Scalable Synthesis of Vonoprazan Intermediate via Telescoped Toluene Process
Introduction to the Novel Synthetic Route
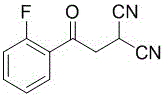
The pharmaceutical industry constantly seeks robust, scalable, and environmentally friendly pathways for producing high-value active pharmaceutical ingredient (API) precursors. Patent CN113896655A introduces a significant advancement in the manufacturing of Vonoprazan intermediates, specifically targeting the key building block known chemically as 3,3-dicyano-1-(2-fluorophenyl)propan-1-one. This innovation addresses critical bottlenecks in traditional synthesis by employing a telescoped four-step reaction sequence conducted entirely within a toluene medium. By eliminating the need to isolate unstable and odorous liquid intermediates, this method not only streamlines the operational workflow but also drastically reduces solvent consumption and waste generation. The technical breakthrough lies in the identification of toluene as a universal solvent that maintains high solubility for all transient species while facilitating efficient phase separations during workup. This approach ensures a mass conversion rate of raw materials reaching up to 90 percent and an overall four-step mass yield exceeding 70 percent, setting a new benchmark for efficiency in gastrointestinal drug intermediate manufacturing.
For procurement specialists and supply chain managers, the implications of this technology are profound. The ability to produce high-purity intermediates with minimal unit operations translates directly into reduced capital expenditure (CAPEX) and operational expenditure (OPEX). The process avoids the handling of hazardous isolated intermediates, thereby enhancing workplace safety and reducing the regulatory burden associated with volatile organic compound (VOC) emissions. Furthermore, the final crude product achieves a purity of more than 95 percent, which can be effortlessly upgraded to over 99.5 percent through a simple methanol pulping procedure. This level of quality control is essential for downstream API synthesis, ensuring that the final Vonoprazan fumarate meets stringent pharmacopeial standards. As a reliable supplier of complex pharmaceutical intermediates, understanding and adopting such patented methodologies is crucial for maintaining competitiveness in the global market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Vonoprazan intermediates often suffer from significant inefficiencies related to intermediate isolation and solvent switching. In conventional processes, the intermediates corresponding to Formula I, Formula II, and Formula III are typically isolated as liquids. These compounds are characterized by unpleasant, irritating odors and physical properties that make concentration and purification difficult and energy-intensive. The necessity to remove solvents between each step not only prolongs the production cycle but also leads to substantial material loss due to adhesion to reactor walls and filtration media. Moreover, the thermal instability of some of these intermediates during concentration can lead to decomposition, generating complex impurity profiles that are challenging to remove in later stages. This fragmented approach increases the demand for multiple reactors, extensive solvent recovery systems, and prolonged labor hours, all of which inflate the cost of goods sold (COGS) and extend the lead time for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN113896655A revolutionizes this landscape by implementing a continuous, telescoped synthesis strategy. By utilizing toluene as the sole reaction medium for the first three steps, the process allows the reaction mixture to proceed directly from condensation to hydrolysis and finally to halogenation without isolating the intermediate species. This "one-pot" philosophy minimizes exposure to air and moisture, reducing the risk of side reactions such as hydrolysis of the acid chloride or oxidation of sensitive functional groups. The patent data indicates that attempting this telescoping in other solvents like dichloromethane or tetrahydrofuran results in incomplete conversion and lower product purity, highlighting the unique solvation properties of toluene in this specific chemical environment. Consequently, the novel approach significantly shortens the process period, reduces the volume of waste liquid requiring treatment, and simplifies the overall equipment train, making it highly suitable for industrial production.
Mechanistic Insights into the Telescoped Condensation and Substitution
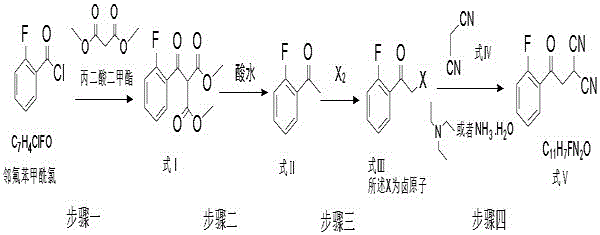
The chemical elegance of this synthesis lies in the seamless integration of four distinct reaction mechanisms. The process initiates with the condensation of o-fluorobenzoyl chloride and dimethyl malonate, likely proceeding through a nucleophilic acyl substitution followed by enolate formation to yield the beta-keto ester (Formula I). This step is exothermic and requires careful temperature control to prevent runaway reactions. Subsequently, the addition of 6 mol/L hydrochloric acid facilitates the hydrolysis of the ester groups and subsequent decarboxylation, transforming Formula I into the methyl ketone (Formula II). The patent emphasizes that maintaining the temperature between 15-25°C during this acidification is critical; deviations can lead to reverse reactions or incomplete hydrolysis, negatively impacting the yield of the final intermediate. The precise control of acid concentration ensures that the protonation state of the intermediates favors the forward reaction pathway while minimizing the formation of polymeric byproducts.
