Revolutionizing Sulfonylurea Herbicide Production via Safe Sulfuryl Chloride Routes
Revolutionizing Sulfonylurea Herbicide Production via Safe Sulfuryl Chloride Routes
The global demand for high-efficiency herbicides continues to drive innovation in agrochemical intermediate manufacturing, specifically within the realm of sulfonylurea compounds. Patent CN1045205C introduces a transformative synthetic methodology that addresses critical safety and economic bottlenecks inherent in traditional production routes. This intellectual property outlines a robust process for preparing compounds of Formula I, which serve as potent plant protection agents with significant herbicidal activity. By shifting away from hazardous precursors, this technology enables manufacturers to achieve superior purity profiles while mitigating operational risks. The structural versatility of Formula I allows for extensive substitution patterns, accommodating various alkyl, alkoxy, and heterocyclic groups to tune biological efficacy. 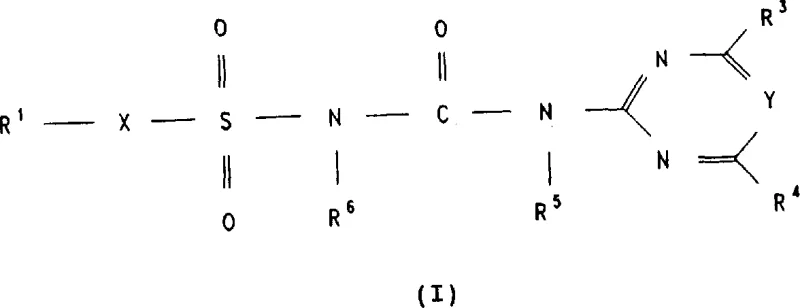 For R&D directors and process chemists, understanding the nuances of this patent is essential for developing next-generation crop protection solutions that meet stringent regulatory and safety standards.
For R&D directors and process chemists, understanding the nuances of this patent is essential for developing next-generation crop protection solutions that meet stringent regulatory and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of sulfonylurea herbicides has relied heavily on the use of Sulfuryl Chloride Isocyanate (CSI) as a key building block. While chemically effective, CSI is characterized by its hypergolic nature and high energy content, which presents severe industrial safety challenges during storage, transport, and reaction handling. The acquisition of CSI is often difficult and costly due to these inherent dangers, creating a supply chain vulnerability for manufacturers dependent on this reagent. Furthermore, the exothermic nature of reactions involving CSI requires rigorous temperature control and specialized equipment to prevent runaway scenarios, thereby increasing capital expenditure. From an economic perspective, the high cost of CSI directly inflates the cost of goods sold (COGS), making the final herbicide less competitive in price-sensitive agricultural markets. Consequently, reliance on this conventional pathway is increasingly viewed as unfavorable for large-scale industrial production from both safety and economic viewpoints.
The Novel Approach
The methodology disclosed in CN1045205C offers a paradigm shift by utilizing readily available and safer raw materials to construct the sulfonylurea backbone. Instead of pre-formed CSI, the process generates the reactive species in situ through the interaction of sulfonamide derivatives (Formula II), isocyanate salts or esters (Formula III), and sulfuryl chloride (Formula IV). This strategic substitution eliminates the need to handle bulk quantities of hazardous isocyanates, drastically simplifying the safety protocols required for operation. The reaction proceeds effectively under moderate conditions, allowing for the synthesis of complex heterocyclic substituted sulfonylureas with high efficiency. 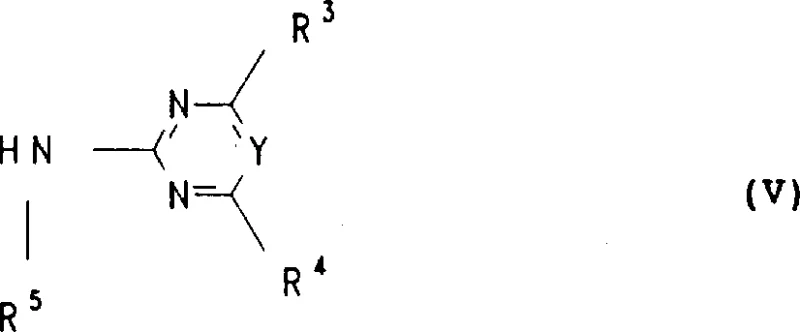 By decoupling the synthesis from unstable intermediates, this novel approach not only enhances operator safety but also opens up new possibilities for cost reduction in agrochemical intermediate manufacturing through the use of commodity chemicals.
By decoupling the synthesis from unstable intermediates, this novel approach not only enhances operator safety but also opens up new possibilities for cost reduction in agrochemical intermediate manufacturing through the use of commodity chemicals.
Mechanistic Insights into In Situ Sulfonyl Isocyanate Formation
The core mechanistic innovation of this patent lies in the generation of a sulfonyl isocyanate intermediate (R1-X-SO2-N=C=O) within the reaction vessel, rather than introducing it as a discrete reagent. The process initiates with the reaction of a sulfonamide derivative and an isocyanate source in the presence of sulfuryl chloride, likely forming an adduct that subsequently rearranges or eliminates to yield the reactive isocyanate species. This in situ generation ensures that the concentration of the highly reactive isocyanate remains low and controlled, minimizing side reactions such as polymerization or hydrolysis that often plague batch processes. The presence of sulfuryl chloride acts as a dehydrating and activating agent, facilitating the transformation of the sulfonamide nitrogen into the necessary electrophilic center. This mechanistic pathway is particularly advantageous because it tolerates a wide range of functional groups on the R1 and X moieties, including alkyl, alkenyl, and phenoxy substituents, without compromising the integrity of the sensitive isocyanate functionality.
