Advanced Synthesis of Monomolecular Phosphorus-Nitrogen Intumescent Flame Retardants for Commercial Scale-Up
The global demand for high-performance, environmentally compliant polymer additives has necessitated a shift away from traditional halogenated solutions toward advanced phosphorus-nitrogen systems. Patent CN101168547A introduces a groundbreaking synthesis method for a monomolecular phosphorus-nitrogen intumescent flame retardant that addresses critical limitations in current market offerings. This technology leverages a sophisticated two-step reaction sequence starting with the phosphorylation of pentaerythritol to form a caged phosphate intermediate, followed by chain extension with diisocyanates. For R&D Directors and Procurement Managers seeking a reliable polymer additive supplier, this patent represents a significant opportunity to enhance product safety profiles while maintaining rigorous performance standards. The resulting material offers a unified molecular architecture that inherently improves compatibility with polymer matrices, reducing the need for excessive loading rates that often compromise mechanical properties. By integrating the acid source, carbon source, and gas source into a single chemical entity, this innovation ensures a more consistent and predictable char formation process during thermal decomposition events.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional intumescent flame retardant systems typically rely on physical blends of ammonium polyphosphate (APP) and pentaerythritol (PER), which present substantial challenges for large-scale manufacturing and end-user application. These mixtures often suffer from severe hygroscopicity, leading to moisture absorption that degrades the mechanical integrity of the final polymer product and complicates storage logistics for supply chain teams. Furthermore, the physical incompatibility between the inorganic acid source and the organic carbon source frequently results in phase separation within the polymer matrix, causing blooming issues and inconsistent flame retardant performance across production batches. The high loading levels required to achieve V-0 ratings in these conventional systems often drastically reduce the impact strength and tensile properties of the host resin, limiting their utility in high-performance engineering plastics. Additionally, the thermal stability of these blends is often insufficient for high-temperature processing conditions, leading to premature decomposition and the release of ammonia gas during extrusion, which creates voids and surface defects in the finished goods.
The Novel Approach
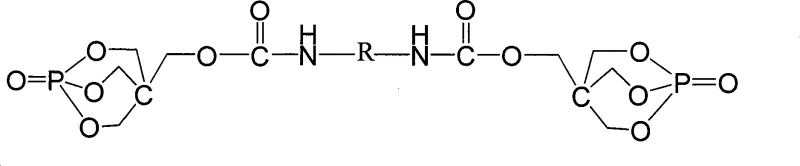
The novel approach detailed in the patent data overcomes these structural deficiencies by synthesizing a single-molecule system where the reactive components are covalently bonded rather than physically mixed. This monomolecular design eliminates the hygroscopic nature associated with ammonium salts, significantly enhancing the hydrolytic stability of the additive during storage and processing. By reacting the caged phosphate intermediate PEPA with diisocyanates such as TDI or MDI, the method creates a robust urethane-linked framework that exhibits superior thermal resistance and compatibility with a wide range of thermoplastic resins. This chemical integration allows for cost reduction in flame retardant manufacturing by reducing the total additive loading required to achieve equivalent fire safety ratings, thereby preserving the physical mechanics of the base polymer. The process utilizes readily available industrial raw materials and operates under relatively mild conditions, facilitating easier commercial scale-up of complex polymer additives without requiring exotic catalysts or extreme pressure equipment. This strategic shift from blending to molecular design ensures a more homogeneous dispersion within the polymer, leading to consistent UL-94 V-0 performance and higher oxygen index values.
Mechanistic Insights into PEPA-Based Urethane Formation
The core of this synthesis lies in the initial formation of 1-oxo-4-hydroxymethyl-1-phospho-hetero-2,6,7-trioxabicyclo[2,2,2]octane (PEPA), achieved through the reaction of pentaerythritol with phosphorus oxychloride at temperatures between 80-120°C. This cyclization reaction creates a stable caged phosphate structure that serves as both the acid source and the carbon source for the intumescent mechanism. The subsequent step involves the nucleophilic attack of the hydroxyl group on the PEPA intermediate onto the isocyanate group of TDI or MDI, catalyzed by tertiary amines or organotin compounds. This urethane formation reaction proceeds efficiently at temperatures ranging from 20-100°C, linking the phosphorus-rich cages together via aromatic or aliphatic bridges to form a high molecular weight network. The resulting macromolecule is designed to decompose upon heating to generate a coherent, expanded char layer that insulates the underlying polymer from heat flux and oxygen diffusion. This mechanistic pathway ensures that the volatile combustible gases released by the degrading polymer are trapped within the char matrix, effectively suppressing flame propagation and smoke generation.
Impurity control is meticulously managed through the precise stoichiometric balance of reactants, specifically maintaining the molar ratio of PEPA to isocyanate between 2:1 and 3:1 to prevent incomplete reactions or excessive cross-linking. Deviations from this optimal ratio can lead to unreacted isocyanate residues which may cause discoloration or reduced thermal stability in the final product. The purification process involves precipitation in acidified ice water followed by multiple washing cycles with water and acetone to remove residual solvents and catalyst traces. This rigorous workup procedure ensures that the final white or light yellow solid meets stringent purity specifications required for high-end electronic or automotive applications. The high yields reported, ranging from 90% to 96%, indicate a highly selective reaction pathway with minimal side product formation, which is critical for maintaining consistent batch-to-batch quality in industrial settings. Understanding these mechanistic nuances allows technical teams to optimize reaction parameters for maximizing throughput while minimizing waste generation and energy consumption.
