Advanced Organocatalytic Synthesis of Optically Active Tetrahydroquinoline Derivatives for Commercial Pharmaceutical Applications
Advanced Organocatalytic Synthesis of Optically Active Tetrahydroquinoline Derivatives for Commercial Pharmaceutical Applications
The development of efficient, scalable, and highly stereoselective synthetic routes for nitrogen-containing heterocycles remains a cornerstone of modern medicinal chemistry. Patent CN102766092A introduces a groundbreaking methodology for the synthesis of optically active tetrahydroquinoline derivatives, utilizing a chiral spiro phosphoric acid as a highly effective organocatalyst. This technology addresses critical bottlenecks in the production of complex pharmaceutical intermediates by enabling reactions under exceptionally mild conditions, ranging from -30°C to 50°C, while achieving superior enantiomeric excess. For R&D directors and process chemists, this represents a significant leap forward in accessing biologically active scaffolds that are prevalent in numerous high-value therapeutic agents and natural products. The robustness of this catalytic system allows for the transformation of simple starting materials—aldehydes, arylamines, and N-benzyloxycarbonyl ethylene amines—into complex chiral architectures with remarkable precision.
From a commercial manufacturing perspective, the implications of this patent are profound, particularly regarding supply chain stability and cost efficiency. The ability to utilize a metal-free organocatalytic approach fundamentally alters the purification landscape, removing the necessity for rigorous heavy metal clearance steps that often plague transition-metal catalyzed processes. This not only streamlines the production workflow but also aligns perfectly with the increasingly stringent regulatory requirements for residual metals in active pharmaceutical ingredients (APIs). As a reliable pharmaceutical intermediate supplier, understanding and implementing such advanced catalytic technologies is essential for maintaining a competitive edge in the global market. The following analysis delves into the technical nuances of this invention, contrasting it with legacy methods and highlighting its potential for large-scale commercialization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active tetrahydroquinoline derivatives has relied heavily on the asymmetric Povarov reaction or variations thereof, often employing chiral Lewis acids or transition metal complexes. While these traditional methods have served the industry for decades, they are fraught with significant limitations that hinder their applicability in modern, high-throughput drug discovery and manufacturing environments. A primary drawback is the narrow substrate scope; many conventional catalysts exhibit poor activity or negligible enantioselectivity when challenged with aliphatic aldehydes or sterically hindered substrates, forcing chemists to resort to lengthy linear syntheses or resolution techniques that drastically reduce overall yield. Furthermore, the reliance on sensitive metal catalysts often necessitates strictly anhydrous conditions, cryogenic temperatures well below -78°C, and the use of hazardous reagents, all of which escalate operational costs and safety risks. The downstream processing is equally burdensome, as removing trace amounts of toxic metals like palladium, rhodium, or copper to meet ppm-level specifications requires specialized scavengers and additional unit operations, creating a bottleneck in the supply chain.
The Novel Approach
In stark contrast, the methodology disclosed in CN102766092A leverages the unique structural properties of chiral spiro phosphoric acids to overcome these historical hurdles. This novel approach utilizes a Brønsted acid catalysis mechanism that activates the imine intermediate through a dual hydrogen-bonding network, facilitating a highly organized transition state that ensures exceptional stereocontrol. The reaction proceeds efficiently in common organic solvents such as dichloromethane, chloroform, or toluene, without the need for extreme cryogenic conditions, typically operating comfortably between -30°C and 50°C. This tolerance for milder temperatures significantly reduces energy consumption and simplifies reactor requirements, making the process inherently safer and more scalable. Moreover, the catalyst demonstrates remarkable versatility, successfully accommodating a diverse array of substrates including those with electron-withdrawing nitro groups, halogens, and even bulky aliphatic chains, consistently delivering high yields and enantiomeric excess values often exceeding 90%.
