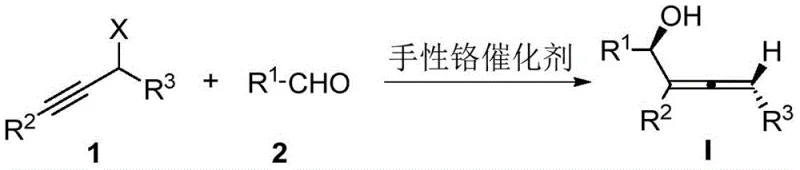
Scalable Synthesis of Dual-Chiral 2,3-Allenols via Chromium Catalysis for Advanced Pharmaceutical Intermediates
The landscape of asymmetric synthesis is constantly evolving, driven by the demand for complex chiral building blocks in the pharmaceutical industry. A significant breakthrough in this domain is documented in Chinese Patent CN113735894A, which details a robust methodology for constructing 2,3-allenol compounds possessing both axial and central chirality. This dual-chirality architecture is notoriously difficult to control simultaneously, yet it is crucial for the bioactivity of many potential drug candidates. The patent discloses a chromium-catalyzed asymmetric addition reaction between propargyl halides and aldehydes, utilizing a novel chiral ligand system to achieve exceptional stereocontrol. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this technology represents a paradigm shift away from restrictive enzymatic methods toward a more universal and scalable chemical catalysis platform. The ability to generate such complex molecular architectures from simple, commodity-grade starting materials under mild conditions underscores the practical value of this invention for modern organic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 2,3-allenols has been fraught with significant challenges that hindered their widespread adoption in process chemistry. Traditional approaches often relied on enzyme-catalyzed kinetic resolution of racemic mixtures. While enzymes offer high specificity, they suffer from narrow substrate tolerance, meaning a specific enzyme might only work for a single structural variant, rendering the method impractical for diverse library synthesis. Furthermore, enzymatic processes are inherently limited to producing compounds with single-center chirality, failing to address the complexity of molecules requiring simultaneous control of axial and central stereogenic elements. Another conventional route involves the use of chiral pool starting materials. However, these chiral raw materials are often expensive, require multi-step preparation and purification, and impose severe structural limitations on the final product. As illustrated in prior art reviews, earlier catalytic methods reported by groups such as List, Feng, and Yin often necessitated specialized activated substrates like alkynyl-substituted ketene acetals or propargyl esters, which restricts the chemical space accessible to medicinal chemists.
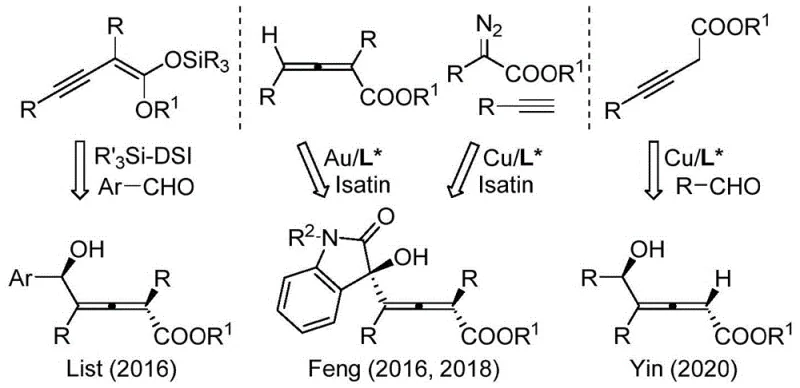
The Novel Approach
The methodology presented in CN113735894A overcomes these historical bottlenecks by employing a chiral chromium catalyst system that operates on simple, unactivated propargyl halides and aldehydes. This approach eliminates the need for pre-functionalized or expensive chiral starting materials. The core innovation lies in the use of specific chiral ligands (designated La through Lk) coordinated with chromium dichloride. This catalyst system facilitates the asymmetric addition reaction with remarkable efficiency, tolerating a wide array of functional groups on both the aldehyde and the alkyne components. Unlike previous methods that were restricted to ester-containing allene fragments, this new protocol allows for the synthesis of simple alkyl-substituted products in the dienyl fragment. The reaction proceeds under mild conditions, typically at room temperature, which is a stark contrast to the harsh conditions often required by older methodologies. This universality and operational simplicity make it an ideal candidate for cost reduction in API manufacturing, as it streamlines the supply chain by relying on bulk chemicals rather than bespoke chiral reagents.
