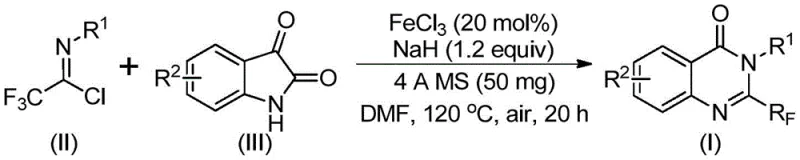
Scalable FeCl3-Catalyzed Process for High-Purity Trifluoromethyl Quinazolinones in Pharmaceutical Manufacturing
The patent CN111675662B discloses a groundbreaking methodology for synthesizing 2-trifluoromethyl-substituted quinazolinone compounds which represent critical pharmacophores in numerous therapeutic agents including anti-cancer and anti-inflammatory drug candidates This innovation addresses longstanding challenges in heterocyclic chemistry by leveraging an iron-catalyzed cascade reaction that operates under mild conditions with exceptional functional group tolerance The process utilizes inexpensive ferric chloride as a catalyst and commercially accessible starting materials including trifluoroethylimidoyl chloride and isatin derivatives enabling scalable production from laboratory to industrial scale Crucially this approach eliminates the need for precious metal catalysts and harsh reaction environments that have plagued conventional syntheses thereby enhancing both economic viability and environmental sustainability The resulting compounds exhibit superior biological activity profiles due to the strategic incorporation of the trifluoromethyl group which significantly improves metabolic stability target binding affinity and lipophilicity in drug molecules This patent thus establishes a new paradigm for manufacturing high-value quinazolinone intermediates essential for next-generation pharmaceuticals while providing a robust foundation for commercial implementation across global supply chains
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing trifluoromethyl-substituted quinazolinones have been severely constrained by multiple critical limitations including harsh reaction conditions requiring elevated temperatures above 150°C under inert atmospheres which significantly increase operational complexity and energy consumption These methods typically rely on expensive transition metal catalysts such as palladium or rhodium complexes that introduce substantial cost burdens while necessitating rigorous purification steps to remove toxic metal residues from final products Furthermore conventional syntheses employ costly trifluoromethyl synthons like trifluoroacetic anhydride which exhibit poor functional group tolerance leading to narrow substrate scope and low yields often below 50% as documented in prior literature The multi-step nature of existing protocols also generates significant waste streams requiring complex environmental management systems while limiting scalability due to exothermic reaction profiles that pose safety risks during industrial implementation These combined factors have created persistent bottlenecks in pharmaceutical intermediate supply chains particularly affecting timely delivery of high-purity materials required for clinical development stages
The Novel Approach
The patented methodology introduces a transformative solution through a streamlined single-pot process utilizing earth-abundant ferric chloride as an efficient catalyst operating under ambient atmospheric conditions without requiring specialized inert gas handling systems This innovative approach employs readily available starting materials including trifluoroethylimidoyl chloride derived from inexpensive aromatic amines and commercially accessible isatin derivatives which substantially reduce raw material costs while expanding substrate versatility across diverse functional groups The two-stage thermal protocol begins at moderate temperatures of 40°C followed by controlled heating to 120°C enabling precise reaction management that minimizes side product formation while achieving consistently high yields exceeding those of conventional methods The elimination of precious metal catalysts not only reduces production costs but also simplifies downstream purification by avoiding complex metal removal procedures thereby enhancing overall process efficiency This novel strategy demonstrates exceptional scalability potential from gram-scale laboratory validation to multi-ton industrial production while maintaining stringent quality standards required by global regulatory frameworks
