Advanced Chiral Salen Catalysts for Enantioselective Epoxidation and Commercial Scale-up
Introduction to Breakthrough Epoxidation Technology
The landscape of asymmetric synthesis has been significantly advanced by the innovations detailed in patent CN1056612C, which introduces a robust methodology for the enantioselective epoxidation of prochiral olefins. This technology centers on the utilization of specialized Salen catalysts in conjunction with a newly identified class of electron-donating ligands, most notably isoquinoline N-oxide. For R&D directors and process chemists seeking reliable pharmaceutical intermediates supplier solutions, this patent represents a critical evolution in generating high-purity chiral epoxides, which are essential precursors for a wide array of bioactive molecules including potassium channel activators. The core breakthrough lies not just in the catalyst structure itself, but in the synergistic relationship between the transition metal complex and the specific donor ligand, which collectively overcome historical limitations regarding reaction rates and stereochemical control.
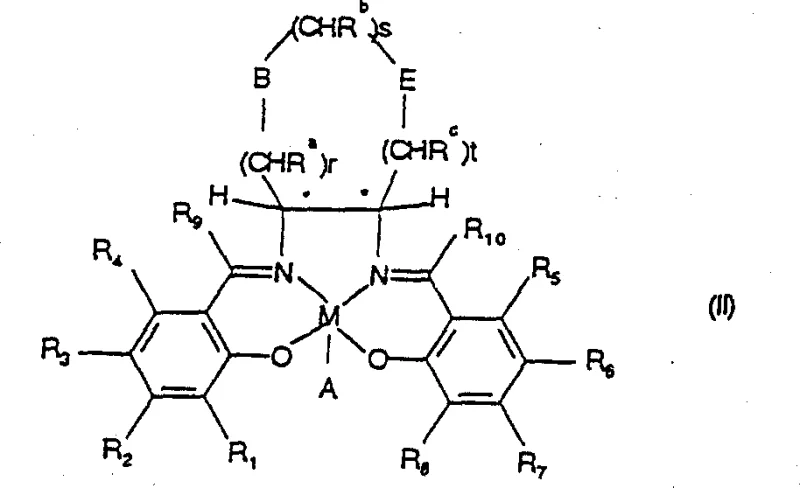
Historically, the production of enriched chiral epoxides required harsh conditions or expensive stoichiometric oxidants, often resulting in poor atom economy and difficult downstream processing. The method described in CN1056612C shifts this paradigm by enabling the use of inexpensive oxygen sources like sodium hypochlorite under mild conditions, typically ranging from 0°C to 40°C. This capability is paramount for the cost reduction in pharmaceutical intermediate manufacturing, as it allows for the safe and efficient production of sensitive intermediates without degrading thermally labile functional groups. Furthermore, the patent outlines a versatile family of catalysts, designated as Formulas I, IA, IB, II, and III, providing process engineers with a toolkit to fine-tune reactivity for specific substrate classes, thereby ensuring consistent quality and yield across diverse synthetic campaigns.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for enantioselective epoxidation, such as those relying on early generation Salen-manganese complexes, frequently encountered substantial bottlenecks when translated to commercial scale. A primary deficiency was the reliance on donor ligands like pyridine N-oxide, which, while effective at accelerating reaction kinetics, posed severe purification challenges. In large-scale two-phase reaction systems, removing trace amounts of these polar ligands from the non-polar organic product layer often required extensive washing protocols or chromatographic steps, leading to significant yield losses and increased solvent consumption. Additionally, conventional catalysts often demanded high loading levels, sometimes exceeding 1-2 mole percent, to achieve acceptable conversion rates, which directly inflated the cost of goods due to the expense of the chiral ligand synthesis. The inability to efficiently recover and recycle these catalysts further exacerbated the economic inefficiency of traditional processes.
Moreover, earlier catalyst generations often struggled to maintain high enantiomeric excess (ee) across a broad range of substrates, particularly those with bulky substituents or electron-withdrawing groups. The rigidity of the chiral pocket in older designs was insufficient to discriminate effectively between the pro-chiral faces of complex olefins, resulting in product mixtures that required costly resolution steps. This lack of selectivity not only impacted the overall yield but also introduced impurities that could be difficult to remove to meet stringent regulatory standards for active pharmaceutical ingredients. Consequently, manufacturers faced prolonged lead times and unpredictable supply chains, hindering the rapid development of new drug candidates that relied on these chiral building blocks.