Following the formation of the ketone, the reaction mixture undergoes alpha-halogenation using elemental bromine to generate the alpha-bromo ketone (Formula III). This electrophilic substitution is conducted at 10-25°C to ensure mono-bromination and prevent poly-halogenation, which would act as a difficult-to-remove impurity. The final step involves the nucleophilic displacement of the bromine atom by the carbanion of malononitrile (Formula IV). This substitution is catalyzed by a base, such as ammonia water or triethylamine, at low temperatures (-10 to 10°C). The low temperature is vital not only for controlling the exotherm but also for promoting the precipitation of the final product (Formula V) from the toluene solution, thereby driving the equilibrium towards completion. This mechanistic understanding allows for precise optimization of stoichiometry and addition rates, ensuring that the mass yield remains consistently above 70 percent even on a multi-ton scale.
How to Synthesize 3,3-Dicyano-1-(2-fluorophenyl)propan-1-one Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding solvent ratios, temperature gradients, and reagent addition rates described in the patent. The process begins with the preparation of a dimethyl malonate solution in toluene, into which o-fluorobenzoyl chloride is slowly dripped to manage the exotherm. Following the condensation, the reaction mass is treated directly with hydrochloric acid for hydrolysis, followed by neutralization and bromination without intermediate workups. The detailed standardized operating procedures (SOPs) for scaling this reaction from laboratory to commercial volumes involve specific controls for phase separation and washing efficiency to remove inorganic salts and residual acids. For a comprehensive guide on the exact molar ratios, stirring speeds, and filtration protocols required to achieve the reported 99.5 percent purity, please refer to the technical instructions below.
- Condense o-fluorobenzoyl chloride with dimethyl malonate in toluene to form the beta-keto ester intermediate (Formula I).
- Perform acid hydrolysis and decarboxylation directly in the reaction mixture using 6 mol/L hydrochloric acid to generate the ketone (Formula II).
- Conduct alpha-halogenation using bromine in the organic layer to produce the alpha-halo ketone (Formula III).
- React the halogenated intermediate with malononitrile (Formula IV) using ammonia or triethylamine as a base to yield the final nitrile intermediate (Formula V).
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this telescoped toluene-based process offers compelling advantages in cost structure and supply reliability. The elimination of intermediate isolation steps drastically reduces the consumption of auxiliary solvents and the energy required for distillation and drying. This reduction in unit operations translates to significant cost savings in manufacturing, as fewer reactor turnovers are needed to produce the same amount of final product. Furthermore, the use of toluene, a commodity solvent with well-established recovery infrastructure, allows for high-efficiency recycling, further lowering the variable costs associated with raw material procurement. The robustness of the process, evidenced by high conversion rates and consistent purity across different scales, minimizes the risk of batch failures, ensuring a steady and predictable supply of critical intermediates for downstream API production.
- Cost Reduction in Manufacturing: The telescoped nature of the synthesis eliminates the need for multiple isolation, drying, and redissolution steps, which are traditionally the most expensive parts of fine chemical manufacturing. By avoiding the handling of odorous and unstable liquid intermediates, the process reduces labor costs and containment requirements. Additionally, the high atom economy of the condensation and substitution steps, combined with the ability to recover and reuse toluene, results in a substantially lower environmental footprint and waste disposal cost. This efficiency allows suppliers to offer competitive pricing without compromising on the rigorous quality standards demanded by the pharmaceutical industry.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as o-fluorobenzoyl chloride and dimethyl malonate ensures that the supply chain is not vulnerable to shortages of exotic reagents. The process has been validated at scales up to 1000 kg, demonstrating that it can be seamlessly transferred from pilot plants to full-scale commercial production without loss of efficiency. This scalability guarantees that suppliers can meet large-volume orders with short lead times, providing security of supply for pharmaceutical companies facing tight production schedules. The simplified workflow also reduces the complexity of logistics, as fewer intermediate shipments and storage requirements are needed.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing solvent usage and waste generation. The ability to conduct multiple reaction steps in a single vessel reduces the equipment footprint and energy consumption per kilogram of product. Moreover, the high purity of the crude product (>95 percent) reduces the burden on downstream purification processes, such as chromatography or recrystallization, which often generate significant amounts of solvent waste. This alignment with environmental, social, and governance (ESG) goals makes the technology attractive for companies aiming to reduce their carbon footprint and comply with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Vonoprazan intermediates using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in CN113896655A, providing clarity on the feasibility and benefits of this route. Understanding these details helps stakeholders make informed decisions about integrating this technology into their supply chains.
Q: Why is toluene preferred over other solvents for this synthesis?
A: Toluene enables a telescoped process where intermediates I, II, and III remain in solution without isolation. Other solvents like dichloromethane or THF led to incomplete conversion and lower purity in patent experiments.
Q: What is the achieved purity of the crude Vonoprazan intermediate?
A: The patent reports a crude product purity of greater than 95%, which can be further refined to over 99.5% with a maximum single impurity of less than 0.2% via methanol pulping.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent demonstrates successful scale-up to 1000 kg batches (Example 4), maintaining high yields (>70% over 4 steps) and consistent quality, proving its viability for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Dicyano-1-(2-fluorophenyl)propan-1-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the synthesis of life-saving medications like Vonoprazan. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patented processes like CN113896655A are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify every batch against the highest industry standards. Our commitment to technical excellence means we can navigate the complexities of telescoped syntheses, managing exotherms and phase separations with precision to deliver products with consistent impurity profiles and reliable performance in your downstream reactions.
We invite you to collaborate with us to optimize your supply chain for Vonoprazan production. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your project timelines and budgetary goals. Together, we can ensure a stable, cost-effective, and high-quality supply of this essential pharmaceutical intermediate.
