Advanced Acidic Recrystallization Technology for Commercial Scale Carbidopa Production
The pharmaceutical industry constantly seeks robust methodologies to enhance the purity and yield of critical active pharmaceutical ingredients (APIs), particularly for treatments managing chronic conditions like Parkinson's disease. A significant technological advancement in this domain is detailed in patent CN102432496A, which discloses a novel method for refining carbidopa. This innovation addresses the longstanding challenge of removing methyldopa, a structurally similar impurity that is difficult to separate using conventional neutral water recrystallization techniques. By shifting the paradigm from neutral to acidic recrystallization conditions, this process ensures the chemical stability of the carbidopa molecule during the critical dissolution phase. For global procurement leaders and R&D directors, understanding this mechanism is vital for securing a reliable carbidopa supplier capable of delivering material that consistently meets stringent pharmacopeia specifications such as USP and EP. The transition to this acidic stabilization protocol represents a major leap forward in process chemistry, offering a pathway to higher quality intermediates with reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of carbidopa crude product has relied heavily on simple recrystallization using water as the sole solvent under neutral or slightly basic conditions. In these traditional processes, the crude material, often obtained from the hydrolysis of dimethyl-carbidopa, is dissolved in large volumes of water at elevated temperatures. However, carbidopa exhibits significant chemical instability in aqueous solutions when the pH value is not strictly controlled, particularly at higher temperatures required for dissolution. Under these neutral conditions, the molecule is prone to spontaneous decomposition, eliminating ammonia to form methyldopa, which is a critical quality attribute impurity. Consequently, conventional methods suffer from low impurity clearance rates, typically removing only about 40% of the methyldopa content in a single pass. This inefficiency forces manufacturers to perform multiple recrystallization cycles to meet the required purity standards of less than 0.5% methyldopa, drastically reducing overall production yield to around 86% and inflating manufacturing costs through increased solvent usage and extended processing time.
The Novel Approach
The innovative approach outlined in the patent data fundamentally alters the physicochemical environment of the recrystallization process to favor product stability over degradation. Instead of relying on neutral water, the novel method introduces a controlled acidic environment by adjusting the pH of the dissolution medium to below 6.5, with optimal results observed between pH 1 and 3. This acidic condition effectively suppresses the ionization and subsequent decomposition of carbidopa, allowing the crude material to dissolve fully at temperatures between 50°C and 110°C without generating additional methyldopa impurities. Furthermore, the process incorporates nitrogen protection or the addition of reducing agents to prevent oxidative degradation, coupled with a hot filtration step to remove insoluble particulates and decolorizing agents. By maintaining the molecule in its stable protonated form throughout the heating and cooling phases, this method achieves a methyldopa removal rate exceeding 90% in a single operation. This breakthrough not only simplifies the workflow but also significantly enhances the final yield, providing a commercially viable route for the cost reduction in pharmaceutical intermediates manufacturing.
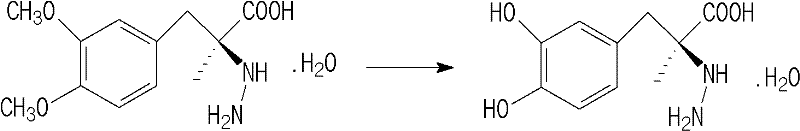
Mechanistic Insights into Acidic Stabilization and Impurity Control
The core mechanistic advantage of this refining technology lies in the pH-dependent stability profile of the carbidopa molecule. Chemically, carbidopa contains a hydrazine moiety and an alpha-methyl dopa structure that is susceptible to elimination reactions in aqueous media. In neutral or alkaline environments, the lone pair electrons on the nitrogen atoms facilitate the elimination of ammonia, resulting in the formation of methyldopa. By introducing mineral or organic acids such as hydrochloric acid, sulfuric acid, or tartaric acid, the system creates a high concentration of protons that protonate the amine groups. This protonation effectively locks the molecular structure, raising the activation energy required for the elimination reaction and thereby kinetically inhibiting the formation of methyldopa during the high-temperature dissolution step. This stabilization is crucial because it prevents the generation of new impurities during the very process intended to remove them, ensuring that the impurity load decreases monotonically throughout the purification cycle rather than fluctuating due to concurrent degradation.
In addition to acid stabilization, the process employs a rigorous control of dissolved oxygen and thermal history to further refine the impurity profile. The inclusion of nitrogen protection or reducing agents like sodium sulfite or vitamin C serves to scavenge free radicals and prevent the oxidation of the catechol ring system, which could otherwise lead to colored impurities and polymeric byproducts. The decolorization step, performed at elevated temperatures (90°C to 100°C) with activated carbon, ensures that any pre-existing colored impurities or trace organics are adsorbed efficiently before crystallization begins. Following hot filtration, the controlled cooling of the filtrate to 0°C to 30°C induces supersaturation in a manner that favors the growth of pure carbidopa crystals while leaving residual methyldopa and other soluble impurities in the mother liquor. This precise thermodynamic control, combined with the kinetic stability provided by the acidic pH, results in a final product with exceptional purity levels, often exceeding 99.5%, making it suitable for direct formulation or further processing without additional purification steps.
