Advanced Acidic Purification Technology for High-Purity Carbidopa Manufacturing
The global demand for effective Parkinson's disease treatments continues to drive rigorous innovation in the synthesis and purification of key active pharmaceutical ingredients (APIs) and their intermediates. Among these, Carbidopa stands out as a critical dopa decarboxylase inhibitor, essential for enhancing the bioavailability of Levodopa. However, the manufacturing landscape has long been challenged by the chemical instability of Carbidopa during downstream processing. A pivotal advancement in addressing these challenges is detailed in patent CN102432496B, which discloses a novel method for refining Carbidopa that fundamentally alters the purification paradigm. Unlike conventional techniques that struggle with yield loss and impurity control, this technology leverages specific acidic conditions and inert atmospheres to stabilize the molecule during recrystallization. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this patent is crucial for securing a robust supply chain. The process not only simplifies the operational workflow but also ensures that the final product meets stringent international pharmacopeia standards, such as USP34 and EP7, regarding the critical methyldopa impurity limit.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Carbidopa involves the hydrolysis of dimethyl carbidopa, typically using strong acids like hydrobromic acid or hydrochloric acid, followed by isolation at the iso-electric point. As illustrated in the reaction scheme below, the conversion from the dimethyl precursor to the active dihydroxy form is chemically demanding. The primary bottleneck arises during the subsequent purification stage, where traditional methods rely on dissolving the crude product in large volumes of water at a neutral pH range of 6 to 7. Under these conditions, Carbidopa exhibits significant instability, particularly when heated to facilitate dissolution. The molecule tends to undergo thermal decomposition, releasing ammonia and converting into methyldopa, a structurally similar impurity that is difficult to separate. Consequently, conventional water-based recrystallization often achieves a methyldopa removal rate of only approximately 40%, necessitating repeated purification cycles that drastically erode overall yield and inflate production costs. Furthermore, the maximum yield achievable through these older methods rarely exceeds 86%, creating substantial material waste and complicating the economic viability of large-scale manufacturing.
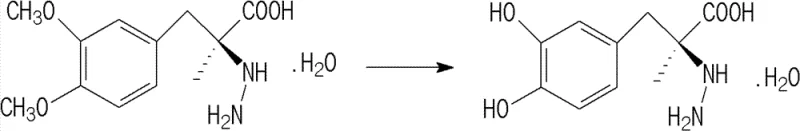
The Novel Approach
The methodology outlined in patent CN102432496B introduces a transformative shift by maintaining an acidic environment throughout the purification process. Instead of adjusting the pH to the iso-electric point, the crude Carbidopa is diluted with water and the pH is rigorously controlled to remain below 6.5, with optimal embodiments operating between pH 1 and 3. This acidic milieu profoundly enhances the chemical stability of Carbidopa, effectively suppressing the decomposition pathway that leads to methyldopa formation even at elevated temperatures ranging from 50°C to 110°C. By preventing the in-situ generation of impurities during the heating phase, the process allows for a highly efficient single-pass purification. The addition of reducing agents or the use of nitrogen protection further safeguards the product against oxidative degradation. This approach not only streamlines the operation by eliminating the need for multiple recrystallizations but also significantly boosts the final yield, with experimental data demonstrating recovery rates approaching 90% while simultaneously driving methyldopa impurity levels well below the 0.5% threshold required by major regulatory bodies.
Mechanistic Insights into Acidic Stabilization and Impurity Control
To fully appreciate the technical superiority of this refinement strategy, one must examine the underlying chemical kinetics governing Carbidopa stability. In aqueous solutions, particularly at higher pH values and temperatures, Carbidopa is prone to a decomposition reaction where the hydrazine moiety breaks down, releasing ammonia gas. This release of ammonia is autocatalytic in nature; as ammonia accumulates, it raises the pH of the solution, which in turn accelerates the decomposition of remaining Carbidopa into methyldopa. The patented process interrupts this vicious cycle by introducing a sufficient concentration of acid—such as sulfuric acid, hydrochloric acid, or organic acids like tartaric acid—to buffer the system. By maintaining a low pH, the equilibrium is shifted away from the decomposition products, ensuring that the Carbidopa molecule remains intact during the critical dissolution and decolorization phases. This mechanistic control is vital for R&D teams focused on impurity profiling, as it addresses the root cause of contamination rather than merely attempting to filter it out post-formation.
Furthermore, the process incorporates robust measures to mitigate oxidative stress, which is another common degradation pathway for catechol-containing compounds like Carbidopa. The protocol mandates the use of nitrogen protection or the addition of specific reducing agents such as sodium bisulfite, vitamin C, or formic acid during the heating and decolorization steps. These agents scavenge dissolved oxygen and free radicals, preventing the oxidation of the phenolic hydroxyl groups. The choice of decolorizing agent, typically activated carbon, is also optimized to operate effectively within this acidic temperature range of 90°C to 100°C. This ensures that colored polymeric impurities are adsorbed without co-adsorbing significant amounts of the active pharmaceutical ingredient. The subsequent crystallization step, conducted by cooling the filtrate to 0°C to 30°C under continued inert conditions, promotes the formation of high-purity crystals while keeping residual impurities in the mother liquor, thereby delivering a product with exceptional chemical integrity.
