Advanced Synthesis of Prucalopride Key Intermediate via Ruthenium-Catalyzed Oxidative Cyclization for Commercial Scale-up
Advanced Synthesis of Prucalopride Key Intermediate via Ruthenium-Catalyzed Oxidative Cyclization for Commercial Scale-up
The pharmaceutical landscape for treating chronic constipation has been significantly transformed by the introduction of Prucalopride succinate, a high-affinity serotonin 5-HT4 receptor agonist marketed under the brand name Resolor. As demand for this active pharmaceutical ingredient (API) grows globally, the efficiency of its supply chain becomes paramount, particularly regarding the synthesis of its critical building block, 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid (Compound 1). Patent CN102942542A discloses a groundbreaking preparation method that utilizes a substituted p-amino m-allyl o-hydroxybenzene methyl ester as a raw material. This innovative approach leverages a series of reactions including oxidation, reduction, protection, substitution, cyclization, and hydrolysis to construct the complex dihydrobenzofuran core. By shifting away from hazardous reagents and cryogenic conditions, this technology offers a robust pathway for industrial production.
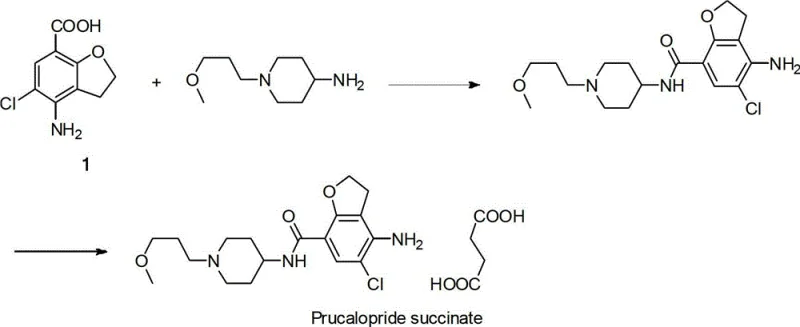
The strategic importance of this patent lies in its ability to streamline the manufacturing of high-purity pharmaceutical intermediates. Traditional methods often struggle with low yields, complex purification requirements, and the use of prohibitively expensive catalysts. In contrast, the methodology described in CN102942542A focuses on simplicity, low cost, and high conversion ratios. For procurement managers and supply chain directors, this represents a tangible opportunity to secure a more reliable pharmaceutical intermediate supplier capable of delivering consistent quality without the volatility associated with scarce precious metal catalysts. The following analysis details how this novel chemistry addresses historical bottlenecks in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for Compound 1 have been plagued by significant operational and economic inefficiencies that hinder large-scale production. For instance, early patents such as US5374637 relied heavily on butyllithium chemistry, necessitating cryogenic temperatures as low as -78°C to manage reactivity. This not only imposes immense energy costs on the manufacturing facility but also introduces severe safety risks associated with handling pyrophoric reagents on a tonnage scale. Furthermore, these legacy processes often yielded undesirable isomers that required tedious column chromatography for separation, resulting in abysmal overall yields of approximately 18%. Other literature methods, such as those published in Synlett (1993), utilized rhodium catalysts and silica reagents, which are economically unsustainable for commodity chemical production due to the high cost of the noble metal and the difficulty in recovering it from the reaction mixture.
Additionally, some prior art routes employed osmium tetroxide for oxidative cleavage, a reagent known for its extreme toxicity and stringent regulatory handling requirements. The use of such hazardous materials creates substantial liabilities for environmental health and safety (EHS) departments, complicating waste disposal and increasing the cost of compliance. The reliance on column chromatography in multiple steps further exacerbates the problem, as it limits batch sizes and increases solvent consumption dramatically. These factors combined make conventional methods unsuitable for the suitability for industrialized production required to meet the global demand for Prucalopride. Consequently, there has been a persistent need for a synthetic strategy that balances chemical elegance with commercial pragmatism.
The Novel Approach
The methodology presented in CN102942542A offers a transformative solution by reimagining the construction of the dihydrobenzofuran ring system. Instead of relying on dangerous organolithium reagents or toxic osmium oxidants, this novel approach initiates the synthesis with an allyl-substituted precursor (Compound 7). The cornerstone of this innovation is the use of a ruthenium trichloride and periodate composite catalyst system to effect the oxidative cleavage of the terminal alkene. This catalytic system is not only significantly cheaper than rhodium alternatives but also operates under much milder conditions, typically between 0°C and 80°C. This shift in reaction conditions drastically reduces the energy footprint of the process and eliminates the need for specialized cryogenic equipment, thereby enhancing the feasibility of cost reduction in API manufacturing.