How to Synthesize Carbidopa Efficiently
Implementing this refined synthesis route requires careful attention to pH control and temperature management to maximize the benefits of the acidic stabilization mechanism. The process begins with the preparation of a dissolution vessel equipped with heating, stirring, and inert gas blanketing capabilities to ensure a consistent reaction environment. Operators must carefully select the appropriate acid to adjust the pH, balancing solubility enhancement with the ease of downstream removal, although the patent indicates that various mineral and organic acids are effective. The following guide outlines the critical operational parameters derived from the patent examples to assist technical teams in replicating this high-efficiency purification protocol.
- Dilute carbidopa crude product with water (10-80x weight) and adjust pH to below 6.5 using mineral or organic acid.
- Heat the mixture to 50-110°C under nitrogen protection or with a reducing agent to ensure full dissolution without oxidation.
- Add a decolorizing agent, filter while hot, cool the filtrate to 0-30°C, and stir to crystallize the refined product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this acidic recrystallization technology translates directly into tangible operational efficiencies and risk mitigation. The primary economic driver is the drastic reduction in processing cycles; whereas traditional methods often require repeated recrystallizations to achieve compliance, this novel approach consistently meets pharmacopeia standards in a single pass. This consolidation of steps eliminates the need for intermediate drying, re-dissolution, and secondary filtration, which are resource-intensive operations that consume significant amounts of energy, water, and labor. By streamlining the production workflow, manufacturers can significantly increase throughput capacity without expanding physical infrastructure, thereby enhancing the overall agility of the supply chain to respond to market demand fluctuations for high-purity carbidopa.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization cycles leads to substantial savings in utility consumption and solvent waste disposal. Since the process achieves high yields in a single operation, the loss of valuable API material associated with repeated handling and mother liquor discard is minimized. Furthermore, the use of common, inexpensive reagents like water and standard acids keeps raw material costs low, avoiding the need for exotic solvents or expensive chromatographic resins. This efficient use of resources ensures that the cost of goods sold (COGS) is optimized, allowing for more competitive pricing structures in the global marketplace while maintaining healthy profit margins for producers.
- Enhanced Supply Chain Reliability: The robustness of this method against variability in crude quality provides a more predictable production schedule. Traditional processes are sensitive to the initial impurity load, often requiring process adjustments or extra cycles if the crude quality fluctuates. In contrast, the high impurity clearance rate of the acidic method acts as a buffer, ensuring consistent output quality regardless of minor variations in the upstream synthesis of the crude carbidopa. This reliability reduces the risk of batch failures and delivery delays, ensuring a steady flow of material to downstream formulation partners and strengthening the resilience of the entire pharmaceutical supply network against disruptions.
- Scalability and Environmental Compliance: From an environmental perspective, the process is inherently greener due to the reduced volume of wastewater generated per kilogram of finished product. The avoidance of organic solvents in favor of aqueous systems simplifies waste treatment protocols and lowers the environmental footprint of the manufacturing site. Additionally, the simplicity of the unit operations—dissolution, filtration, and crystallization—makes the technology highly scalable from pilot plant to commercial production scales of 100 MT or more. This ease of scale-up minimizes the technical risks associated with technology transfer, allowing for rapid deployment of capacity to meet growing global demand for Parkinson's disease medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this refining technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy techniques in terms of purity and yield. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement specialists assessing supplier capabilities.
Q: Why is acidic pH critical during carbidopa recrystallization?
A: Carbidopa is chemically unstable in neutral or alkaline aqueous solutions, where it readily decomposes into methyldopa and ammonia. Maintaining a pH below 6.5, ideally between 1 and 3, stabilizes the molecule, preventing this degradation pathway and ensuring high recovery yields.
Q: How does this method improve methyldopa impurity removal?
A: Traditional water recrystallization removes only about 40% of methyldopa impurities. The patented acidic method leverages solubility differences and stability controls to achieve over 90% removal efficiency in a single pass, meeting strict pharmacopeia standards (<0.5%).
Q: What are the scalability advantages of this refining process?
A: The process utilizes common reagents like water and standard acids, operates at moderate temperatures (50-110°C), and eliminates the need for repeated recrystallization cycles. This simplifies equipment requirements and significantly reduces production time and solvent consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbidopa Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced refining technologies like the acidic recrystallization method is critical for maintaining competitiveness in the global API market. As a leading CDMO and supplier, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102432496A are fully realized in industrial practice. Our facilities are equipped with state-of-the-art reactors capable of precise pH and temperature control, along with rigorous QC labs that enforce stringent purity specifications to guarantee every batch meets international regulatory standards. We are committed to leveraging our technical expertise to deliver high-purity carbidopa that supports the development of safe and effective therapeutic formulations.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to optimize their supply chains for anti-Parkinson agents. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, demonstrating our transparency and commitment to fostering long-term strategic relationships based on technical excellence and supply security.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
