Scalable Chromium-Catalyzed Synthesis of Chiral 2,3-Allenols for Advanced Pharmaceutical Intermediates
Introduction to Dual-Chirality Allenol Synthesis
The development of efficient synthetic routes for constructing complex chiral architectures remains a cornerstone of modern pharmaceutical chemistry. Patent CN113735894B introduces a groundbreaking methodology for the preparation of 2,3-allenol compounds that simultaneously possess both axial and central chirality. These structural motifs are highly valued in organic synthesis due to their unique reactivity profiles, serving as versatile precursors for heterocycles and natural products. The core innovation lies in a chromium-catalyzed asymmetric addition reaction between propargyl halides and aldehydes, which bypasses the limitations of traditional methods.
This technology represents a significant leap forward for manufacturers seeking reliable pharmaceutical intermediate supplier capabilities. By enabling the direct assembly of dual-chirality centers from simple, commercially available starting materials, the process drastically simplifies the supply chain for high-value active ingredients. The ability to access these complex scaffolds with high stereochemical fidelity opens new avenues for drug discovery and process optimization.
The structural versatility of the resulting 2,3-allenols allows for diverse downstream transformations, including transition metal-catalyzed cyclizations and rearrangements. This flexibility makes them indispensable building blocks for synthesizing bioactive molecules such as furanomycins and amphidinolides. For R&D teams, mastering this chemistry means gaining access to a robust platform for generating novel chemical space with defined stereochemistry, essential for meeting the rigorous demands of modern regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral 2,3-allenols has been fraught with challenges that hinder industrial adoption. Traditional approaches often rely on enzymatic kinetic resolution of racemic mixtures, a technique severely constrained by the narrow substrate specificity of enzymes. This limitation restricts the method to producing compounds with only single central chirality, failing to address the need for dual-chirality systems. Furthermore, the requirement for specific substrate structures limits the universality of enzymatic processes, making them unsuitable for broad-scale application.
Alternative strategies involving chiral pool synthesis necessitate the use of expensive, pre-functionalized chiral starting materials. These materials often require complex preparation and purification steps, inflating costs and extending lead times. Additionally, catalytic asymmetric synthesis methods reported prior to this patent, such as those using gold or copper catalysts, frequently demand specialized activated substrates like propargyl esters. These constraints result in product structures that are limited to specific functional groups, such as esters in the allene fragment, thereby excluding simple alkyl-substituted variants crucial for many pharmaceutical targets.
The Novel Approach
The methodology disclosed in CN113735894B overcomes these barriers by utilizing a chromium-based catalytic system that operates under mild conditions with simple substrates. By employing propargyl halides and aldehydes directly, the process eliminates the need for pre-activated esters or complex chiral pools. This shift significantly reduces cost reduction in API manufacturing by streamlining the raw material sourcing and minimizing waste generation associated with multi-step activation sequences.
Moreover, the reaction exhibits exceptional tolerance for a wide range of functional groups on both the aldehyde and the alkyne components. From electron-rich aromatics to heterocycles and aliphatic chains, the system maintains high efficiency. This broad substrate scope ensures that the commercial scale-up of complex pharmaceutical intermediates is feasible without extensive re-optimization for each new target. The operational simplicity, conducted at room temperature in common solvents like DME, further enhances its attractiveness for large-scale production environments.
Mechanistic Insights into Chromium-Catalyzed Asymmetric Addition
The success of this transformation hinges on the precise design of the chiral ligand environment surrounding the chromium center. The patent details a series of bis-oxazoline-type ligands (La-Lk) that dictate the stereochemical outcome of the reaction. Among these, ligand Ld has been identified as particularly effective, facilitating the formation of the carbon-carbon bond with remarkable diastereo- and enantioselectivity. The interaction between the chromium species and the ligand creates a rigid chiral pocket that effectively differentiates the prochiral faces of the incoming aldehyde.
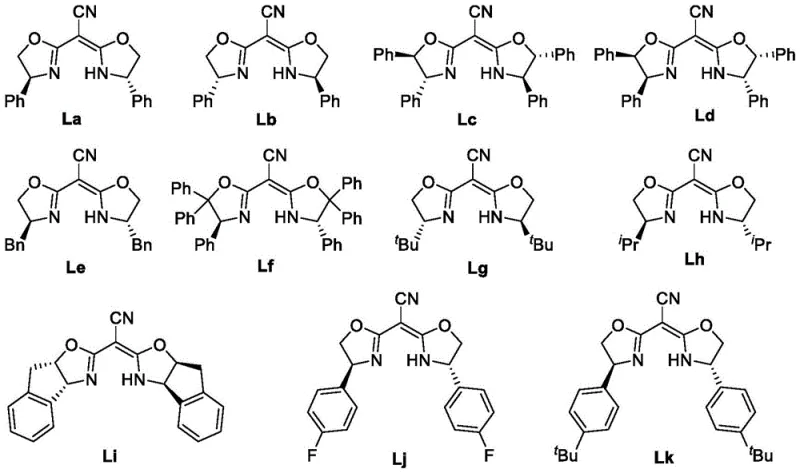
Mechanistically, the reaction proceeds through the generation of an organochromium intermediate via the reduction of the propargyl halide by manganese in the presence of Cp2ZrCl2. This active species then undergoes nucleophilic addition to the aldehyde carbonyl group. The chiral ligand controls the trajectory of this addition, ensuring that the newly formed stereocenters align with the desired configuration. The presence of both axial chirality in the allene moiety and central chirality at the alcohol carbon is achieved simultaneously, a feat difficult to accomplish with other metal systems.