Mechanistic Insights into Chromium-Catalyzed Asymmetric Addition
The success of this transformation hinges on the precise design of the chiral ligand environment around the chromium center. The patent details a series of bis-oxazoline-type ligands (La-Lk) that create a rigid chiral pocket, effectively differentiating the enantiotopic faces of the incoming aldehyde and controlling the geometry of the allenyl-chromium intermediate. The reaction mechanism likely involves the in situ generation of an allenyl-chromium species from the propargyl halide and manganese reductant. This nucleophilic species then attacks the aldehyde carbonyl in a stereocontrolled manner dictated by the chiral ligand. The presence of zirconocene dichloride acts as a dissociating agent, potentially facilitating the turnover of the catalyst or stabilizing reactive intermediates. The result is the formation of a carbon-carbon bond with simultaneous establishment of a chiral axis in the allene moiety and a chiral center at the propargylic position. This dual stereocontrol is evidenced by the high diastereomeric ratios (dr > 20:1) and enantiomeric excesses (up to 98% ee) observed across various substrates. Such high fidelity in stereochemical outcome is critical for pharmaceutical applications where impurity profiles are strictly regulated.
Furthermore, the mechanistic robustness of this system allows for extensive substrate scope exploration. The electronic nature of the aldehyde, whether electron-rich or electron-deficient, has minimal impact on the stereoselectivity, suggesting a highly organized transition state that overrides subtle electronic effects. Heterocyclic aldehydes, such as furfural and thiophene carboxaldehydes, are also well-tolerated, expanding the utility of this method for synthesizing heteroaromatic intermediates common in drug discovery. The tolerance for bulky silyl protecting groups (like TIPS) on the alkyne terminus further demonstrates the steric flexibility of the catalyst. This mechanistic resilience ensures that the process remains viable even when scaling up, where mixing and heat transfer issues might otherwise perturb sensitive catalytic cycles. For technical teams, understanding this mechanism provides confidence in the reproducibility of the process across different batches and scales.
How to Synthesize Chiral 2,3-Allenols Efficiently
The practical execution of this synthesis is designed for ease of operation, aligning with the needs of process development teams looking for robust protocols. The procedure typically begins with the preparation of the chiral chromium catalyst in a glove box to ensure an oxygen-free environment, reacting chromium dichloride with the optimal ligand (such as Ld) in dimethoxyethane (DME). Once the catalyst is formed, the reaction components are added sequentially: the propargyl halide, the aldehyde substrate, manganese powder as the stoichiometric reductant, and zirconocene dichloride. The mixture is stirred at room temperature for approximately 12 hours, allowing the transformation to reach completion without the need for external heating or cooling. Following the reaction, a simple aqueous quench and filtration through a short silica plug removes the metal salts and excess manganese. The final purification is achieved via standard column chromatography. For detailed standardized operating procedures and specific molar ratios, please refer to the guide below.
- Prepare the chiral chromium catalyst by reacting chromium dichloride with chiral ligand Ld in DME solvent at room temperature for 2 hours under inert atmosphere.
- Add propargyl halide, aldehyde substrate, manganese powder as reducing agent, and zirconocene dichloride as dissociating agent to the catalyst mixture.
- Stir the reaction mixture at room temperature for 12 hours, quench with water, and purify the crude product via silica gel column chromatography to obtain the target chiral 2,3-allenol.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this chromium-catalyzed route offers substantial strategic benefits for supply chain managers and procurement officers. The primary advantage lies in the drastic simplification of the raw material portfolio. By utilizing commodity chemicals like benzaldehyde derivatives and simple propargyl bromides, manufacturers can mitigate supply risks associated with exotic or single-source chiral reagents. This shift towards abundant feedstocks directly contributes to cost reduction in pharmaceutical intermediate manufacturing, as the volatility of prices for specialized chiral auxiliaries is eliminated. Moreover, the reaction conditions are exceptionally mild, operating at ambient temperature and pressure. This reduces the energy footprint of the manufacturing process and lowers the capital expenditure required for specialized reactor equipment capable of handling extreme cryogenic or high-temperature conditions.