How to Synthesize Monomolecular Phosphorus-Nitrogen Intumescent Flame Retardant Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for replicating this high-value additive in a pilot or production environment, emphasizing the importance of temperature control and reagent addition rates. The process begins with the careful addition of phosphorus oxychloride to a solution of pentaerythritol in dioxane, requiring efficient stirring to manage the exothermic nature of the phosphorylation step. Following the isolation of the PEPA intermediate, the second stage involves dissolving the solid in a polar aprotic solvent like DMF before the dropwise addition of the diisocyanate component. Detailed standardized synthesis steps are provided below to ensure reproducibility and safety during operation.
- React pentaerythritol with phosphorus oxychloride at 80-120°C to synthesize the PEPA intermediate.
- Dissolve PEPA in a polar solvent and react with TDI or MDI isocyanates at 20-100°C using an amine catalyst.
- Precipitate the final product in acidified ice water, wash thoroughly, and vacuum dry to obtain high-purity solids.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits driven by the simplicity and efficiency of the chemical process. The reliance on bulk commodity chemicals such as pentaerythritol and common diisocyanates ensures a stable and resilient supply chain, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. The high reaction yields observed in the patent data translate directly into improved atom economy, meaning less raw material is wasted as byproducts, which significantly lowers the effective cost per kilogram of the active ingredient. Furthermore, the elimination of halogenated compounds simplifies regulatory compliance and waste disposal procedures, removing the need for expensive hazardous waste treatment protocols associated with brominated or chlorinated flame retardants. The mild reaction conditions also reduce energy consumption during production, contributing to a lower carbon footprint and aligning with corporate sustainability goals without compromising on performance metrics.
- Cost Reduction in Manufacturing: The synthesis pathway utilizes inexpensive, widely available starting materials that are produced on a massive industrial scale, ensuring competitive pricing and long-term supply security. By integrating the acid and carbon sources into a single molecule, the formulation efficiency is increased, allowing manufacturers to achieve target fire ratings with lower additive loadings which reduces the overall compound cost. The high yield of 90-96% minimizes material loss during production, directly improving the gross margin potential for manufacturers adopting this technology. Additionally, the simplified purification process involving water and acetone washing avoids the need for complex chromatographic separations or expensive recrystallization solvents, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including phosphorus oxychloride and TDI/MDI, are established commodities with multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions allows for flexible manufacturing scheduling, as the process is not overly sensitive to minor fluctuations in ambient temperature or humidity compared to moisture-sensitive alternatives. This stability ensures consistent lead times for high-purity flame retardants, enabling downstream polymer processors to maintain lean inventory levels without fear of production stoppages. The non-hygroscopic nature of the final product also simplifies logistics and warehousing, as special climate-controlled storage is not required to maintain product integrity during transit.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton reactors due to the absence of extreme pressure or cryogenic requirements, facilitating rapid commercial scale-up of complex polymer additives. The halogen-free composition ensures full compliance with increasingly stringent global environmental regulations such as RoHS and REACH, future-proofing the supply chain against regulatory bans on toxic substances. Waste streams generated during the washing phases are primarily aqueous and organic solvents that can be readily treated or recycled, minimizing the environmental impact of the manufacturing facility. This alignment with green chemistry principles enhances the brand value of the final polymer products, appealing to eco-conscious consumers and OEMs in the automotive and electronics sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this monomolecular flame retardant technology in industrial applications. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation to provide clarity on performance and handling. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this additive into existing polymer formulations.
Q: What are the primary advantages of this monomolecular IFR over traditional APP/PER blends?
A: Unlike physical blends of ammonium polyphosphate and pentaerythritol which suffer from hygroscopicity and poor compatibility, this monomolecular structure integrates the acid, carbon, and gas sources into a single molecule, ensuring superior thermal stability and matrix compatibility.
Q: Is this flame retardant suitable for halogen-free polymer applications?
A: Yes, the synthesis method explicitly avoids halogenated reagents, resulting in a non-toxic, environmentally friendly product that releases no corrosive gases during combustion, making it ideal for strict environmental compliance.
Q: What yields can be expected from this synthesis route?
A: According to the patent data, the process demonstrates high efficiency with reported yields ranging from 90% to 96%, indicating a robust and economically viable pathway for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monomolecular Phosphorus-Nitrogen Intumescent Flame Retardant Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative chemistries like this phosphorus-nitrogen system are translated into reliable supply realities. Our commitment to stringent purity specifications and rigorous QC labs guarantees that every batch of flame retardant meets the exacting standards required for high-performance polymer applications. We understand the critical balance between cost efficiency and technical performance, leveraging our CDMO expertise to optimize synthesis routes for maximum yield and minimal environmental impact. Our team is dedicated to supporting your R&D efforts with custom synthesis capabilities that can adapt this patent technology to your specific polymer matrix requirements.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current formulation needs. By collaborating with us, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this advanced monomolecular system. Let us help you secure a sustainable and high-performance supply chain for your next generation of flame-retardant materials.
