Advanced Solvent-Free Manufacturing of Quinazolinone Polycyclic Intermediates for Global Pharma
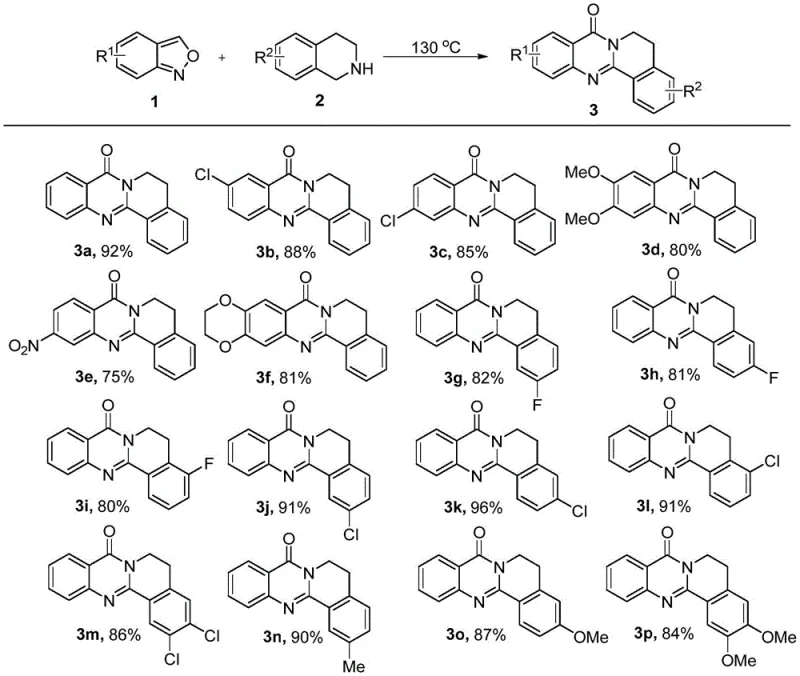
Introduction to Next-Generation Quinazolinone Synthesis
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to access biologically active heterocyclic scaffolds, and the recent disclosure in patent CN110483511B represents a significant leap forward in this domain. This groundbreaking technology introduces a novel synthetic method for quinazolinone polycyclic compounds that fundamentally alters the traditional manufacturing landscape by eliminating the need for solvents and additives. By utilizing benzisoxazole derivatives and tetrahydroisoquinoline as direct raw materials, this process achieves rapid construction of the quinazolinone nucleus under simple heating conditions. For R&D directors and process chemists, this development is particularly compelling because it addresses the critical pain points of step economy and environmental impact simultaneously. The ability to generate complex polycyclic structures in a single operational step with yields ranging from 75% to 96% suggests a paradigm shift away from laborious multi-step sequences. As a reliable quinazolinone supplier, understanding and adopting such green chemistry principles is essential for maintaining competitiveness in the global API intermediate market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of quinazolinone derivatives has been plagued by significant operational complexities and safety hazards that hinder large-scale commercial adoption. Traditional routes often rely on cyclic carbonylation reactions which necessitate the use of toxic carbon monoxide gas under high pressure, creating severe safety risks for plant operators and requiring specialized, expensive reactor infrastructure. Alternative approaches involving oxidative cyclization of anthranilamide derivatives or photocatalytic intramolecular reductive synthesis frequently suffer from long reaction times, the need for precious metal catalysts, and the requirement for specific ligands that drive up raw material costs. Furthermore, many existing protocols demand strictly anhydrous conditions or inert atmospheres, adding layers of procedural difficulty and increasing the overall cost of goods sold. These factors collectively create a bottleneck for procurement managers who are tasked with reducing lead time for high-purity pharmaceutical intermediates while managing tight budgets. The reliance on volatile organic solvents in these conventional methods also generates substantial chemical waste, complicating environmental compliance and waste disposal logistics for supply chain heads.