Mechanistic Insights into FeCl3-Catalyzed Quinazolinone Formation
The reaction mechanism proceeds through a sophisticated cascade involving initial alkali-promoted carbon-nitrogen bond formation between trifluoroethylimidoyl chloride and isatin derivatives facilitated by sodium hydride which generates a key trifluoroacetamidine intermediate This critical step occurs under mild conditions at elevated temperatures where the base promotes nucleophilic attack while molecular sieves maintain optimal moisture control throughout the transformation Subsequent iron-catalyzed decarbonylation initiates a cyclization sequence where ferric chloride coordinates with carbonyl oxygen atoms lowering activation energy barriers through Lewis acid catalysis This coordination enables selective ring closure while simultaneously facilitating isomerization to form the stable quinazolinone core structure The catalytic cycle operates efficiently at low loadings of only 20 mol% FeCl3 demonstrating remarkable turnover frequency without requiring additional ligands or co-catalysts This mechanistic pathway represents a significant advancement over traditional methods by avoiding high-energy transition states associated with alternative cyclization approaches thereby minimizing unwanted side reactions
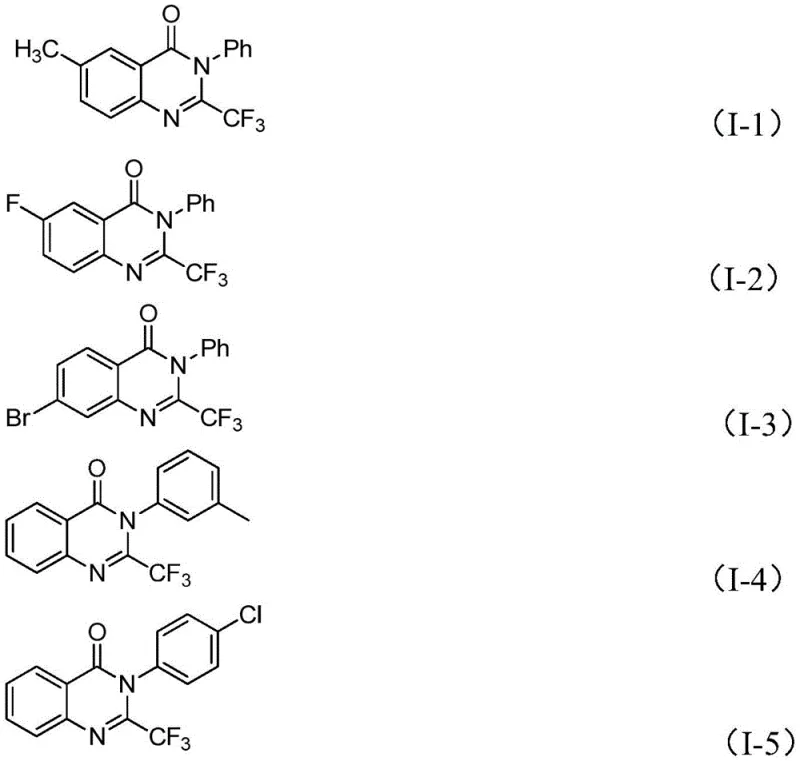
Impurity control is achieved through the method's inherent selectivity where the iron-catalyzed cyclization pathway favors formation of the desired quinazolinone regioisomer while suppressing common side products such as dimeric species or hydrolysis byproducts The excellent functional group tolerance documented across diverse aryl substitutions including halogenated methylated and methoxy variants demonstrates minimal interference from electron-donating or electron-withdrawing groups during the transformation This selectivity stems from the precise coordination chemistry between ferric chloride and carbonyl functionalities which directs reaction progression along a single dominant pathway The absence of transition metals eliminates potential metal-induced impurities while the simplified workup procedure involving filtration followed by standard column chromatography ensures removal of any residual catalyst or unreacted starting materials This robust purification profile consistently delivers products meeting pharmaceutical-grade purity specifications without requiring additional specialized cleaning steps that would otherwise complicate commercial manufacturing
How to Synthesize Trifluoromethyl Quinazolinones Efficiently
This patented methodology provides a reliable pathway for producing high-purity trifluoromethyl quinazolinones through a carefully optimized sequence that balances reactivity with operational simplicity The process begins with precise stoichiometric control where trifluoroethylimidoyl chloride is used in slight excess relative to isatin derivatives to drive complete conversion while minimizing unreacted starting material residues Critical attention must be paid to solvent selection with DMF proving optimal due to its ability to dissolve both polar and non-polar components while facilitating proton transfer during key cyclization steps The two-stage thermal profile requires meticulous temperature monitoring where initial moderate heating prevents premature decomposition while subsequent elevated temperatures enable complete ring closure Detailed standardized synthesis steps are provided below to ensure consistent results across different production scales while maintaining stringent quality control parameters throughout the manufacturing process
- Combine ferric chloride catalyst (20 mol%), sodium hydride (1.2 equiv), and activated molecular sieve in an inert atmosphere reaction vessel with DMF solvent.
- Introduce trifluoroethylimidoyl chloride and isatin derivatives at controlled stoichiometry while maintaining initial reaction temperature at 40°C.
