Revolutionizing Valsartan Production: A Low-Toxicity, Solvent-Free Synthetic Route for Commercial Scale
Introduction to Advanced Valsartan Manufacturing Technologies
The pharmaceutical landscape for Angiotensin II receptor blockers continues to evolve, driven by the urgent need for greener, safer, and more cost-effective manufacturing processes. Patent CN102010381B introduces a transformative methodology for the preparation of Valsartan, a critical cardiovascular medication, by fundamentally reengineering the tetrazole ring formation step. Historically, the synthesis of this pharmacophore has been plagued by the use of highly toxic organotin reagents and complex protection-deprotection sequences that burden both the environment and the production budget. This innovative protocol substitutes hazardous tributyltin compounds with trimethylsilyl azide (TMSA) and employs a solvent-free reaction system catalyzed by tetrabutylammonium fluoride (TBAF). By shifting the paradigm from toxic heavy metal chemistry to silicon-based azide transfer, this technology offers a robust pathway for reliable pharmaceutical intermediate supplier networks seeking to modernize their production capabilities while adhering to stringent global safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for Valsartan, as documented in prior art such as Organic Process R&D 2007 and various international patents, heavily rely on organotin chemistry to construct the essential tetrazole moiety. These legacy methods typically utilize tributyltin chloride or azide, reagents known for their extreme toxicity and persistence in the environment, necessitating elaborate and costly purification steps to ensure residual tin levels meet regulatory limits for active pharmaceutical ingredients. Furthermore, conventional approaches often require the use of expensive starting materials where the tetrazole ring is pre-installed and protected, adding multiple synthetic steps including protection and subsequent deprotection which drastically reduce overall atom economy. The reliance on high-boiling solvents like xylene at reflux temperatures further exacerbates energy consumption and complicates solvent recovery systems, creating a bottleneck for efficient commercial scale-up of complex pharmaceutical intermediates.
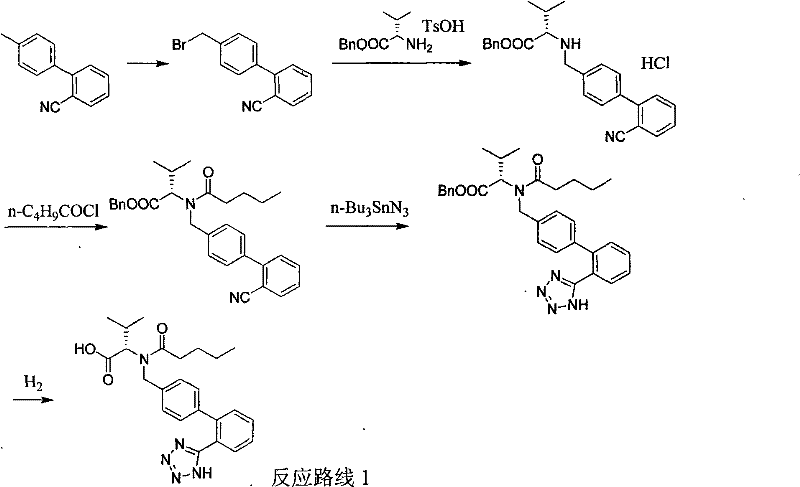
The Novel Approach
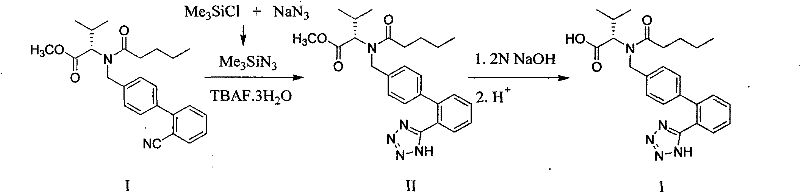
In stark contrast to these cumbersome legacy protocols, the novel approach detailed in CN102010381B streamlines the synthesis by introducing the tetrazole ring directly onto a cyano-precursor using TMSA under solvent-free conditions. This methodology eliminates the need for toxic organotin catalysts entirely, replacing them with a safer silicon-based azide source that generates benign byproducts. The absence of bulk solvent during the critical cyclization step not only simplifies the reactor setup but also intensifies the reaction rate through higher effective concentrations of reactants. By avoiding the use of protected tetrazole starting materials, the process reduces the total number of unit operations, thereby minimizing material loss and handling time. This direct cyclization strategy represents a significant leap forward in process chemistry, offering a cleaner, more direct route to the target molecule that aligns perfectly with modern principles of green chemistry and sustainable manufacturing.
