Advanced Synthesis of Novel Naphthalimide Derivatives for High-Purity Antitumor API Manufacturing
The landscape of antitumor drug development has long recognized the potential of naphthalimide derivatives, yet the quest for compounds with superior efficacy and manageable toxicity profiles continues to drive innovation in medicinal chemistry. Patent CN101575315A introduces a significant advancement in this field by disclosing a series of novel naphthalimide derivatives, specifically designed to enhance DNA intercalation capabilities and cytotoxicity against resistant tumor strains. Building upon the foundational work of early compounds like Amonafide and Mitonafide, which established the critical role of the naphthalimide chromophore in inhibiting DNA and RNA synthesis, this invention leverages structural modifications to the imide side chain and the naphthalene nucleus. The disclosed compounds, characterized by specific substitutions at the 4, 5, 7, 8, or 9 positions and varied amine linkers, represent a strategic evolution from mono-intercalators to more potent bis-intercalating agents. For research and development teams seeking high-purity pharmaceutical intermediates, this technology offers a robust platform for generating next-generation anticancer candidates with demonstrated activity superior to standard controls like 5-Fluorouracil in vitro assays.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of naphthalimide-based therapeutics has been constrained by the limitations of traditional synthetic routes which often yield mono-intercalating structures with moderate potency. Conventional methods typically rely on direct alkylation of naphthalic anhydride with simple amines, resulting in compounds that may lack the necessary conjugate plane size or side-chain flexibility to effectively penetrate multidrug-resistant tumor cells. Furthermore, older synthesis pathways frequently involve harsh conditions or expensive catalysts that complicate scale-up and introduce difficult-to-remove impurities, posing significant challenges for cost reduction in API manufacturing. The structural rigidity of many first-generation derivatives limits their ability to adapt to the DNA helix, reducing their binding affinity and overall therapeutic index. Consequently, there is a persistent industry need for more versatile synthetic strategies that allow for the precise tuning of side-chain length and terminal functional groups to optimize pharmacokinetic properties without compromising yield or purity.
The Novel Approach
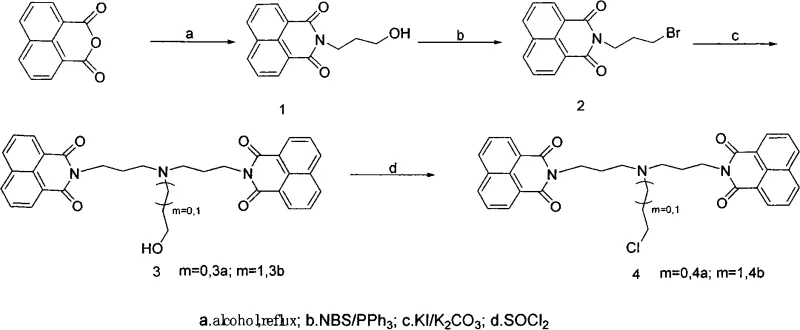
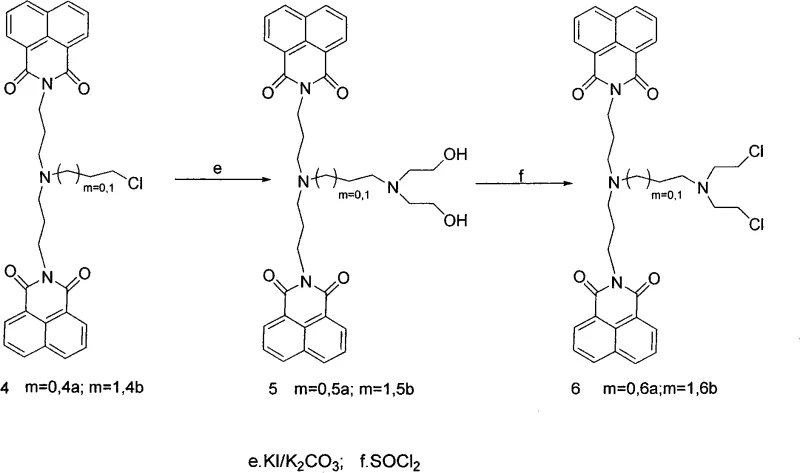
The methodology presented in this patent overcomes these hurdles through a modular synthetic strategy that prioritizes flexibility and efficiency. By utilizing naphthalic anhydride and hydroxyamines as primary building blocks, the process enables the systematic extension of the amine chain and the introduction of diverse derivatizing groups. This approach facilitates the creation of both symmetric and asymmetric bis-naphthalimide structures, such as the highly active Compound 3b and Compound 5b, which exhibit IC50 values in the nanomolar range against human colon cancer cells. The novelty lies in the stepwise conversion of hydroxyl terminals into reactive leaving groups (bromine or chlorine), allowing for subsequent nucleophilic substitution with various diamines or amino-alcohols. This controlled assembly not only enhances the conjugate plane interaction via pi-stacking forces but also improves solubility and cellular uptake. As a reliable pharmaceutical intermediate supplier, adopting this route allows for the rapid generation of structural analogs to probe structure-activity relationships (SAR) effectively.