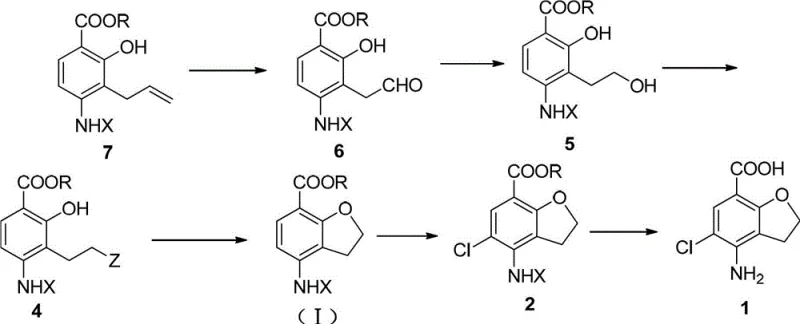
Following the oxidation step, the resulting aldehyde is reduced to an alcohol and subsequently converted into a leaving group, setting the stage for an intramolecular cyclization. This sequence allows for the formation of the five-membered ether ring with high regioselectivity and minimal byproduct formation. Crucially, the workup procedures described involve standard extractions and crystallizations rather than chromatographic separations. This simplification of downstream processing is a major advantage for supply chain reliability, as it enables larger batch sizes and faster throughput. By avoiding the use of hypertoxic reagents and expensive noble metals, this route aligns perfectly with modern green chemistry principles while simultaneously driving down the cost of goods sold (COGS) for the final intermediate.
Mechanistic Insights into Ruthenium-Catalyzed Oxidative Cleavage
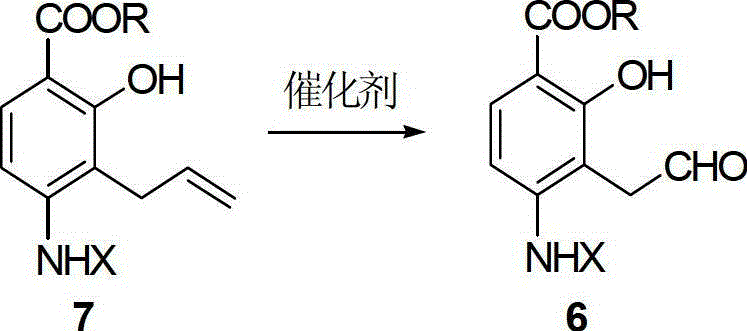
The heart of this synthetic innovation is the ruthenium-catalyzed oxidation of the allyl side chain to an aldehyde functionality. In this mechanism, ruthenium trichloride (or its hydrate) acts in concert with a periodate oxidant, such as sodium periodate or potassium periodate, to generate a high-valent ruthenium-oxo species in situ. This active catalytic species is responsible for the cleavage of the carbon-carbon double bond in the allyl group of Compound 7. The reaction proceeds through a cyclic ruthenate ester intermediate, which subsequently decomposes to release the desired aldehyde (Compound 6) and regenerate the catalyst. The mole dosage of the ruthenium catalyst is kept remarkably low, typically between 1% and 8%, with preferred embodiments utilizing only 2% to 4%. This high turnover number demonstrates the efficiency of the catalytic cycle and minimizes the residual metal content in the final product, a critical parameter for pharmaceutical quality control.
Following the oxidation, the process employs a straightforward reduction of the aldehyde to a primary alcohol using sodium borohydride. This step is highly chemoselective and proceeds rapidly at ambient temperatures (20°C to 30°C). The subsequent conversion of the alcohol to a leaving group (Z), such as a chloride or tosylate, activates the side chain for nucleophilic attack by the phenolic oxygen. This intramolecular substitution is the key ring-closing step that forms the 2,3-dihydrobenzofuran scaffold. The use of organic bases or mineral alkalis facilitates this cyclization under reflux conditions, ensuring complete conversion without the need for harsh acidic conditions that might degrade the sensitive amino or ester functionalities. This mechanistic pathway effectively bypasses the need for protecting group manipulations that are common in other routes, thereby shortening the overall synthetic timeline and improving the impurity profile of the intermediate.
