Advanced Industrial Synthesis of Perindopril Intermediates for Scalable API Production
Advanced Industrial Synthesis of Perindopril Intermediates for Scalable API Production
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, driven by the relentless demand for cost-effective and high-purity Active Pharmaceutical Ingredients (APIs). Patent CN1753906A introduces a transformative methodology for the synthesis of Perindopril and its pharmaceutically acceptable salts, addressing critical bottlenecks in traditional manufacturing. This innovation is particularly significant for global supply chains seeking reliable pharmaceutical intermediate suppliers who can deliver consistent quality without the prohibitive costs associated with legacy synthetic routes. By leveraging a novel enamine-based strategy, this process bypasses the reliance on scarce natural precursors, offering a robust pathway from commodity chemicals to complex heterocyclic scaffolds. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing the production of this vital ACE inhibitor.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Perindopril, as described in earlier intellectual property such as EP 0 308 341, has been constrained by the availability and cost of key starting materials. The conventional route necessitates the use of (2S,3aS,7aS)-octahydroindole-2-carboxylic acid benzyl ester, a specialized intermediate that is not commercially available off-the-shelf. Producing this precursor typically involves a cumbersome multi-step sequence starting from indole-2-carboxylic acid, which invariably includes difficult resolution steps to achieve the required stereochemistry. These additional synthetic operations not only inflate the cost of goods sold (COGS) but also introduce potential points of failure regarding yield loss and impurity generation. For a procurement manager, this dependency on a custom-synthesized, low-volume starting material creates significant supply chain vulnerability and limits the ability to negotiate favorable pricing terms with vendors.
The Novel Approach
In stark contrast, the methodology disclosed in CN1753906A revolutionizes the entry point into the Perindopril scaffold by utilizing 1-(1-cyclohexen-1-yl)-pyrrolidine, designated as Formula (II) in the patent documentation. This enamine derivative is significantly more accessible and cost-effective than the octahydroindole esters used in prior art. The strategic shift allows manufacturers to construct the bicyclic indole core in situ through a streamlined alkylation and cyclization sequence. 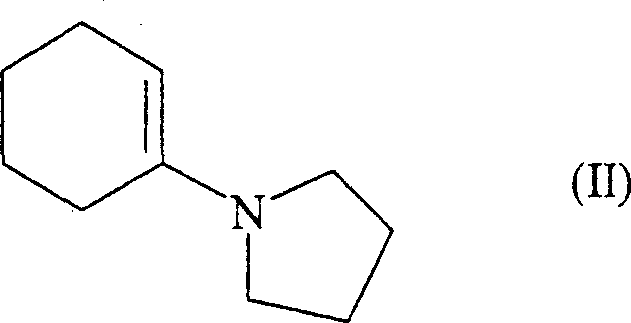 By starting with this abundant enamine, the process eliminates the need for the aforementioned resolution steps and reduces the total number of unit operations required to reach the advanced intermediate stage. This simplification translates directly into enhanced process efficiency and a more resilient supply chain, making it an ideal candidate for cost reduction in API manufacturing.
By starting with this abundant enamine, the process eliminates the need for the aforementioned resolution steps and reduces the total number of unit operations required to reach the advanced intermediate stage. This simplification translates directly into enhanced process efficiency and a more resilient supply chain, making it an ideal candidate for cost reduction in API manufacturing.