Mechanistic Insights into Nucleophilic Substitution and Intercalation
The core chemical mechanism driving the efficacy of these derivatives involves a dual action of DNA intercalation and potential covalent binding, facilitated by the specific architecture of the side chains. The planar naphthalimide moiety inserts itself between DNA base pairs through pi-pi stacking interactions, a process that is thermodynamically favored when the conjugate system is sufficiently large and planar. The patent highlights that replacing the naphthalene nucleus with larger systems or optimizing the side chain length (n and m variables) significantly boosts this interaction. Chemically, the synthesis relies on a classic nucleophilic substitution mechanism where the nitrogen atom of the amine linker attacks the electrophilic carbon adjacent to the leaving group (bromine or chlorine) on the naphthalimide precursor. This reaction is catalyzed by bases like potassium carbonate in polar aprotic solvents such as acetonitrile, ensuring high conversion rates. The presence of hydroxyl or chloroethyl terminals on the side chains further modulates the compound's reactivity, potentially allowing for cross-linking with DNA or enhancing water solubility for better bioavailability.
From an impurity control perspective, the mechanism offers distinct advantages due to the crystallinity and polarity differences between intermediates and byproducts. The conversion of hydroxyl groups to halides using reagents like thionyl chloride or N-bromosuccinimide (NBS) is highly selective, minimizing the formation of regioisomers that often plague aromatic substitutions. The subsequent coupling steps are driven to completion by the use of excess amine reagents or iodide catalysts (KI), which accelerate the displacement of the halide leaving groups. This kinetic control ensures that the final products, such as the bis-naphthalimides, are formed with high regioselectivity. For R&D directors focused on purity, understanding this mechanistic pathway is crucial for designing effective purification protocols, such as the silica gel column chromatography described in the examples, which effectively separates the target molecules from unreacted starting materials and minor side products, ensuring the stringent purity specifications required for clinical grade materials.
How to Synthesize Novel Naphthalimide Derivatives Efficiently
The synthesis of these potent antitumor agents follows a logical, multi-step sequence that balances reaction efficiency with operational simplicity, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates. The process begins with the condensation of naphthalic anhydride with hydroxyamines under reflux conditions to establish the core imide structure with a functionalized terminal hydroxyl group. This intermediate is then activated via bromination, transforming the hydroxyl into a reactive bromide capable of undergoing nucleophilic attack. The detailed standardized synthesis steps below outline the precise conditions, stoichiometry, and workup procedures required to achieve the high yields reported in the patent examples, ensuring reproducibility from gram-scale laboratory experiments to multi-kilogram production runs.
- Condense naphthalic anhydride with hydroxyamines under reflux to form the initial imide structure with a hydroxyl terminal.
