Advanced LED Excitable Sulfonium Salts for High-Performance Curing Applications
Advanced LED Excitable Sulfonium Salts for High-Performance Curing Applications
The rapid transition from traditional mercury lamp curing systems to energy-efficient LED technology has created a critical demand for specialized photoinitiators capable of operating at longer wavelengths. Patent CN112961085B introduces a groundbreaking class of LED excitable intramolecular sensitization type sulfonium salt compounds that address this specific technological gap. These novel non-conjugated sulfonium salt derivatives, represented by Formula (I), are engineered to overcome the spectral mismatch between conventional initiators and modern LED light sources. By linking a sulfonium salt to a conjugated system via a flexible alkyl chain, the invention achieves a significant red-shift in absorption peaks, ensuring high photoinitiation efficiency under 365nm LED irradiation. This development is particularly vital for industries seeking sustainable, high-speed curing solutions for coatings, inks, and adhesives without the environmental drawbacks of ozone-generating mercury lamps.
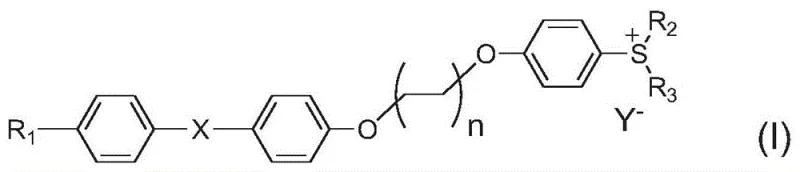
As a reliable photoinitiator supplier, understanding the structural nuances of Formula (I) is essential for optimizing formulation performance. The molecule features a versatile architecture where R1 can be hydrogen or various substituted alkyl groups, while R2 and R3 provide further tunability through benzyl or phenyl substituents. The flexibility of the linker chain, defined by integer n (preferably 2, 4, or 6), plays a pivotal role in facilitating the intramolecular energy transfer that defines the compound's efficacy. This structural design not only enhances photosensitivity but also maintains high thermal stability, making these compounds ideal candidates for demanding industrial applications where consistent curing depth and speed are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional photocuring technologies have long relied on mercury lamps, which suffer from high energy consumption, ozone generation, and significant environmental hazards. While LED light sources offer a sustainable alternative, their longer emission wavelengths often fail to activate conventional photoinitiators effectively, leading to poor curing rates and incomplete polymerization. Previous attempts to solve this by directly introducing photocleavable groups into stilbene or biphenyl structures often resulted in overly complex and costly organic synthesis processes. These conventional routes frequently require harsh reaction conditions, expensive catalysts, and multi-step purifications that drive up manufacturing costs and limit commercial viability. Furthermore, the rigid structures of many traditional initiators restrict their solubility and compatibility with diverse resin systems, creating formulation challenges for end-users seeking versatile curing agents.
The Novel Approach
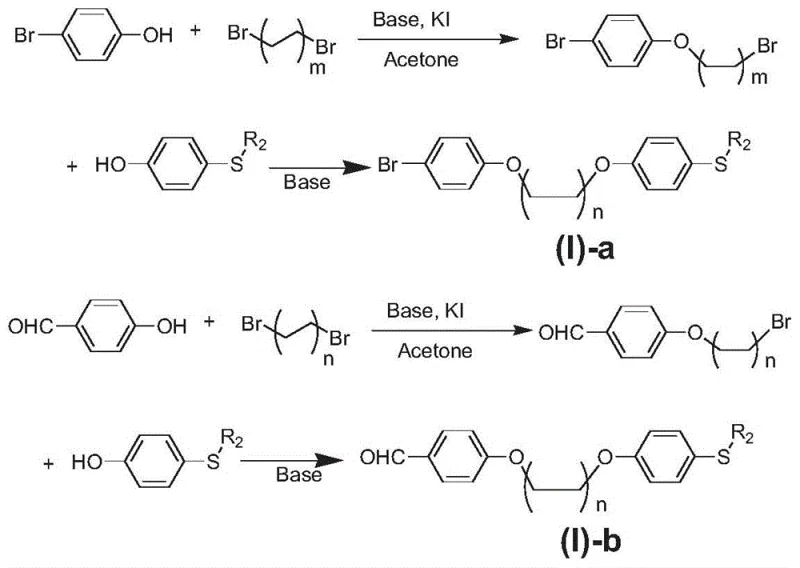
The innovative strategy outlined in the patent circumvents these issues by employing a modular synthetic route that separates the chromophore formation from the salt generation. Instead of forcing a direct conjugation that complicates synthesis, the method utilizes a flexible ether linkage to connect the light-absorbing conjugated system with the cationic sulfonium center. This approach allows for the independent optimization of the light-absorbing moiety through standard cross-coupling reactions while maintaining the cationic character necessary for acid generation. The result is a simplified preparation technology that yields high-purity products with excellent absorption characteristics in the near-ultraviolet-visible region. This decoupling of functions enables cost reduction in electronic chemical manufacturing by utilizing readily available starting materials like bromophenol and dibromoalkanes, streamlining the supply chain for high-volume production.