How to Synthesize 4-Amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic Acid Efficiently
The synthesis of this critical pharmaceutical intermediate involves a carefully orchestrated sequence of transformations designed to maximize yield and purity while minimizing operational complexity. The process begins with the oxidative cleavage of the allyl precursor, followed by reduction and activation of the side chain. Once the linear precursor is activated, a base-mediated cyclization closes the furan ring. The final stages involve regioselective chlorination of the aromatic ring and hydrolysis of the ester to reveal the free carboxylic acid. Each step has been optimized to avoid chromatographic purification, relying instead on crystallization and extraction to achieve the necessary purity standards. For a detailed breakdown of the specific reaction conditions, stoichiometry, and workup procedures required to execute this synthesis in a GMP environment, please refer to the standardized protocol below.
- Oxidize the allyl precursor (Compound 7) using a ruthenium trichloride/periodate composite catalyst to form the aldehyde intermediate (Compound 6).
- Reduce the aldehyde group to a hydroxyl group using sodium borohydride to obtain Compound 5, followed by substitution to introduce a leaving group Z.
- Perform base-catalyzed cyclization to form the dihydrobenzofuran ring (Compound I), followed by chlorination and hydrolysis to yield the final acid (Compound 1).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN102942542A offers distinct strategic advantages over legacy manufacturing methods. The primary benefit is the substantial cost savings achieved through the elimination of expensive noble metal catalysts like rhodium and the avoidance of cryogenic operations. By utilizing a ruthenium-based system that operates at near-ambient temperatures, manufacturers can significantly reduce energy consumption and capital expenditure on specialized cooling infrastructure. Furthermore, the removal of column chromatography from the process flow allows for a drastic increase in batch throughput, directly addressing the need for reducing lead time for high-purity pharmaceutical intermediates. This efficiency gain translates into a more resilient supply chain capable of responding quickly to market fluctuations.
- Cost Reduction in Manufacturing: The replacement of rhodium and osmium reagents with a ruthenium/periodate system results in a direct decrease in raw material costs. Additionally, the simplified workup procedures eliminate the massive solvent volumes associated with silica gel chromatography, leading to lower waste disposal fees and reduced solvent recovery costs. The overall process design prioritizes atom economy and operational simplicity, ensuring that the cost of goods remains competitive even at large production scales.
- Enhanced Supply Chain Reliability: The starting materials for this route, specifically the allyl-substituted benzene derivatives, are readily accessible and do not rely on single-source suppliers for exotic reagents. The robustness of the reaction conditions means that production is less susceptible to disruptions caused by equipment failure or utility fluctuations. This stability ensures a continuous supply of the key intermediate, mitigating the risk of stockouts that could delay the production of the final API.
- Scalability and Environmental Compliance: The absence of highly toxic reagents like osmium tetroxide simplifies environmental compliance and reduces the burden on EHS teams. The process generates less hazardous waste, making it easier to permit and operate in diverse geographic locations. The scalability is further enhanced by the use of standard unit operations such as filtration and crystallization, which are easily transferred from pilot plants to multi-ton commercial reactors without significant re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Prucalopride intermediates using this patented methodology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing networks. The answers highlight the balance between chemical performance and operational practicality that defines this innovation.
Q: How does the ruthenium-catalyzed route improve upon traditional rhodium or osmium methods?
A: The patented method replaces expensive rhodium reagents and highly toxic osmium tetroxide with a cost-effective ruthenium trichloride and periodate system. This significantly reduces raw material costs and eliminates severe environmental and safety hazards associated with heavy metal toxicity, simplifying waste treatment and regulatory compliance.
Q: What are the scalability advantages of this synthesis route?
A: The process operates under mild conditions (0-80°C) compared to cryogenic methods (-78°C) required by butyllithium routes. Furthermore, it avoids complex column chromatography purification steps, relying instead on standard extraction and crystallization, which facilitates easier scale-up from pilot to commercial production volumes.
Q: Is the starting material for this route readily available?
A: Yes, the starting material, substituted p-amino m-allyl o-hydroxybenzene methyl ester (Compound 7), can be synthesized using established literature methods or purchased commercially. Its accessibility ensures a stable supply chain foundation for continuous manufacturing of the downstream intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex APIs like Prucalopride depends on a robust and scalable supply of high-quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical markets. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid meets the exacting standards required for downstream coupling reactions. Our facility is equipped to handle the specific catalytic and cyclization chemistries described in CN102942542A with precision and safety.
We invite potential partners to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and quality requirements. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain and enhance your competitive position in the marketplace.
