an inert atmosphere to drive the equilibrium towards the tertiary product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced isomerization technology translates into tangible strategic advantages beyond mere technical specifications. The primary benefit lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures and enhanced supply reliability. By minimizing the formation of difficult-to-remove by-products, the downstream purification burden is drastically lowered, leading to faster batch turnover times. This efficiency gain allows manufacturers to respond more agilely to market fluctuations and urgent customer demands without compromising on quality standards. Furthermore, the robustness of the tungsten catalyst system implies a more stable production schedule with fewer interruptions caused by catalyst failure or inconsistent reaction outcomes. These factors collectively contribute to a more resilient supply chain capable of sustaining long-term contracts with key global partners.
- Cost Reduction in Manufacturing: The elimination of expensive and complex purification steps required to remove acid-catalyzed by-products results in substantial cost savings throughout the production lifecycle. By achieving higher selectivity, the consumption of raw materials per unit of finished product is optimized, reducing the overall cost of goods sold. The ability to operate at moderate temperatures also lowers energy consumption compared to processes requiring extreme thermal conditions. Additionally, the extended utility of the catalyst system reduces the frequency of catalyst replenishment, further driving down variable costs. These cumulative efficiencies allow for a more competitive pricing structure in the global marketplace without sacrificing margin integrity.
- Enhanced Supply Chain Reliability: The improved conversion rates and reaction stability ensure a consistent output of high-purity intermediates, mitigating the risk of supply shortages. Because the process is less sensitive to minor variations in operating conditions, it offers greater predictability in production planning and inventory management. The compatibility of this method with continuous processing techniques, such as reactive distillation, enables manufacturers to scale up production volumes seamlessly to meet surging demand. This scalability ensures that buyers can secure reliable long-term supplies of critical fragrance and pharmaceutical intermediates. Consequently, partners can build more robust inventory strategies with reduced safety stock requirements.
- Scalability and Environmental Compliance: The reduction in hazardous by-products and the potential for solvent-free or low-solvent operation align with increasingly strict environmental regulations and sustainability goals. The process generates less waste stream volume, simplifying effluent treatment and lowering the environmental footprint of the manufacturing facility. This compliance advantage reduces regulatory risk and avoids potential fines or shutdowns associated with non-compliant waste disposal. Moreover, the ease of scaling this chemistry from pilot plants to full commercial production minimizes the technical risk associated with technology transfer. Companies can confidently invest in capacity expansion knowing that the underlying chemistry is proven to perform reliably at large scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tungsten-catalyzed isomerization technology. These answers are derived directly from the patent specifications and are intended to clarify the operational capabilities and limitations of the process. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines. The responses cover aspects ranging from catalyst composition to process adaptability, providing a comprehensive overview for decision-makers.
Q: What distinguishes this tungsten catalyst system from traditional acid catalysis?
A: Unlike traditional acid catalysis which often leads to significant dehydration and cyclization by-products, this tungsten oxo(VI) complex system with amino alcohol ligands offers superior selectivity for the desired isomerization without promoting destructive side reactions.
Q: Can this process be adapted for continuous large-scale production?
A: Yes, the patent explicitly describes that the process can be carried out continuously or discontinuously, and reactive distillation techniques can be employed to remove low-boiling products like linalool, thereby shifting the equilibrium and enhancing space-time yield for commercial scale-up.
Q: How does the addition of amino alcohols impact reaction efficiency?
A: The inclusion of amino alcohols as additional nitrogen-containing ligands significantly increases the reaction rate and conversion levels compared to using tungsten catalysts with only standard nitrogenous bases, allowing for high activity even at moderate temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Linalool Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of deploying cutting-edge synthetic methodologies to deliver superior chemical intermediates to the global market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the tungsten-catalyzed isomerization process are implemented with precision. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that verify every batch against the highest international standards. Our commitment to technical excellence means that we do not just supply chemicals; we provide validated solutions that enhance your downstream manufacturing efficiency. By leveraging our deep expertise in fine chemical synthesis, we help our partners navigate the complexities of modern supply chains with confidence and reliability.
We invite you to engage with our technical procurement team to discuss how this advanced isomerization technology can be tailored to your specific production requirements. Request a Customized Cost-Saving Analysis to understand the potential economic impact of switching to this high-efficiency catalytic system. Our experts are ready to provide specific COA data and route feasibility assessments to support your internal validation processes. Partnering with us ensures access to a stable supply of high-purity allyl alcohols backed by a commitment to continuous process improvement and innovation. Contact us today to initiate a dialogue about optimizing your fragrance or pharmaceutical intermediate supply chain.