How to Synthesize Carbidopa Efficiently
Implementing this purification technology requires precise control over reaction parameters to maximize the benefits of acidic stabilization. The process begins with the dilution of the crude Carbidopa material, followed by careful pH adjustment and heating under inert gas. Detailed operational guidelines are essential for reproducibility and scale-up success. The following section outlines the standardized synthesis steps derived from the patent data, providing a clear roadmap for technical teams aiming to adopt this superior purification route.
- Dilute crude Carbidopa with water (10-80x weight), adjust pH to below 6.5 using acid, and heat to 50-110°C under nitrogen protection.
- Add a decolorizing agent (e.g., activated carbon) to the heated system and maintain temperature for 5-60 minutes to remove colored impurities.
- Filter while hot, cool the filtrate to 0-30°C under nitrogen, stir for crystallization, then filter, wash, and vacuum dry to obtain the finished product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this acidic purification method represents a strategic opportunity to optimize cost structures and enhance supply reliability. The traditional reliance on multiple recrystallization cycles to meet purity specifications creates bottlenecks in production scheduling and consumes excessive utilities and solvents. By contrast, the novel approach described in CN102432496B enables the achievement of pharmacopeia-grade purity in a single purification pass. This consolidation of processing steps translates directly into reduced manufacturing lead times and lower operational expenditures. The elimination of redundant cycles means that production capacity can be effectively increased without the need for additional capital investment in equipment, offering a scalable solution for meeting growing market demand for Parkinson's disease medications.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven primarily by the substantial improvement in yield and the reduction of material waste. In conventional processing, the degradation of Carbidopa into methyldopa results in a permanent loss of valuable mass, forcing manufacturers to process larger batches of crude material to obtain the same amount of finished product. By stabilizing the molecule and preventing this decomposition, the new method preserves the mass balance, leading to significantly higher recovery rates. Additionally, the reduction in processing cycles lowers the consumption of water, energy for heating and cooling, and labor hours associated with filtration and drying operations. These cumulative efficiencies result in a markedly lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often jeopardized by complex purification processes that are sensitive to variability and prone to failure. The robustness of the acidic purification method enhances supply chain resilience by simplifying the critical path of production. With fewer unit operations and a wider operating window for temperature and pH control, the risk of batch rejection due to out-of-specification impurity levels is drastically minimized. This reliability ensures consistent on-time delivery to downstream API manufacturers. Furthermore, the raw materials required for this process, such as common mineral or organic acids and activated carbon, are readily available commodities, reducing the risk of supply disruptions associated with specialized reagents. This accessibility supports a stable and predictable manufacturing schedule.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The reduction in the number of recrystallization cycles inherently decreases the volume of wastewater generated, easing the burden on effluent treatment facilities. The use of water as the primary solvent, combined with the absence of toxic organic solvents in the purification step, simplifies regulatory compliance and reduces the environmental footprint of the manufacturing site. The method is designed for easy industrial implementation, with parameters that are straightforward to control in large-scale reactors. This scalability ensures that the technology can be seamlessly transferred from pilot plants to commercial production lines ranging from hundreds of kilograms to multi-ton annual capacities, supporting long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Carbidopa purification technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing clarity for stakeholders evaluating this manufacturing route. Understanding these details is essential for making informed decisions about process adoption and supplier qualification.
Q: Why is acidic pH critical for Carbidopa purification?
A: Carbidopa is chemically unstable in neutral or alkaline aqueous solutions, especially at elevated temperatures, where it decomposes into methyldopa and ammonia. Maintaining a pH below 6.5 (optimally 1-3) suppresses this decomposition pathway, ensuring high recovery yields and minimizing the formation of the critical methyldopa impurity.
Q: How does this method improve methyldopa removal compared to traditional processes?
A: Traditional water-based recrystallization at the iso-electric point (pH 6-7) often results in only about 40% removal of methyldopa impurities, requiring multiple cycles. The patented acidic method stabilizes the molecule during heating, allowing for a single-pass purification that achieves methyldopa levels well below pharmacopeia limits (e.g., <0.5%).
Q: What role do reducing agents play in this synthesis?
A: Reducing agents such as sodium bisulfite, vitamin C, or formic acid are added to prevent oxidative degradation of Carbidopa by atmospheric oxygen during the heating and decolorization steps. This ensures the chemical integrity of the final API intermediate and prevents the formation of oxidation byproducts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbidopa Supplier
The technological advancements detailed in patent CN102432496B underscore the importance of partnering with a manufacturer that possesses both deep chemical expertise and robust production capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of Carbidopa meets the highest international standards. We understand the critical nature of this intermediate in the treatment of Parkinson's disease and are dedicated to providing a supply chain that is both resilient and responsive to the evolving needs of the global pharmaceutical market.
We invite potential partners to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our optimized purification process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your supply of high-purity Carbidopa is secure, cost-effective, and ready for the next generation of therapeutic innovations.