Mechanistic Insights into Enamine Alkylation and Cyclization
The core chemical innovation of this process lies in the precise construction of the chiral centers and the fused ring system. The synthesis initiates with the alkylation of the enamine nitrogen or the alpha-carbon (depending on specific tautomeric conditions controlled by the solvent and temperature) with a chiral iodide derivative, represented as Formula (III). 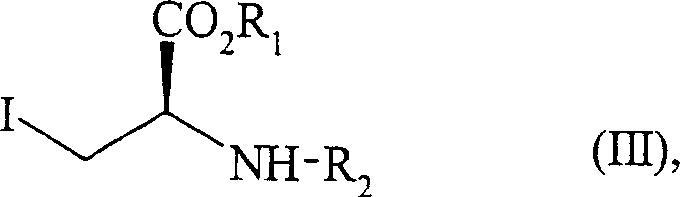 This step is critical as it installs the side chain containing the protected amine and ester functionalities that will eventually become part of the peptide-like backbone of Perindopril. The reaction is typically conducted in polar aprotic solvents like acetonitrile under reflux, ensuring complete conversion while maintaining the integrity of the sensitive functional groups. The use of a chiral iodide ensures that the stereochemistry at the alpha-position is established early with high fidelity, setting the stage for the subsequent diastereoselective transformations.
This step is critical as it installs the side chain containing the protected amine and ester functionalities that will eventually become part of the peptide-like backbone of Perindopril. The reaction is typically conducted in polar aprotic solvents like acetonitrile under reflux, ensuring complete conversion while maintaining the integrity of the sensitive functional groups. The use of a chiral iodide ensures that the stereochemistry at the alpha-position is established early with high fidelity, setting the stage for the subsequent diastereoselective transformations.
Following the alkylation and subsequent deprotection of the amine group, the molecule undergoes a pivotal acid-catalyzed cyclization. This step involves the condensation of the free amine with the ketone functionality generated from the hydrolysis of the enamine moiety. The reaction is facilitated by strong organic acids such as p-toluenesulfonic acid in azeotropic solvents like toluene, which drive the equilibrium forward by removing water. This cyclization effectively closes the five-membered nitrogen-containing ring fused to the cyclohexane ring, yielding the hexahydroindole core, shown as Formula (V). 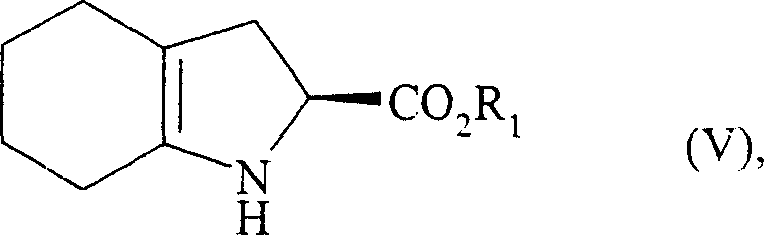 The mechanism relies on the thermodynamic stability of the fused bicyclic system and the specific spatial arrangement of the substituents, which favors the formation of the desired cis-fused stereochemistry. This intrinsic stereocontrol is vital for minimizing the formation of diastereomeric impurities, thereby simplifying downstream purification and ensuring the final API meets stringent regulatory specifications for chiral purity.
The mechanism relies on the thermodynamic stability of the fused bicyclic system and the specific spatial arrangement of the substituents, which favors the formation of the desired cis-fused stereochemistry. This intrinsic stereocontrol is vital for minimizing the formation of diastereomeric impurities, thereby simplifying downstream purification and ensuring the final API meets stringent regulatory specifications for chiral purity.
How to Synthesize Perindopril Intermediates Efficiently
The execution of this synthetic route requires careful attention to reaction parameters to maximize yield and purity. The process begins with the alkylation of the enamine in acetonitrile, followed by a workup that involves aqueous extraction and solvent evaporation to isolate the ketone intermediate. Subsequent deprotection using trifluoroacetic acid must be carefully quenched to prevent degradation of the acid-sensitive groups. The cyclization step demands rigorous water removal, often achieved through Dean-Stark apparatus or molecular sieves, to push the reaction to completion. Finally, the coupling of the indole core with the proline-like side chain utilizes standard peptide coupling reagents such as DCC and HOBt, followed by a catalytic hydrogenation step to saturate the double bond and finalize the stereochemistry.
- Alkylate 1-(1-cyclohexen-1-yl)-pyrrolidine with a protected amino acid iodide derivative in acetonitrile under reflux to form the ketone intermediate.