- Convert the hydroxyl group into a better leaving group (bromine) using N-bromosuccinimide (NBS) and triphenylphosphine.
- Perform nucleophilic substitution with diamines or amino-alcohols, followed by optional chlorination with thionyl chloride to finalize the side chain.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route presents compelling economic and logistical benefits that directly impact the bottom line and operational continuity. The reliance on commodity chemicals such as naphthalic anhydride, thionyl chloride, and common diamines eliminates the dependency on scarce or geopolitically sensitive raw materials, thereby enhancing supply chain reliability. Furthermore, the avoidance of precious metal catalysts, which are often required in cross-coupling reactions for similar scaffolds, results in significant cost reduction in API manufacturing by removing the need for expensive metal scavenging steps and reducing heavy metal residue concerns in the final product. The robustness of the reaction conditions, which tolerate standard heating and stirring without requiring cryogenic temperatures or high-pressure equipment, simplifies the engineering requirements for production facilities.
- Cost Reduction in Manufacturing: The synthetic pathway utilizes inexpensive, bulk-available reagents and avoids the use of costly transition metal catalysts, which drastically simplifies the downstream processing and purification stages. By eliminating the need for specialized metal removal resins or complex filtration systems, the overall production cost is substantially lowered, allowing for more competitive pricing of the final active pharmaceutical ingredients. Additionally, the high yields reported in the patent examples indicate efficient atom economy, reducing waste disposal costs and maximizing the output per batch of raw materials consumed.
- Enhanced Supply Chain Reliability: The starting materials and reagents identified in this process are widely produced by multiple global chemical suppliers, mitigating the risk of single-source bottlenecks or supply disruptions. The synthetic steps are chemically robust and do not require highly specialized or fragile reagents that might have short shelf-lives or strict storage requirements. This stability ensures consistent production schedules and reduces lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond quickly to market demands and clinical trial requirements without fear of raw material shortages.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory flasks to industrial reactors due to the use of standard unit operations like reflux, filtration, and solvent extraction. The waste streams generated are primarily organic solvents and inorganic salts, which can be managed through established recovery and treatment protocols, aligning with modern environmental compliance standards. The absence of toxic heavy metals in the reaction mixture further simplifies wastewater treatment and regulatory approval processes, facilitating a smoother path to commercial production and reducing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel naphthalimide derivatives, based on the detailed specifications and experimental data provided in the patent documentation. These insights are intended to clarify the feasibility of integrating this technology into existing drug development pipelines and to highlight the specific advantages it offers over conventional synthetic methods. Understanding these details is essential for stakeholders evaluating the potential of these compounds for oncology applications.
Q: What distinguishes these new naphthalimide derivatives from existing drugs like Amonafide?
A: The novel derivatives described in CN101575315A feature optimized side chains and potential bis-intercalating structures that demonstrate significantly higher antitumor activity (lower IC50 values) against cell lines like MCF-7 and A549 compared to traditional monomers.
Q: Is the synthesis route scalable for commercial production?
A: Yes, the process utilizes readily available starting materials like naphthalic anhydride and standard reagents such as NBS and thionyl chloride, avoiding expensive transition metal catalysts, which facilitates cost reduction in API manufacturing and ensures supply chain reliability.
Q: How is purity controlled in this synthesis?
A: The patent outlines rigorous purification methods including column chromatography and recrystallization, ensuring high-purity pharmaceutical intermediates suitable for stringent QC labs and clinical development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalimide Derivative Supplier
As the demand for advanced oncology therapeutics grows, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and unparalleled manufacturing expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, guaranteeing that your project transitions smoothly from preclinical discovery to full-scale market supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of naphthalimide intermediate delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with our technical team to explore the full potential of this patented synthesis for your specific drug candidates. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your R&D and supply chain planning, ensuring a secure and efficient supply of these critical antitumor intermediates.