Following the formation of the sulfonyl isocyanate intermediate, the second stage involves the nucleophilic attack by an amino-heterocycle (Formula V), typically a pyrimidine or triazine derivative. This coupling step forms the critical urea linkage (-NH-CO-NH-) that defines the biological activity of the herbicide. The patent specifies that this reaction can be conducted over a broad temperature range, from -20°C to +120°C, allowing process engineers to optimize kinetics versus selectivity. Impurity control is inherently managed by the stoichiometry of the reagents; excess sulfuryl chloride can be distilled off prior to the addition of the amine, preventing chlorination side products on the heterocyclic ring. Furthermore, the resulting product often precipitates directly from the reaction medium upon the addition of water or polar solvents, facilitating isolation in a highly purified state without the need for chromatographic purification, which is a significant advantage for industrial scalability.
How to Synthesize Sulfonylurea Compounds Efficiently
The execution of this synthesis route is designed to be flexible, supporting both solvent-free conditions and the use of inert organic solvents such as acetonitrile, toluene, or dimethylformamide. The process generally follows a two-step or multi-step sequence where the initial components are reacted to form the intermediate, followed by the addition of the heterocyclic amine to complete the molecule. Operational parameters such as stirring speed and temperature gradients are critical; for instance, vigorous stirring or ultrasonic acceleration may be employed if solubility issues arise with the starting materials. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures derived from the patent examples, are outlined below to guide process development teams in replicating this high-yield methodology.
- React sulfonamide derivatives (Formula II) with alkali metal isocyanates (Formula III) and sulfuryl chloride (Formula IV) in a polar aprotic solvent.
- Maintain reaction temperature between 0°C and 200°C to form the reactive sulfonyl isocyanate intermediate in situ.
- Add the amino-heterocycle (Formula V) to the reaction mixture at controlled temperatures (-20°C to 120°C) to finalize the urea linkage.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the methodology described in CN1045205C represents a strategic opportunity to optimize the cost structure and reliability of the herbicide supply chain. By replacing expensive and hard-to-source CSI with commodity chemicals like sulfuryl chloride and simple isocyanates, the raw material costs are significantly reduced. The ability to recover and reuse solvents and unreacted reagents quantitatively further drives down the variable costs associated with production. Additionally, the simplified safety profile reduces the insurance and compliance overheads typically associated with handling hypergolic materials. These factors combine to create a more resilient supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The elimination of Sulfuryl Chloride Isocyanate (CSI) from the bill of materials results in substantial cost savings, as CSI is a high-value specialty chemical with complex logistics. The new route utilizes sulfuryl chloride and isocyanate salts, which are produced on a massive industrial scale and are therefore significantly cheaper and more stable in price. Furthermore, the process allows for the quantitative recovery of spent solvents and excess reagents, meaning that the effective consumption of raw materials per kilogram of product is drastically lowered. This efficiency translates directly into a lower cost of goods sold, providing a competitive margin advantage in the global agrochemical market without sacrificing quality.
- Enhanced Supply Chain Reliability: Dependence on a single source for a hazardous reagent like CSI creates a single point of failure in the supply chain; by switching to widely available commodity chemicals, supply continuity is greatly enhanced. Sulfuryl chloride and common isocyanates are produced by multiple global suppliers, ensuring that procurement teams can negotiate better terms and secure backup sources easily. The robustness of the reaction conditions, which tolerate a variety of solvents and do not require cryogenic temperatures for the initial steps, also means that production can be maintained across different geographical locations with varying infrastructure capabilities. This flexibility ensures consistent delivery of high-purity agrochemical intermediates to downstream formulators.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids the thermal hazards associated with bulk CSI handling, allowing for larger batch sizes in standard stainless steel reactors. The ability to isolate the product by precipitation and filtration, rather than complex distillation or chromatography, simplifies the downstream processing equipment requirements. From an environmental standpoint, the quantitative recovery of solvents and the minimization of hazardous waste streams align with modern green chemistry principles and strict environmental regulations. This compliance reduces the risk of regulatory shutdowns and lowers the costs associated with waste treatment and disposal, making the facility more sustainable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this sulfonylurea synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of adopting this route for commercial production of herbicide intermediates.
Q: Why is the conventional CSI method considered unsafe for industrial scale-up?
A: The conventional method utilizes Sulfuryl Chloride Isocyanate (CSI), which is hypergolic and possesses high energy, posing significant industrial safety hazards and handling difficulties that complicate large-scale operations.
Q: What are the purity levels achievable with this novel sulfuryl chloride route?
A: The patented process consistently yields products with purity exceeding 94% by weight, often reaching 96% or higher, eliminating the need for expensive and time-consuming purification steps.
Q: Can solvents and unreacted reagents be recovered in this process?
A: Yes, a key advantage of this method is the quantitative recovery of non-reacted components and spent solvents, which can be mechanically separated and reused, significantly enhancing process economics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfonylurea Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting safer and more efficient synthetic routes for the production of high-value agrochemical intermediates. Our technical team has extensively analyzed the methodology presented in CN1045205C and possesses the expertise to translate this laboratory-scale innovation into robust commercial processes. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of sulfonylurea intermediate meets the exacting standards required by global crop protection leaders.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific product portfolio. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this sulfuryl chloride-based method for your specific targets. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both your supply chain security and your bottom line.