The Novel Approach
In stark contrast to these cumbersome legacy methods, the innovative strategy outlined in the patent data utilizes a direct thermal coupling between benzisoxazole and tetrahydroisoquinoline derivatives that proceeds remarkably efficiently without any solvent mediation. This solvent-free protocol operates under air conditions at temperatures between 100 °C and 130 °C, completing the transformation in as little as 1 hour with exceptional conversion rates. The simplicity of mixing two solid or liquid precursors and applying heat eliminates the need for complex workup procedures associated with solvent removal and catalyst filtration. This approach not only streamlines the synthetic route but also drastically improves the atom economy of the process, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing. The versatility of this method is evidenced by its tolerance to various substituents on both the benzisoxazole and tetrahydroisoquinoline rings, allowing for the rapid generation of diverse chemical libraries for drug discovery.
Mechanistic Insights into Solvent-Free Thermal Cyclization
The mechanistic underpinning of this transformation likely involves a thermally induced ring-opening of the benzisoxazole moiety followed by a nucleophilic attack by the secondary amine of the tetrahydroisoquinoline. Under the applied thermal energy of 130 °C, the weak N-O bond in the benzisoxazole ring becomes labile, generating a reactive nitrile oxide or similar electrophilic intermediate in situ. This reactive species is then intercepted by the nucleophilic nitrogen of the tetrahydroisoquinoline, initiating a cascade of cyclization events that ultimately forge the fused quinazolinone skeleton. The absence of solvent is crucial here, as the high concentration of reactants in the neat phase accelerates the reaction kinetics through increased collision frequency, effectively driving the equilibrium towards the product side. For technical teams, understanding this mechanism highlights the importance of precise temperature control to ensure complete ring opening without inducing thermal decomposition of the sensitive polycyclic product. The robustness of this mechanism allows for the accommodation of electron-withdrawing groups like nitro or chloro, as well as electron-donating groups like methoxy, without significant loss in efficiency.
Regarding impurity control, the clean nature of this solvent-free reaction minimizes the formation of side products that typically arise from solvent participation or catalyst degradation. The primary impurities are likely unreacted starting materials or minor regioisomers, which can be easily removed via standard silica gel column chromatography using a petroleum ether and ethyl acetate system. The patent examples demonstrate that even with diverse substitution patterns, such as 5-chloro or 6-nitro groups, the purification process remains straightforward, yielding analytically pure materials suitable for biological testing. This high level of purity is critical for pharmaceutical applications where strict limits on genotoxic impurities and heavy metals are enforced. By avoiding transition metal catalysts entirely, this method inherently produces a cleaner crude product profile, reducing the burden on downstream purification units and ensuring consistent quality across different batches of commercial scale-up of complex pharmaceutical intermediates.
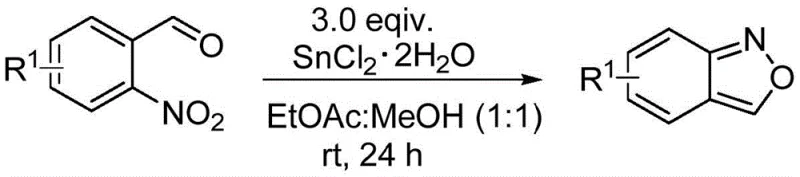
How to Synthesize Quinazolinone Polycyclic Compounds Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must adhere to specific thermal parameters to maximize yield and safety. The process begins with the precise weighing of benzisoxazole and tetrahydroisoquinoline derivatives in a strict 1:1 molar ratio to ensure complete consumption of both reagents. These materials are charged directly into a pressure-resistant reaction vessel, such as a sealed tube or autoclave, without the addition of any diluents, ligands, or drying agents. The reaction mixture is then heated to a target temperature of 130 °C and maintained under stirring for approximately 1 hour, although optimization may allow for temperatures as low as 100 °C over extended periods. Upon completion, the crude reaction mass can be directly subjected to purification, bypassing the energy-intensive steps of solvent evaporation and aqueous workup. Detailed standardized synthetic steps for this procedure are provided in the guide below to ensure reproducibility and safety compliance.
- Prepare benzisoxazole derivatives and tetrahydroisoquinoline derivatives in a 1: 1 molar ratio.