- Deprotect the amine group using trifluoroacetic acid, followed by acid-catalyzed cyclization with p-toluenesulfonic acid in toluene to form the hexahydroindole core.
- Couple the indole core with N-[(S)-ethoxycarbonyl-1-butyl]-(S)-alanine using DCC/HOBt, followed by catalytic hydrogenation to establish the final stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and operational continuity, the advantages of this patented process are manifold. The shift away from resolution-dependent starting materials fundamentally alters the cost structure of Perindopril production. By utilizing commodity-grade enamines and avoiding the expensive enzymatic or chromatographic resolutions required in older methods, manufacturers can achieve substantial cost savings in raw material procurement. Furthermore, the high yields reported in the patent examples—ranging from 80% to 97% across the initial steps—indicate a highly efficient process that minimizes waste and maximizes throughput. This efficiency is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing suppliers to respond more rapidly to market fluctuations and urgent procurement needs.
- Cost Reduction in Manufacturing: The elimination of complex resolution steps and the use of readily available starting materials significantly lower the barrier to entry for production. The process avoids the need for specialized chiral catalysts or expensive resolving agents that characterize legacy routes. Instead, it relies on robust, scalable chemistry that utilizes standard reagents like p-toluenesulfonic acid and common solvents. This simplification of the bill of materials (BOM) directly contributes to a lower cost of goods, enabling more competitive pricing strategies in the global generic pharmaceutical market without compromising on quality standards.
- Enhanced Supply Chain Reliability: Dependence on single-source suppliers for exotic intermediates is a major risk factor in pharmaceutical supply chains. This new route mitigates that risk by relying on 1-(1-cyclohexen-1-yl)-pyrrolidine and standard amino acid derivatives, which are produced by multiple chemical manufacturers globally. This diversification of the supply base ensures greater continuity of supply and reduces the likelihood of production stoppages due to raw material shortages. Additionally, the robustness of the reaction conditions means that the process is less sensitive to minor variations in reagent quality, further stabilizing the supply chain against upstream volatility.
- Scalability and Environmental Compliance: The synthetic steps described are inherently scalable, utilizing unit operations such as reflux, distillation, and filtration that are standard in multi-purpose chemical plants. The avoidance of heavy metal catalysts in the early stages (prior to the final hydrogenation) simplifies waste treatment and reduces the environmental footprint of the manufacturing process. The high atom economy of the cyclization step, which primarily generates water as a byproduct, aligns well with modern green chemistry principles. This makes the process not only economically attractive but also easier to permit and operate within increasingly strict environmental regulatory frameworks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for commercial production. The answers are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making.
Q: What is the primary advantage of this new Perindopril synthesis route over conventional methods?
A: The primary advantage is the use of readily available starting materials, specifically 1-(1-cyclohexen-1-yl)-pyrrolidine, which eliminates the need for the expensive and hard-to-source (2S,3aS,7aS)-octahydroindole-2-carboxylate required in older patents like EP 0 308 341.
Q: How does this process ensure high optical purity for the final API?
A: High optical purity is maintained by introducing the chiral center early in the synthesis via a protected chiral amino acid iodide derivative. The subsequent cyclization and hydrogenation steps preserve this stereochemical integrity, achieving up to 99% enantiomeric purity.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method is designed for industrial scalability. It utilizes robust reaction conditions such as reflux in common solvents like acetonitrile and toluene, and avoids complex resolution steps, resulting in high overall yields suitable for metric ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Perindopril Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one detailed in CN1753906A for the global supply of cardiovascular medications. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of critical intermediates. Our state-of-the-art facilities are equipped to handle the specific requirements of this chemistry, including rigorous moisture control for the cyclization steps and specialized hydrogenation capabilities for the final stereochemical adjustments. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite potential partners to engage with our technical team to explore how this optimized synthesis route can benefit your specific supply chain requirements. By leveraging our expertise, you can secure a Customized Cost-Saving Analysis tailored to your production volumes and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, ensuring that your transition to this advanced manufacturing process is seamless and commercially successful.