Mechanistic Insights into TBAF-Catalyzed Tetrazole Cyclization
The core innovation of this process lies in the mechanism of the [3+2] cycloaddition between the nitrile group of the biphenyl precursor and the azide source, facilitated by the fluoride ion. In the absence of solvent, the tetrabutylammonium fluoride (TBAF) acts as a potent nucleophilic catalyst that activates the trimethylsilyl azide (TMSA) by cleaving the silicon-nitrogen bond to generate a reactive azide species in situ. This activated azide then attacks the electrophilic carbon of the nitrile group, initiating the ring closure to form the tetrazole structure. The solvent-free environment is crucial here, as it prevents the dilution of these reactive intermediates, ensuring that the collision frequency between the nitrile and the azide remains high enough to drive the reaction to completion even at moderate temperatures ranging from 80°C to 140°C. This mechanistic efficiency allows for high conversion rates without the need for harsh Lewis acids or toxic metal promoters that are typically required to activate nitriles in solution-phase reactions.
From an impurity control perspective, this mechanism offers distinct advantages by suppressing side reactions associated with organotin chemistry. Traditional tin-mediated pathways often generate difficult-to-remove tin-containing byproducts and oligomers that can co-elute with the product, complicating downstream purification. The silicon-based pathway, however, generates volatile siloxanes or easily hydrolyzable silyl species that are readily removed during the aqueous workup and acidification steps. Furthermore, the specificity of the fluoride-catalyzed activation ensures that the azide reacts preferentially with the nitrile group rather than other functional groups present on the valine scaffold, such as the amide or ester moieties. This high chemoselectivity results in a cleaner crude product profile, reducing the burden on crystallization steps and ensuring that the final active pharmaceutical ingredient meets rigorous purity specifications with minimal effort.
How to Synthesize Valsartan Efficiently
The implementation of this synthetic route involves a straightforward three-stage sequence that begins with the generation of the azide source, followed by the key cyclization, and concludes with hydrolysis. The process is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions often associated with azide chemistry. The initial preparation of trimethylsilyl azide can be performed in situ or sourced commercially, reacting trimethylchlorosilane with sodium azide in a suitable medium. The subsequent coupling with the valine derivative is the heart of the process, where precise temperature control between 100°C and 120°C optimizes the balance between reaction rate and thermal stability. Detailed standardized operating procedures for scaling this reaction from laboratory to pilot plant are critical to maintaining safety and consistency, particularly when handling azide reagents on a large tonnage scale.
- Preparation of Trimethylsilyl Azide (TMSA): React trimethylchlorosilane with sodium azide in a low-boiling solvent like n-butyl ether or high-boiling silicone oil at elevated temperatures (100°C) to generate the azide source.
- Solvent-Free Tetrazole Cyclization: Mix the cyano-biphenyl valine ester precursor with TMSA and a catalytic amount of tetrabutylammonium fluoride (TBAF). Heat the mixture to 100-120°C without additional solvent to form the tetrazole ring.