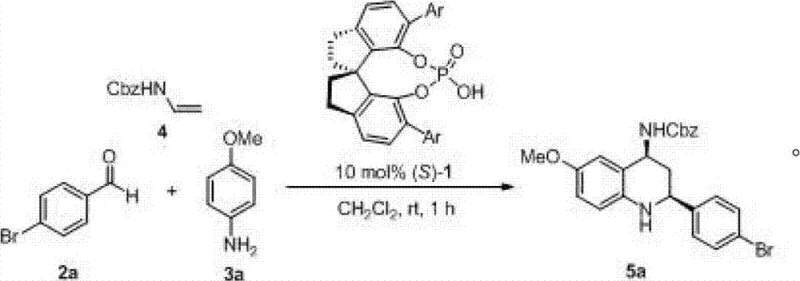
The generalized reaction pathway illustrates the convergence of three readily available components: an aldehyde, an aromatic amine, and an N-protected vinyl amine. This three-component coupling strategy is atom-economical and convergent, allowing for rapid diversification of the chemical library. By simply varying the R1 and R2 substituents on the starting materials, manufacturers can access a vast array of structurally distinct tetrahydroquinoline cores without re-optimizing the entire synthetic route. This modularity is a key advantage for contract development and manufacturing organizations (CDMOs) aiming to support clients through various stages of drug development, from lead optimization to commercial launch. The elimination of stoichiometric chiral auxiliaries and the use of a catalytic amount (1-15 mol%) of the chiral phosphoric acid further enhance the economic viability of this process, positioning it as a superior alternative for cost reduction in API manufacturing.
Mechanistic Insights into Chiral Spiro Phosphoric Acid Catalysis
The success of this synthetic route hinges on the sophisticated design of the chiral spiro phosphoric acid catalyst, which acts as a bifunctional activator. Unlike simple Brønsted acids that merely protonate substrates, these spirocyclic structures possess a rigid, three-dimensional framework that creates a well-defined chiral pocket around the reactive center. The mechanism likely involves the simultaneous activation of the imine electophile via hydrogen bonding with the phosphoryl oxygen and the stabilization of the nucleophilic enamine or vinyl amine species. This dual activation lowers the activation energy of the cycloaddition step while enforcing a specific spatial orientation that favors the formation of one enantiomer over the other. The steric bulk of the substituents on the binaphthyl or spiro-backbone (such as the 3,5-bis(trifluoromethyl)phenyl groups mentioned in related art) plays a critical role in shielding one face of the reacting species, thereby dictating the stereochemical outcome with high fidelity.
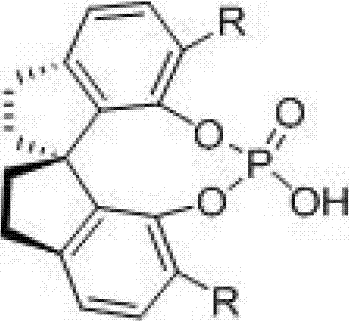
Understanding the impurity profile is crucial for R&D teams tasked with scaling this chemistry. The high diastereoselectivity observed (cis:trans > 20:1 in many cases) suggests that the transition state is tightly controlled, minimizing the formation of unwanted diastereomers that are notoriously difficult to separate. The primary impurities typically arise from hydrolysis of the imine intermediate or polymerization of the vinyl amine, both of which can be mitigated by controlling the water content in the solvent and optimizing the addition rate of reagents. The robustness of the catalyst against moisture and air, compared to sensitive metal complexes, further contributes to a cleaner reaction profile. This mechanistic clarity allows process chemists to implement robust in-process controls (IPCs), ensuring consistent quality batch after batch. The ability to predict and control the stereochemistry at the C2 and C4 positions of the tetrahydroquinoline ring is invaluable for synthesizing specific biological isomers required for clinical efficacy.
How to Synthesize Optically Active Tetrahydroquinoline Derivatives Efficiently
Implementing this catalytic protocol in a laboratory or pilot plant setting requires attention to detail regarding reagent quality and reaction monitoring. The process begins with the in situ formation of the imine from the aldehyde and amine components, followed by the introduction of the vinyl amine and the chiral catalyst. Maintaining an inert atmosphere is recommended to prevent oxidation of sensitive intermediates, although the catalyst itself shows reasonable stability. Reaction progress can be effectively monitored using thin-layer chromatography (TLC) or HPLC, with typical completion times ranging from 1 to 5 hours depending on the electronic nature of the substrates. Work-up generally involves quenching the reaction, extraction, and purification via standard silica gel chromatography, yielding the target compound as a white solid with high optical purity.
- Prepare the reaction mixture by combining aldehyde, aromatic amine, and N-benzyloxycarbonyl ethylene amine in an organic solvent such as dichloromethane under nitrogen protection.