- Cost Reduction in Manufacturing: The economic viability of this process is enhanced by the use of inexpensive reagents such as manganese powder and chromium dichloride, which are significantly cheaper than noble metal catalysts like gold or palladium often used in similar transformations. The elimination of expensive chiral starting materials and the high atom economy of the addition reaction further drive down the cost of goods sold (COGS). Additionally, the high stereoselectivity minimizes the formation of unwanted diastereomers and enantiomers, reducing the burden and cost associated with downstream purification and waste disposal. This efficiency translates into a more competitive pricing structure for the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Relying on widely available chemical building blocks ensures a stable and resilient supply chain. Unlike enzymatic processes that may depend on specific fermentation outputs or biocatalysts with limited shelf-life, this chemical catalysis approach uses stable, shelf-stable reagents that can be sourced from multiple global suppliers. This diversification of the supply base reduces the risk of production stoppages due to raw material shortages. The robustness of the reaction also implies consistent batch-to-batch quality, which is essential for maintaining long-term contracts with pharmaceutical clients who require stringent quality assurance.
- Scalability and Environmental Compliance: The patent explicitly notes the ease of reaction amplification, indicating that the process is amenable to commercial scale-up from kilogram to tonnage levels. The use of standard organic solvents like DME and the absence of hazardous high-pressure gases simplify the safety profile of the plant operations. Furthermore, the high selectivity of the reaction reduces the generation of chemical waste, aligning with green chemistry principles and easing the regulatory burden associated with environmental compliance. This scalability ensures that the technology can meet the growing demand for complex chiral intermediates without compromising on quality or delivery timelines.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating this technology, we have compiled answers to common questions regarding the implementation and scope of this chromium-catalyzed synthesis. These insights are derived directly from the experimental data and technical disclosures within the patent documentation. Understanding these nuances is vital for assessing the feasibility of integrating this route into existing production workflows.
Q: What are the key advantages of this chromium-catalyzed method over previous enzymatic or chiral pool approaches?
A: Unlike enzymatic kinetic resolution which is limited to specific substrates, or chiral pool synthesis which requires expensive starting materials, this chromium-catalyzed method utilizes cheap and readily available propargyl halides and aldehydes. It offers broad substrate scope, mild reaction conditions (room temperature), and achieves high stereoselectivity (up to 98% ee and >20:1 dr) for products containing both axial and central chirality.
Q: Can this synthetic route be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the patent explicitly highlights the ease of reaction amplification. The use of inexpensive reagents like manganese powder and standard solvents like DME, combined with room temperature operation, significantly simplifies process engineering compared to cryogenic or high-pressure alternatives, making it highly suitable for commercial scale-up.
Q: What types of downstream transformations are possible with these chiral 2,3-allenol products?
A: The resulting 2,3-allenols are versatile building blocks. As demonstrated in the patent data, they can undergo electrophilic cyclization to form complex tetrahydrofuran derivatives with multiple chiral centers. Furthermore, they serve as precursors for the total synthesis of bioactive natural products such as (+)-varitriol, demonstrating their high utility in medicinal chemistry.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Allenol Supplier
The technological potential of chiral 2,3-allenols as versatile building blocks for natural products and APIs is immense, yet realizing this potential requires a partner with deep expertise in process chemistry. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of chiral intermediate meets the highest global standards. We understand the critical nature of stereochemical purity in drug development and are committed to delivering materials that facilitate your regulatory filings and clinical trials.
We invite you to collaborate with us to optimize your supply chain for these high-value intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. By leveraging our expertise in chromium catalysis and asymmetric synthesis, we can help you navigate the complexities of scale-up and secure a reliable source of high-purity chiral building blocks. Contact us today to request specific COA data and route feasibility assessments for your next generation of pharmaceutical candidates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