The Novel Approach
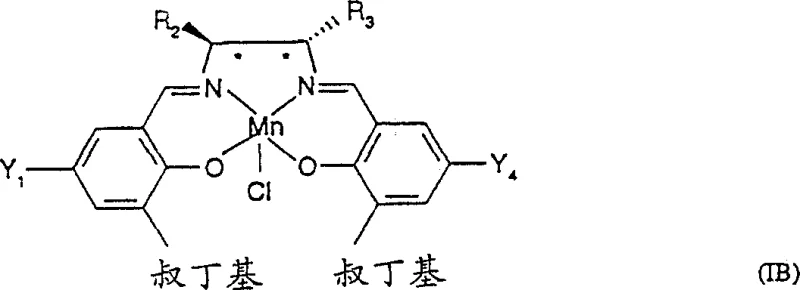
The innovative strategy presented in CN1056612C addresses these deficiencies through the introduction of isoquinoline N-oxide as a superior donor ligand. Unlike its predecessors, isoquinoline N-oxide possesses solubility characteristics that allow it to function effectively within the catalytic cycle while being significantly easier to remove from the final product stream. This improvement simplifies the workup procedure, reducing the number of unit operations required and minimizing solvent waste, which aligns perfectly with modern green chemistry initiatives. When combined with the novel Salen catalysts of Formula II and III, this ligand system demonstrates a remarkable ability to enhance both the reaction rate and the enantioselectivity simultaneously, allowing for catalyst loadings to be reduced to as low as 0.1 to 0.2 mole percent in optimized examples. This drastic reduction in catalyst usage translates directly into substantial cost savings and improved process sustainability.
Furthermore, the structural diversity of the new catalyst family allows for precise tuning of the steric and electronic environment around the manganese center. By modifying the backbone of the diamine component—incorporating rings such as tetrahydrofuran, dioxepane, or piperidine—the catalyst can be tailored to accommodate specific substrate geometries. This modularity ensures that high levels of stereocontrol can be maintained even for challenging substrates like 2,2-dimethyl-6-pentafluoroethyl-2H-1-benzopyran. The result is a process that delivers high-purity chiral epoxides with ee values consistently exceeding 99% after simple crystallization, eliminating the need for complex chiral separations. This reliability makes the technology an ideal candidate for the commercial scale-up of complex pharmaceutical additives and intermediates, providing a stable foundation for long-term production contracts.
Mechanistic Insights into Salen-Mn Catalyzed Epoxidation
The efficacy of this epoxidation process is rooted in the sophisticated coordination chemistry of the Salen-manganese complex. The catalyst operates via a mechanism where the manganese ion, typically in the +3 oxidation state, coordinates with the oxygen source (hypochlorite) to form a high-valent manganese-oxo species, which is the active oxidant. The presence of the electron-donating ligand, such as isoquinoline N-oxide, plays a critical role by coordinating axially to the manganese center. This axial coordination modifies the electronic density of the metal, facilitating the formation of the reactive oxo-species at a faster rate and stabilizing the transition state during oxygen transfer to the olefin substrate. This interaction is not merely kinetic; it profoundly influences the stereochemical outcome by locking the catalyst into a specific conformation that favors the approach of the olefin from one specific face, thereby ensuring high enantioselectivity.
Impurity control is another critical aspect managed by the mechanistic design of these catalysts. In traditional systems, side reactions such as non-selective background epoxidation or over-oxidation of the product could occur, leading to complex impurity profiles. The novel Salen catalysts described in the patent mitigate these risks by creating a highly shielded active site. The bulky tert-butyl groups on the salicylaldehyde moieties, combined with the rigid cyclic backbone of the diamine, effectively block access to the metal center for anything other than the intended substrate in the correct orientation. This steric shielding minimizes non-productive decomposition of the oxidant and prevents the formation of regio-isomers or diastereomers. Consequently, the crude reaction mixture is much cleaner, simplifying the purification process and ensuring that the final API intermediate meets the rigorous purity specifications required by global regulatory bodies.
How to Synthesize Chiral Epoxides Efficiently
Implementing this technology requires a systematic approach to reactor setup and parameter control to maximize the benefits of the novel catalyst system. The process generally involves preparing a biphasic system where the organic phase contains the prochiral olefin, the Salen-Mn catalyst, and the isoquinoline N-oxide ligand dissolved in a water-immiscible solvent like dichloromethane. The aqueous phase consists of the oxidant, typically commercial bleach (sodium hypochlorite), buffered to a specific pH range of 10 to 13 using phosphate buffers to maintain catalyst stability. Maintaining the correct pH is crucial, as deviations can lead to catalyst decomposition or reduced selectivity. The reaction is typically conducted at temperatures between 0°C and 40°C, with vigorous stirring to ensure efficient mass transfer between the two phases, which is often the rate-limiting step in biphasic catalysis.
- Prepare the reaction system by establishing a two-phase mixture of an water-immiscible organic solvent (such as dichloromethane) containing the prochiral olefin substrate and the Salen-Mn catalyst, and an aqueous phase containing the oxygen source (sodium hypochlorite) buffered to a pH between 10 and 13.