- Add the chiral spiro phosphoric acid catalyst (1-15 mol%) to the mixture and maintain the reaction temperature between -30°C and 50°C for 1 to 5 hours.
- Upon completion, perform standard work-up procedures including separation and purification via silica gel column chromatography to isolate the optically active product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this organocatalytic technology offers tangible benefits that extend beyond mere chemical elegance. The shift from metal-based catalysis to metal-free organocatalysis fundamentally reshapes the cost structure of manufacturing complex heterocycles. By eliminating the need for expensive precious metal catalysts and the associated ligands, raw material costs are significantly reduced. More importantly, the removal of heavy metals from the process flow negates the requirement for costly metal scavenging resins and the extensive analytical testing required to certify low residual metal levels. This simplification of the downstream processing train leads to substantial cost savings in terms of both materials and labor, directly impacting the bottom line of commercial production campaigns.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the high turnover number of the catalyst and the avoidance of precious metals. Traditional methods often require loadings of expensive rhodium or palladium complexes that cannot be recovered, whereas the chiral phosphoric acid can sometimes be recovered or is used in relatively low loadings (1-15 mol%) relative to the value of the product. Furthermore, the mild reaction conditions (-30°C to 50°C) imply lower energy demands for heating and cooling compared to cryogenic processes, resulting in reduced utility costs. The simplified purification process, which avoids complex metal removal steps, shortens the overall cycle time per batch, increasing facility throughput and asset utilization without requiring capital investment in new equipment.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. Aldehydes, anilines, and protected vinyl amines are widely available from multiple global suppliers, reducing the risk of single-source dependency. The catalyst itself, while specialized, is based on a scaffold that can be synthesized from commercially available binaphthol derivatives, ensuring long-term availability. The robustness of the reaction to minor variations in conditions means that production is less susceptible to delays caused by strict environmental controls or reagent quality fluctuations. This reliability is critical for maintaining continuous supply to downstream API manufacturers, preventing stock-outs that could disrupt clinical trials or commercial launches.
- Scalability and Environmental Compliance: Scaling this process from gram to multi-ton quantities is facilitated by the absence of hazardous reagents and the use of standard organic solvents. The reaction does not generate toxic metal waste streams, simplifying wastewater treatment and disposal compliance, which is a growing concern for chemical manufacturers facing stricter environmental regulations. The high atom economy of the three-component coupling minimizes waste generation, aligning with green chemistry principles. This environmental compatibility not only reduces disposal costs but also enhances the corporate sustainability profile of the manufacturing site, a key factor for pharmaceutical companies seeking eco-friendly supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral spiro phosphoric acid catalyzed synthesis. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What are the primary advantages of using chiral spiro phosphoric acid over traditional metal catalysts?
A: The use of chiral spiro phosphoric acid eliminates the need for toxic transition metals, thereby simplifying downstream purification processes and ensuring the final pharmaceutical intermediate meets stringent heavy metal residue specifications without expensive scavenging steps.
Q: Does this catalytic method support a broad range of substrate types?
A: Yes, unlike many conventional methods that struggle with aliphatic aldehydes, this protocol demonstrates excellent versatility and high enantioselectivity across a wide variety of substrates, including those with electron-withdrawing or electron-donating groups.
Q: What are the typical reaction conditions required for this synthesis?
A: The reaction operates under remarkably mild conditions, typically proceeding at temperatures ranging from -30°C to 50°C in common organic solvents like dichloromethane, completing within 1 to 5 hours with high yields.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of advanced catalytic technologies in driving innovation within the pharmaceutical sector. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries like the chiral spiro phosphoric acid catalyzed synthesis of tetrahydroquinolines can be successfully translated into robust industrial processes. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation including chiral HPLC and NMR. Our capability to handle complex asymmetric syntheses allows us to offer customized solutions that optimize both yield and enantiomeric purity, catering to the specific needs of global drug developers.
We invite you to collaborate with us to leverage this cutting-edge synthetic methodology for your next project. Whether you require a Customized Cost-Saving Analysis to evaluate the economic feasibility of switching to this organocatalytic route or need specific COA data to verify the quality of our pilot batches, our technical procurement team is ready to assist. Contact us today to discuss route feasibility assessments and discover how our expertise in chiral synthesis can accelerate your drug development timeline while reducing overall manufacturing costs.