Mechanistic Insights into Intramolecular Sensitization and Coupling
The core mechanism driving the superior performance of these compounds is intramolecular electron or energy transfer facilitated by the flexible alkyl chain. Unlike rigid conjugated systems where electronic communication is direct but synthetically difficult, this design allows the excited state of the chromophore (such as a stilbene or biphenyl derivative) to transfer energy to the sulfonium salt moiety efficiently. This process triggers the homolytic or heterolytic cleavage of the carbon-sulfur bond, generating the active cationic species required to initiate the polymerization of epoxy or vinyl monomers. The patent highlights that this non-conjugated linkage surprisingly does not hinder efficiency; rather, it red-shifts the absorption maximum to align perfectly with commercial LED emission spectra, solving the fundamental wavelength mismatch problem that plagues the industry.
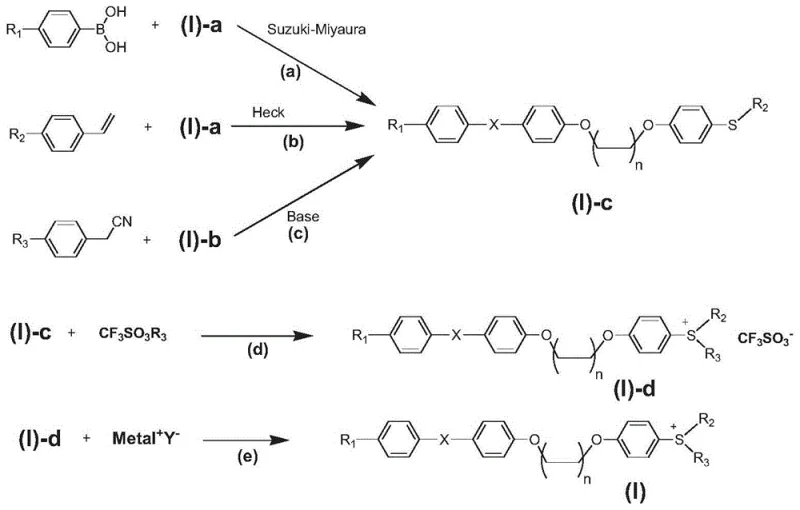
From a synthetic chemistry perspective, the construction of the molecular framework relies heavily on robust palladium-catalyzed cross-coupling reactions. The intermediates (I)-a and (I)-b serve as versatile platforms for introducing diverse functional groups via Suzuki-Miyaura or Heck reactions. For instance, reacting intermediate (I)-a with substituted phenylboronic acids allows for the precise installation of electron-donating or withdrawing groups on the aromatic ring, fine-tuning the absorption profile. Alternatively, the use of substituted styrenes in Heck reactions introduces vinyl linkages that extend the conjugation length. The patent notes that these reactions proceed under anhydrous and oxygen-free conditions to ensure high yields, typically exceeding 85-90%. Impurity control is managed through careful selection of bases and catalysts, minimizing the formation of cis-isomers in stilbene derivatives which could otherwise affect the crystallinity and solubility of the final photoinitiator.
How to Synthesize LED Excitable Sulfonium Salts Efficiently
The synthesis of these high-performance photoinitiators follows a logical, stepwise progression that balances chemical complexity with operational simplicity. The process begins with the formation of the ether backbone, followed by the construction of the conjugated system, and concludes with the quaternization of the sulfur atom. This sequence ensures that the sensitive sulfonium group is introduced only at the final stage, preventing premature decomposition during the harsher coupling steps. The detailed standardized synthesis steps below outline the critical parameters for achieving optimal yield and purity, serving as a guide for process chemists aiming to replicate or scale this technology.