Impurity control is inherently built into this mechanism due to the high selectivity of the catalyst. The stringent steric constraints imposed by the ligand minimize side reactions such as homocoupling of the alkyne or non-selective background reactions. This results in crude products with high purity profiles, reducing the burden on downstream purification processes. For quality control laboratories, this translates to more consistent batch-to-batch reproducibility and easier compliance with stringent purity specifications required for GMP manufacturing.
How to Synthesize Chiral 2,3-Allenols Efficiently
Implementing this synthesis requires careful attention to the preparation of the catalytic species and the handling of reagents under inert conditions. The protocol involves the in situ generation of the chiral chromium catalyst followed by the sequential addition of substrates and additives. This standardized approach ensures that the high levels of stereocontrol observed in the patent examples can be replicated reliably in a production setting.
- Prepare the chiral chromium catalyst by reacting chromium dichloride with a specific chiral ligand (such as Ld) in an organic solvent like DME at room temperature for over two hours.
- In a glove box, combine the prepared catalyst with propargyl halide, aldehyde, manganese powder as a reducing agent, and Cp2ZrCl2 as a dissociating agent.
- Allow the reaction mixture to stir at room temperature for approximately 12 hours, then quench with water and purify the crude product via column chromatography to isolate the target chiral 2,3-allenol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chromium-catalyzed route offers tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals like propargyl halides and benzaldehydes mitigates the risk of supply disruptions often associated with exotic chiral reagents. This stability in raw material availability ensures enhanced supply chain reliability, allowing for consistent production schedules and reduced inventory holding costs for specialized precursors.
From a cost perspective, the elimination of expensive noble metal catalysts (like gold or palladium) in favor of abundant chromium significantly lowers the direct material costs. Additionally, the reaction proceeds at room temperature, removing the energy expenditures associated with cryogenic cooling or high-temperature heating. These factors contribute to substantial cost savings in manufacturing operations, improving the overall margin profile of the final active pharmaceutical ingredient. The simplified workup procedure, involving basic filtration and chromatography, further reduces processing time and solvent consumption.
Scalability and environmental compliance are also markedly improved. The mild reaction conditions and the use of less hazardous solvents align with green chemistry principles, facilitating easier permitting and waste management. The robustness of the reaction allows for seamless translation from gram-scale laboratory experiments to multi-kilogram pilot runs without loss of selectivity. This ease of scale-up reduces the time-to-market for new drug candidates, providing a competitive edge in the fast-paced pharmaceutical landscape.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for evaluating the fit of this chemistry within your existing process development pipeline. The answers are derived directly from the experimental data and scope defined in the patent documentation.
Q: What distinguishes this chromium-catalyzed method from previous enzymatic or chiral pool syntheses?
A: Unlike enzymatic kinetic resolution which is limited to single central chirality and specific substrates, or chiral pool synthesis which requires complex purification of starting materials, this chromium-catalyzed method utilizes simple propargyl halides and aldehydes. It achieves simultaneous control of both axial and central chirality with broad substrate scope and high stereoselectivity under mild conditions.
Q: Can this synthetic route be applied to the production of natural products?
A: Yes, the patent explicitly demonstrates the utility of these chiral 2,3-allenols as key intermediates. For instance, the methodology was successfully employed to synthesize the natural product (+)-varitriol through subsequent electrophilic cyclization reactions, proving its viability for complex molecule construction.
Q: What are the typical stereoselectivity outcomes for this reaction?
A: The optimized protocol using ligand Ld typically yields products with excellent diastereoselectivity (dr > 20:1) and high enantiomeric excess (up to 98% ee). This level of stereocontrol is critical for pharmaceutical applications where isomeric purity directly impacts biological activity and safety profiles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Allenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chromium-catalyzed technology for advancing drug development programs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop to plant floor is seamless. We are committed to delivering high-purity 2,3-allenol intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation.
We invite you to collaborate with us to leverage this innovative synthetic route for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can accelerate your timeline and optimize your budget.