- Hydrolysis and Purification: Treat the resulting tetrazole ester intermediate with aqueous sodium hydroxide to hydrolyze the methyl ester, followed by acidification and recrystallization to isolate pure valsartan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend far beyond simple yield improvements. The elimination of organotin reagents removes a major regulatory and disposal hurdle, significantly lowering the costs associated with hazardous waste management and environmental compliance. By switching to a solvent-free protocol for the key step, manufacturers can drastically reduce the volume of organic solvents required, leading to substantial cost savings in solvent purchase, recovery, and disposal. This reduction in material intensity directly impacts the cost of goods sold (COGS), making the final API more competitive in the global market. Additionally, the simplified workflow reduces the overall cycle time per batch, allowing facilities to increase throughput without requiring capital investment in new reactor trains, thereby enhancing supply chain resilience and responsiveness to market demand fluctuations.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic tributyltin compounds with cost-effective trimethylsilyl azide creates a direct reduction in raw material expenditure. Furthermore, the avoidance of complex protection and deprotection steps for the tetrazole ring eliminates the need for costly protecting group reagents and the associated labor for additional synthetic stages. The solvent-free nature of the reaction minimizes utility costs related to heating and cooling large volumes of solvent, while also reducing the load on solvent recovery distillation columns. These cumulative efficiencies result in a leaner manufacturing process that delivers significant economic value without compromising on product quality or safety standards.
- Enhanced Supply Chain Reliability: Sourcing high-purity organotin reagents can often be a bottleneck due to strict transportation regulations and limited supplier bases. By shifting to silicon-based reagents and common catalysts like TBAF, the supply chain becomes more robust and less susceptible to disruptions. The starting materials, such as the cyano-biphenyl valine ester, are more readily accessible and stable compared to sensitive tetrazole-protected precursors. This stability allows for longer shelf-life and easier logistics management, ensuring that production schedules can be maintained consistently. The simplified process also reduces the risk of batch failures due to reagent variability, providing a more predictable and reliable supply of critical cardiovascular intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling up organotin chemistry presents significant challenges regarding worker safety and environmental discharge limits. The proposed solvent-free, tin-free route inherently mitigates these risks, facilitating a smoother transition from pilot scale to multi-ton commercial production. The reduction in hazardous waste generation aligns with increasingly strict global environmental regulations, future-proofing the manufacturing site against tighter compliance standards. Moreover, the simplified workup procedure, which relies on standard aqueous extractions and crystallizations, is easily adaptable to existing infrastructure, allowing for rapid capacity expansion. This scalability ensures that the manufacturer can meet growing global demand for Valsartan while maintaining a sustainable and environmentally responsible operational footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. Understanding these details is crucial for R&D teams evaluating process transfer and procurement officers assessing vendor capabilities. The answers provided are derived directly from the technical specifications and experimental data outlined in the patent literature, ensuring accuracy and relevance for decision-makers. These insights highlight the practical advantages of adopting this methodology over traditional approaches, focusing on safety, efficiency, and economic viability in a commercial setting.
Q: How does this new method improve safety compared to traditional organotin routes?
A: Traditional methods rely on toxic tributyltin azide or chloride, posing severe environmental and health risks. This patented approach replaces organotin compounds with trimethylsilyl azide (TMSA), significantly reducing toxicity and eliminating the need for complex heavy metal removal processes.
Q: What are the yield advantages of the solvent-free tetrazole formation?
A: By utilizing a solvent-free system catalyzed by TBAF, the reaction concentration is maximized, leading to improved kinetics. The patent reports yields exceeding 80%, a substantial improvement over the 40-70% yields typical of older organotin-based methodologies.
Q: Is the starting material for this route commercially viable?
A: Yes, the route avoids expensive tetrazole-protected bromomethylbiphenyl starting materials. Instead, it utilizes a cyano-precursor which is more cost-effective and readily available, streamlining the supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Valsartan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced, greener synthetic routes requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102010381B are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace impurities, guaranteeing that every batch of Valsartan intermediate meets the highest international standards. Our commitment to continuous improvement drives us to adopt technologies that enhance safety and sustainability, positioning us as a forward-thinking leader in the pharmaceutical intermediate sector.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can deliver superior value, reliability, and quality for your cardiovascular drug portfolio.