- Execute two-stage thermal protocol involving initial stirring at moderate temperature followed by elevated temperature cyclization under ambient conditions.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative synthesis methodology delivers substantial value across procurement and supply chain operations by addressing critical pain points inherent in traditional manufacturing approaches The elimination of expensive transition metal catalysts represents a fundamental cost driver reduction while the use of readily available starting materials ensures consistent supply chain resilience against market fluctuations Furthermore the simplified process flow minimizes equipment requirements reducing capital expenditure needs while enhancing operational flexibility across different manufacturing sites The method's demonstrated scalability from laboratory validation to commercial production provides procurement teams with confidence in reliable long-term supply continuity while meeting evolving quality demands from global regulatory bodies These advantages collectively position this technology as a strategic solution for pharmaceutical manufacturers seeking sustainable competitive advantages in intermediate sourcing
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes significant material costs while simplifying purification workflows reduces solvent consumption and waste treatment expenses The use of inexpensive iron-based catalysis combined with commercially available starting materials creates substantial cost savings through reduced raw material expenditures and lower energy requirements during processing This approach also minimizes quality control costs by producing fewer impurities that would otherwise require additional analytical testing and remediation steps ultimately delivering significant economic benefits without compromising product quality standards
- Enhanced Supply Chain Reliability: Sourcing flexibility is dramatically improved through reliance on widely available starting materials that are not subject to single-source dependencies or geopolitical supply constraints The robust nature of the process allows seamless transfer between manufacturing facilities without requiring specialized equipment modifications ensuring consistent output regardless of production location Furthermore the method's tolerance to minor variations in raw material quality provides additional buffer against supply chain disruptions while maintaining product specifications This reliability translates directly into reduced lead times through more predictable production scheduling and inventory management practices
- Scalability and Environmental Compliance: The demonstrated scalability from gram-scale validation to multi-ton production ensures smooth transition from development to commercial manufacturing without requiring fundamental process re-engineering Environmental benefits arise from reduced energy consumption during milder reaction conditions along with minimized waste generation through higher atom economy The elimination of toxic metal residues simplifies waste stream management while meeting increasingly stringent regulatory requirements for sustainable manufacturing practices This combination enables pharmaceutical companies to achieve both commercial scale-up objectives and environmental stewardship goals simultaneously
Frequently Asked Questions (FAQ)
The following questions address critical technical and commercial considerations based on detailed analysis of patent CN111675662B specifications These inquiries reflect common concerns raised by procurement supply chain and R&D teams during technology evaluation processes Each response draws directly from experimental data and process descriptions documented within the patent framework ensuring accurate representation of technical capabilities and commercial implications Without further ado here are the most frequently asked questions regarding this innovative synthesis methodology
Q: How does this method overcome limitations of traditional quinazolinone synthesis?
A: The patented process eliminates harsh reaction conditions and expensive substrates by utilizing readily available starting materials with iron catalysis achieving higher yields under milder temperatures while maintaining excellent functional group tolerance compared to conventional methods requiring precious metal catalysts.
Q: What makes this synthesis suitable for commercial scale-up?
A: The methodology demonstrates robust scalability from gram-scale to industrial production through simple operation procedures cost-effective catalysts and straightforward purification techniques that ensure consistent high-purity output without specialized equipment requirements.
Q: Why is the trifluoromethyl group critical in pharmaceutical intermediates?
A: The strategic incorporation of trifluoromethyl significantly enhances metabolic stability bioavailability and lipophilicity of drug molecules directly contributing to improved therapeutic efficacy and pharmacokinetic profiles in final pharmaceutical products through electronegativity modulation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethyl Quinazolinone Supplier
Our company brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production ensuring seamless transition from laboratory validation to full-scale manufacturing operations We maintain stringent purity specifications through rigorous QC labs equipped with advanced analytical instrumentation capable of detecting impurities at trace levels required by global regulatory authorities Our technical team possesses deep expertise in optimizing complex heterocyclic syntheses including specialized knowledge in fluorinated compound manufacturing which directly supports successful implementation of this patented methodology NINGBO INNO PHARMCHEM's commitment to quality assurance provides pharmaceutical partners with confidence in consistent supply chain performance meeting even the most demanding specifications across multiple production sites worldwide
We invite you to request a Customized Cost-Saving Analysis tailored to your specific manufacturing requirements Our technical procurement team stands ready to provide detailed COA data route feasibility assessments and scalability projections upon inquiry Contact us today to explore how our expertise in high-purity intermediate production can enhance your pharmaceutical development pipeline while delivering significant operational efficiencies throughout your supply chain