- Perform etherification by reacting p-hydroxy-phenyl-methyl-sulfide with dibromoalkane using potassium carbonate and potassium iodide in acetone to form the bromo-alkoxy intermediate.
- React the bromo-alkoxy intermediate with p-bromophenol or p-hydroxybenzaldehyde under basic conditions to extend the ether chain and form intermediates (I)-a or (I)-b.
- Conduct Suzuki-Miyaura or Heck coupling reactions with substituted phenylboronic acids or styrenes using palladium catalysts to introduce the conjugated chromophore system.
- Complete the synthesis by quaternizing the sulfur atom with methyl trifluoromethanesulfonate or performing anion exchange to obtain the final sulfonium salt product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new synthetic route offers tangible benefits beyond mere technical performance. The reliance on commodity chemicals such as acetone, potassium carbonate, and common halogenated aromatics significantly de-risks the supply chain compared to processes requiring exotic reagents. The high yields reported in the patent examples, often ranging from 90% to 95% for intermediate steps, translate directly into reduced raw material consumption and lower waste generation. This efficiency is crucial for maintaining healthy margins in the competitive electronic chemicals market, where cost pressures are relentless. Furthermore, the ability to perform anion exchange as a post-synthesis modification allows manufacturers to produce a family of products from a single intermediate, maximizing asset utilization and inventory flexibility.
- Cost Reduction in Manufacturing: The elimination of complex, multi-step sequences required for direct conjugation significantly lowers the overall cost of goods sold. By utilizing high-yielding etherification and coupling reactions that operate under relatively mild conditions, the process reduces energy consumption and reactor time. The avoidance of expensive transition metal removal steps, often required in other catalytic processes, further simplifies the downstream processing. Additionally, the use of simple recrystallization or filtration for purification instead of preparative chromatography makes the process inherently more economical for tonnage-scale production.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including p-hydroxy-phenyl-methyl-sulfide and various dibromoalkanes, are widely available from global chemical suppliers. This abundance ensures a stable supply base that is less susceptible to the geopolitical or logistical disruptions that often plague niche fine chemical intermediates. The robustness of the reaction conditions, which tolerate standard industrial solvents and bases, means that production can be easily transferred between different manufacturing sites without significant re-validation, ensuring continuity of supply for key customers.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing batch processes that are easily adapted to large reactors. The waste streams generated are primarily inorganic salts and common organic solvents, which can be managed through standard wastewater treatment and solvent recovery systems. This aligns with increasingly stringent environmental regulations regarding volatile organic compounds (VOCs) and heavy metal discharge. The high atom economy of the coupling reactions minimizes waste generation at the source, supporting corporate sustainability goals and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and sourcing of these advanced sulfonium salt photoinitiators. These answers are derived directly from the technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for potential partners.
Q: What is the primary advantage of these sulfonium salts over traditional photoinitiators?
A: Unlike traditional photoinitiators designed for mercury lamps, these non-conjugated sulfonium salts feature a flexible chain linking the conjugated system to the sulfonium salt. This structure allows for intramolecular electron or energy transfer, red-shifting the absorption peak to overlap effectively with commercial LED light sources (365nm and above), thereby significantly improving excitation efficiency.
Q: Is the synthesis process suitable for large-scale industrial production?
A: Yes, the preparation method is described as simple and convenient with high yields. The reactions utilize common reagents like potassium carbonate and standard solvents like acetone and DMF. The workup procedures involve straightforward filtration and recrystallization, avoiding complex purification steps that would hinder scalability.
Q: Can the anion of the sulfonium salt be customized for specific applications?
A: Absolutely. While the direct synthesis yields the trifluoromethanesulfonate anion, the patent describes a simple salt exchange method. By reacting the initial sulfonium salt with saturated solutions of salts like potassium hexafluorophosphate or sodium hexafluoroantimonate, various anions such as PF6-, SbF6-, and BF4- can be introduced to tune solubility and performance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonium Salt Photoinitiator Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating cutting-edge patent technologies into commercial reality. With extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, we possess the technical expertise to optimize the synthesis of these LED excitable sulfonium salts for maximum efficiency. Our rigorous QC labs and stringent purity specifications ensure that every batch meets the exacting standards required for high-performance electronic materials and coating applications. We understand that consistency is key in photocuring formulations, and our dedicated process development team works closely with clients to refine crystallization and drying protocols for optimal particle size distribution.
We invite you to collaborate with us to leverage this transformative technology for your next-generation curing systems. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your growth in the rapidly expanding LED curing market.
