Advanced Carbidopa Manufacturing: High-Yield Oxaziridine Route for Global Pharma Supply Chains
Introduction to Next-Generation Carbidopa Synthesis
The pharmaceutical landscape for Parkinson's disease treatment relies heavily on the consistent supply of high-quality Carbidopa, a critical peripheral dopa decarboxylase inhibitor. Patent CN102702019B introduces a transformative methodology that addresses long-standing inefficiencies in the industrial production of this vital API intermediate. By leveraging the unique reactivity of oxaziridine derivatives, this novel synthetic pathway bypasses the cumbersome multi-step protections and hazardous reagents characteristic of legacy processes. For R&D directors and procurement strategists, this represents a pivotal shift towards greener, more cost-effective manufacturing that does not compromise on the stringent purity standards required for neurological therapeutics. The transition from traditional hydrazine-based condensations to direct oxaziridine amination marks a significant technological leap, offering a robust framework for securing supply chains against regulatory and operational volatility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Carbidopa has been plagued by low efficiency and significant safety concerns associated with raw material toxicity. The earliest industrial routes, such as the one depicted below, utilized dimethoxymethyldopa hydrochloride (DMMD) as a starting point, necessitating a complex sequence involving urea formation, sodium hypochlorite oxidation, and subsequent hydrazine conversion.
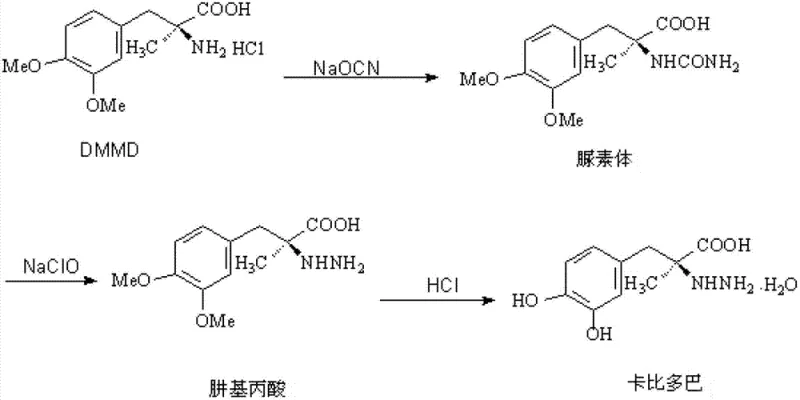
This archaic pathway suffered from abysmal overall yields, often stagnating around merely 20%, rendering it economically unsustainable for modern large-scale demands. Furthermore, alternative routes described in prior art, such as those utilizing boric acid for hydroxyl protection or cyclohexanone-derived oxadiazoles, introduced severe bottlenecks. The reliance on cyclohexanone, a known carcinogen with a high boiling point of 155.6°C, created substantial challenges in solvent recovery and residual control, posing unacceptable risks for GMP-compliant pharmaceutical manufacturing. Additionally, the use of toluene at elevated temperatures (80-85°C) in these older methods frequently led to不合格 solvent residues, necessitating costly and time-consuming purification steps that eroded profit margins.
The Novel Approach
In stark contrast, the innovative process detailed in the patent utilizes 3,3-dimethyloxyaziridine as a potent aminating agent to directly functionalize methyldopa esters. This streamlined approach, illustrated in the reaction scheme below, collapses multiple synthetic steps into a concise two-stage operation involving imido ester formation followed by hydrolysis.
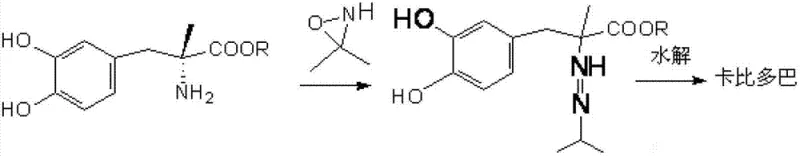
By employing this direct amination strategy, the process achieves remarkable yields of approximately 87% with product purity exceeding 98.5%, drastically outperforming the 40-70% yields of previous generations. The elimination of heavy metal catalysts and carcinogenic ketones simplifies the downstream processing workflow, allowing for a more agile and responsive production schedule. This methodological shift not only enhances the economic viability of Carbidopa manufacturing but also aligns perfectly with modern environmental, health, and safety (EHS) mandates, positioning it as the preferred choice for reliable pharmaceutical intermediate suppliers seeking long-term sustainability.
Mechanistic Insights into Oxaziridine-Mediated Amination
The core of this technological advancement lies in the unique electronic and structural properties of the oxaziridine ring. Oxaziridines are three-membered heterocycles containing carbon, nitrogen, and oxygen, characterized by significant ring strain and a relatively weak nitrogen-oxygen bond. This inherent instability renders them highly reactive towards nucleophiles, functioning dually as either oxidizing or aminating agents depending on the substituents attached to the nitrogen atom. In the specific context of Carbidopa synthesis, the use of 3,3-dimethyloxyaziridine, where the nitrogen substituent is a small hydrogen atom, directs the reactivity specifically towards amination rather than oxidation. When the primary amine group of the methyldopa ester attacks the electrophilic nitrogen of the oxaziridine ring, the N-O bond cleaves, facilitating the formation of the crucial hydrazino linkage found in the final drug molecule.