- Mix the raw materials in a pressure-resistant tube without adding any solvent or additive.
- Heat the mixture to 100-130 °C under air conditions for 0.5 to 3 hours to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solvent-free methodology offers transformative economic and logistical benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic simplification of the Bill of Materials (BOM), as the elimination of solvents removes a major cost center associated with purchasing, storing, and disposing of volatile organic compounds. This reduction in material usage directly correlates to a lower E-factor (environmental factor), meaning less waste is generated per kilogram of product, which significantly lowers waste treatment fees and environmental compliance burdens. Furthermore, the absence of transition metal catalysts eliminates the need for expensive scavenging resins or complex filtration steps to meet residual metal specifications, thereby shortening the overall production cycle time. These efficiencies combine to create a leaner, more agile manufacturing process that is highly responsive to market demands for key pharmaceutical building blocks.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the complete removal of solvent costs and the associated energy required for their recovery and distillation. In traditional batch processing, solvent purchase and disposal can account for a substantial portion of variable costs, and eliminating this line item results in significant margin expansion. Additionally, the reaction does not require specialized high-pressure equipment for carbon monoxide or inert gas lines, allowing the process to be run in standard heating manifolds which reduces capital expenditure (CAPEX) for new production lines. The high yields reported, reaching up to 96% for certain substrates, ensure that raw material utilization is maximized, further driving down the cost per kilogram of the final API intermediate. This holistic reduction in operational expenses makes the process highly attractive for generic drug manufacturers looking to optimize their supply chains.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the starting materials for this synthesis, namely benzisoxazole and tetrahydroisoquinoline derivatives, are commercially available and structurally simple, reducing the risk of supply disruptions. Unlike methods relying on exotic ligands or unstable reagents that may have long lead times, these precursors can be sourced from multiple global vendors, ensuring a robust and redundant supply network. The simplicity of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive re-validation of complex parameters. This flexibility is crucial for maintaining continuity of supply in the face of geopolitical or logistical challenges, ensuring that downstream drug production schedules are not compromised by intermediate shortages.
- Scalability and Environmental Compliance: Scaling this solvent-free reaction presents fewer engineering challenges compared to solution-phase chemistry, as issues related to solvent volume, heat transfer limitations of large solvent masses, and flammability are largely mitigated. The neat reaction mixture allows for higher throughput per unit volume of reactor space, effectively increasing the capacity of existing facilities without the need for physical expansion. Moreover, the green chemistry credentials of this method align perfectly with increasingly stringent global environmental regulations regarding VOC emissions and hazardous waste generation. By adopting this technology, companies can proactively meet sustainability goals and reduce their carbon footprint, which is becoming a key differentiator in supplier selection processes for major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel quinazolinone synthesis platform. These answers are derived directly from the experimental data and technical specifications provided in the patent literature to ensure accuracy and reliability for decision-makers. Understanding these details is vital for assessing the feasibility of integrating this method into your current production portfolio or R&D pipeline. We encourage technical teams to review these points carefully to fully appreciate the operational advantages offered by this solvent-free approach.
Q: What are the primary advantages of this solvent-free method over traditional carbonylation?
A: Unlike traditional methods requiring high CO pressure or expensive ligands, this method operates under simple heating conditions without solvents, significantly reducing safety risks and operational costs.
Q: What is the typical yield range for these quinazolinone derivatives?
A: The patent data indicates a robust yield range of 75% to 96% across various substituted derivatives, demonstrating excellent substrate tolerance.
Q: Is this process scalable for industrial production?
A: Yes, the absence of solvents and the use of simple thermal conditions make this process highly amenable to scale-up, minimizing waste treatment and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Quinazolinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of this solvent-free cyclization technology to revolutionize the production of high-value quinazolinone intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped to handle solvent-free and high-temperature reactions safely, backed by rigorous QC labs that guarantee stringent purity specifications for every batch we produce. We are committed to delivering not just chemicals, but comprehensive solutions that enhance your competitive edge in the marketplace through superior quality and reliability.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this green method can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence and precision.