- Introduce the electron-donating ligand, specifically isoquinoline N-oxide or a compound with similar solubility characteristics, into the organic phase to coordinate with the transition metal center and enhance both reaction rate and enantioselectivity.
- Maintain the reaction temperature between 0°C and 40°C while stirring vigorously to ensure efficient mass transfer between phases, followed by separation, washing, and crystallization to isolate the high-purity chiral epoxide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this epoxidation technology offers compelling strategic advantages that extend beyond simple technical performance. The primary value proposition lies in the significant optimization of the cost structure associated with chiral synthesis. By enabling the use of lower catalyst loadings and cheaper oxidants like sodium hypochlorite, the direct material costs are drastically reduced compared to processes requiring stoichiometric chiral oxidants or precious metal catalysts. Furthermore, the ease of ligand removal reduces the consumption of purification solvents and the time spent on downstream processing, which are often hidden cost drivers in fine chemical manufacturing. This efficiency gain allows for more competitive pricing models for the final intermediates, enhancing the margin potential for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The ability to operate with catalyst loadings as low as 0.1 mole percent represents a paradigm shift in economic efficiency for asymmetric catalysis. Traditional methods often required 10 to 20 times more catalyst to achieve similar conversions, representing a massive expenditure on chiral ligands which are expensive to synthesize. By minimizing this input, the overall cost of goods sold is significantly lowered. Additionally, the use of commodity chemicals like bleach and dichloromethane, rather than specialized peracids or metal oxidants, ensures that raw material costs remain stable and predictable, shielding the supply chain from volatility in the pricing of exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of the catalyst system contributes directly to supply security. The catalysts are stable under ambient storage conditions and the reaction tolerates minor fluctuations in operating parameters better than fragile enzymatic or organocatalytic systems. This resilience reduces the risk of batch failures, which can cause significant delays in production schedules. Moreover, the starting materials for the catalyst synthesis, such as substituted salicylaldehydes and chiral diamines, are readily available from multiple global suppliers, preventing single-source bottlenecks. This diversification of the supply base ensures continuous availability of the catalyst, supporting uninterrupted manufacturing campaigns for critical pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations familiar to any multipurpose chemical plant. The biphasic nature of the reaction simplifies heat management, a critical safety factor when scaling exothermic oxidations. From an environmental perspective, the reduction in catalyst waste and the use of less hazardous oxidants align with increasingly strict environmental regulations. The simplified workup reduces the volume of aqueous and organic waste streams requiring treatment, lowering disposal costs and improving the overall environmental footprint of the manufacturing site. This compliance facilitates smoother regulatory approvals and enhances the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxidation technology. These insights are derived directly from the experimental data and claims within patent CN1056612C, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of integrating this route into their existing process portfolios. The answers highlight the versatility of the catalyst system and its compatibility with various substrate classes, reinforcing its value as a platform technology for chiral synthesis.
Q: What is the primary advantage of using isoquinoline N-oxide over pyridine N-oxide in this epoxidation process?
A: Isoquinoline N-oxide offers superior solubility characteristics that facilitate its complete removal from the final epoxide product, addressing a significant purification challenge associated with traditional donor ligands in large-scale two-phase reactions.
Q: How does the novel Salen catalyst structure improve enantioselectivity compared to prior art?
A: The novel Salen catalysts, particularly those of Formula II and III featuring specific cyclic backbones like tetrahydrofuran or dioxepane rings, create a more rigid and defined chiral environment around the manganese center, resulting in significantly higher enantiomeric excess (ee) values.
Q: Can this process be scaled for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard industrial reagents like sodium hypochlorite and common solvents like dichloromethane in a biphasic system, making it highly amenable to commercial scale-up from kilogram to multi-ton production levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Salen Catalyst Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the epoxidation technologies described in CN1056612C for the production of high-value chiral intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to industrial reality is seamless. Our facilities are equipped with state-of-the-art reactors capable of handling biphasic oxidation chemistries safely and efficiently, adhering to stringent purity specifications and rigorous QC labs to guarantee product quality. We understand that consistency is key in pharmaceutical supply, and our robust quality management systems are designed to deliver batch-after-batch reproducibility that meets the exacting standards of global regulatory agencies.
We invite you to engage with our technical procurement team to discuss how we can tailor this catalytic epoxidation process to your specific molecular targets. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this novel catalyst system or need specific COA data to validate the purity profiles of our pilot batches, we are ready to assist. Our experts can provide detailed route feasibility assessments to identify potential scale-up risks early in the development cycle, ensuring a smooth and accelerated path to market. Partner with us to leverage this advanced chemistry and secure a competitive edge in the supply of high-purity chiral epoxides.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