Furthermore, the choice of solvent plays a critical mechanistic role in controlling the reaction trajectory and minimizing impurity formation. The patent specifies the use of dichloromethane or dichloroethane, solvents that offer superior solubility for both the methyldopa ester substrate and the resulting imido ester intermediate. This enhanced solubility profile allows the reaction to proceed efficiently at significantly lower temperatures, optimally between 25°C and 35°C. Operating at these mild thermal conditions is paramount for suppressing competing side reactions, such as racemization or thermal decomposition, which are prevalent in high-temperature protocols. Consequently, the stereochemical integrity of the chiral center is preserved, ensuring the production of the biologically active (S)-enantiomer with high optical purity, a critical quality attribute for neurological medications.
How to Synthesize Carbidopa Efficiently
The implementation of this oxaziridine-based route requires precise control over reagent stoichiometry and temperature gradients to maximize the formation of the imido ester intermediate. The process begins with the in-situ generation of the oxaziridine reagent, followed by its immediate consumption in the amination step to prevent decomposition. Detailed standard operating procedures regarding mixing rates, addition sequences, and quenching protocols are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the guide below.
- Preparation of 3,3-dimethyloxyaziridine by reacting chloramine with acetone in a dichloromethane medium under alkaline conditions.
- Reaction of the prepared oxaziridine solution with methyldopa methyl ester at controlled temperatures (25-35°C) to form the imido ester intermediate.
- Acidic hydrolysis of the imido ester using dilute hydrochloric acid followed by pH adjustment and crystallization to isolate high-purity Carbidopa.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route translates into tangible strategic benefits that extend beyond simple yield improvements. The restructuring of the synthetic pathway fundamentally alters the cost basis and risk profile of Carbidopa production, offering a more resilient supply model in a volatile global market. By removing dependency on hazardous and difficult-to-source reagents, manufacturers can stabilize their raw material inventory and reduce the overhead associated with specialized waste disposal and safety compliance.
- Cost Reduction in Manufacturing: The elimination of expensive and toxic reagents like cyclohexanone derivatives and the avoidance of high-boiling solvents like toluene significantly lowers the variable cost of goods sold. The simplified workup procedure, which avoids complex extraction and distillation steps required for removing stubborn solvent residues, reduces energy consumption and labor hours per batch. Furthermore, the substantial increase in yield from roughly 40% in older methods to nearly 87% in this new process effectively doubles the output from the same amount of starting material, driving down the unit cost of the active pharmaceutical ingredient dramatically without requiring capital expenditure on new equipment.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, specifically acetone, chloramine, and methyldopa esters, are commodity chemicals with robust and diversified global supply chains. This contrasts sharply with older routes that relied on specialized, custom-synthesized intermediates which were prone to supply disruptions. The mild reaction conditions also reduce the risk of batch failures due to thermal runaway or equipment corrosion, ensuring consistent on-time delivery performance. This reliability is crucial for maintaining continuous production schedules for finished dosage forms, preventing costly stockouts in the Parkinson's disease therapeutic market.
- Scalability and Environmental Compliance: The process is inherently scalable, having been designed with industrial constraints in mind, such as heat transfer efficiency and mixing dynamics. The use of dichloromethane, while requiring careful handling, allows for easier solvent recovery and recycling compared to the high-boiling toluene used in previous iterations, aligning with stricter environmental regulations regarding volatile organic compound (VOC) emissions. The absence of heavy metals and carcinogenic byproducts simplifies the environmental impact assessment and reduces the burden on wastewater treatment facilities, facilitating faster regulatory approvals for new manufacturing sites and ensuring long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxaziridine-mediated synthesis. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this technology for your specific production needs.
Q: What are the primary advantages of the oxaziridine route over traditional Carbidopa synthesis methods?
A: The oxaziridine route eliminates the need for carcinogenic cyclohexanone derivatives and high-boiling solvents like toluene, resulting in significantly higher yields (up to 87%) and superior product purity (>98.5%) with easier solvent removal.
Q: How does this method address impurity control in pharmaceutical manufacturing?
A: By utilizing dichloromethane or dichloroethane as solvents and maintaining reaction temperatures below 40°C, the process minimizes side reactions and thermal degradation, ensuring a cleaner impurity profile compared to high-temperature traditional routes.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the process uses readily available raw materials like acetone and chloramine and operates under mild conditions, making it highly suitable for scale-up from pilot batches to multi-ton commercial production without complex safety constraints.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbidopa Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes requires a partner with deep technical expertise and proven scale-up capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the oxaziridine route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Carbidopa meets the highest international pharmacopoeia standards, providing you with a secure and high-quality supply of this critical intermediate.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless integration of this high-performance intermediate into your global pharmaceutical portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
