Advanced Purification Strategy for High-Purity Prucalopride Manufacturing
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
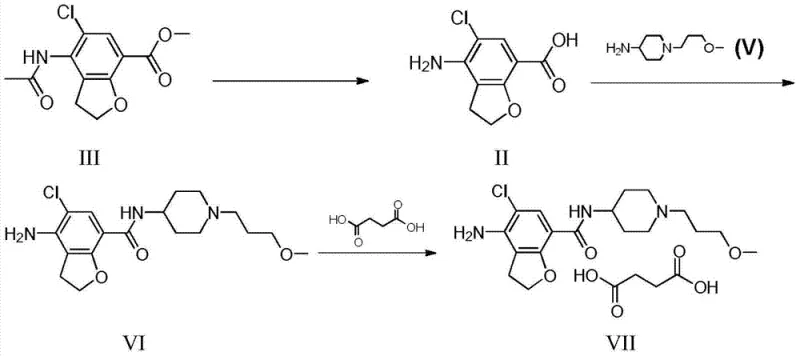
In the traditional synthesis pathways for Prucalopride, a significant bottleneck arises from the quality of the commercially available starting material, specifically acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid methyl ester. This raw material invariably contains trace amounts of a bromo analogue impurity due to the halogenation processes used in its own manufacture. Because the chemical properties of this bromo impurity are strikingly similar to the desired chloro substrate, conventional purification techniques such as standard column chromatography or simple recrystallization of the ester precursor often fail to achieve sufficient separation. Consequently, this persistent bromo contaminant survives the initial hydrolysis steps and propagates through the entire synthetic sequence. This leads to the formation of structurally similar bromo-byproducts in the final active pharmaceutical ingredient, which are notoriously difficult to remove in the late stages of synthesis, thereby compromising the overall purity profile and potentially affecting the safety and efficacy of the final gastrointestinal therapeutic agent.
The Novel Approach
The innovative methodology disclosed in patent CN103664912A introduces a strategic intervention point that fundamentally alters the purification landscape for this critical intermediate. Instead of attempting to purify the ester precursor directly, the process first subjects the raw material to hydrolysis to generate the free carboxylic acid form, known as 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid. It is at this specific stage, where the molecule exists as a free acid, that a targeted solvent recrystallization step is employed. By leveraging the distinct differential solubility profiles between the desired chloro-acid and the unwanted bromo-acid impurity in specific polar aprotic solvent systems, the process achieves a level of purification that was previously unattainable. This approach effectively sequesters the bromo impurity in the mother liquor, allowing the highly purified chloro-intermediate to crystallize out, thus breaking the chain of impurity propagation and ensuring a much cleaner substrate for the subsequent amide coupling reactions.
Mechanistic Insights into Recrystallization-Based Impurity Control
The core mechanistic advantage of this synthesis lies in the thermodynamic manipulation of solubility during the crystallization of the key intermediate. When the crude 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid is dissolved in solvents such as DMF or DMA and subsequently treated with water as an anti-solvent, the system reaches a supersaturated state favorable for the nucleation of the chloro-species. The bromo-analogue, possessing slightly different lattice energy and solvation characteristics due to the larger atomic radius and different electronegativity of the bromine atom compared to chlorine, remains preferentially solvated in the liquid phase. This phenomenon is critical because it prevents the co-crystallization of the impurity. Furthermore, the use of activated carbon during the hot filtration step prior to cooling aids in the adsorption of colored organic byproducts and polymeric residues, further refining the quality of the crystal lattice as it forms. The controlled cooling rate, maintained between 5 to 10 degrees Celsius per hour, ensures the growth of well-defined crystals with minimal occlusion of the mother liquor, which is the primary vehicle for the rejected bromo impurities.
Following the purification of the acid intermediate, the subsequent condensation reaction with 1-(3-methoxy-propyl)-4-piperidylhydrazine proceeds with significantly higher fidelity. The use of carbonyldiimidazole (CDI) as the coupling agent activates the carboxylic acid to form a reactive acyl imidazole intermediate, which is then attacked by the hydrazine nucleophile. Because the starting acid is now virtually free of the bromo contaminant, the resulting amide product, Prucalopride, is formed with a drastically reduced burden of related substances. This high fidelity in the coupling step minimizes the need for aggressive downstream purification, such as preparative HPLC, which is often cost-prohibitive at a commercial scale. The final salification with succinic acid in ethanol yields the target succinate salt with exceptional purity, demonstrating how early-stage intermediate control dictates the success of the entire manufacturing campaign.
How to Synthesize Prucalopride Efficiently
The synthesis of Prucalopride described in this technical disclosure offers a robust roadmap for manufacturers seeking to optimize their production of this gastrointestinal agent. The process is defined by its simplicity and reliance on standard unit operations such as hydrolysis, filtration, and crystallization, which are easily adaptable to existing reactor setups. The critical innovation is the timing of the purification; by shifting the recrystallization to the free acid stage rather than the ester stage, manufacturers can exploit solubility differences that are otherwise inaccessible. This strategy not only improves the chemical purity but also enhances the physical properties of the intermediate, such as filterability and drying characteristics. For detailed operational parameters regarding temperature controls, solvent ratios, and reaction times, please refer to the standardized synthesis guide provided below, which encapsulates the optimal conditions identified in the patent literature.
- Hydrolyze acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid methyl ester using alkali aqueous solution to obtain crude 4-amino-5-chloro-2,3-dihydrobenzofuran-7-carboxylic acid.
- Perform solvent recrystallization on the crude acid using a DMF/Water or DMA/Water system to selectively remove 4-amino-5-bromo impurities based on solubility differences.
- Condense the purified acid with 1-(3-methoxy-propyl)-4-piperidylhydrazine using CDI coupling, followed by salification with succinic acid to yield Prucalopride Succinate.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the implementation of this recrystallization-focused synthesis route offers substantial strategic benefits that extend beyond mere chemical purity. Traditional methods that rely on late-stage purification or chromatography often suffer from variable yields and high solvent consumption, leading to unpredictable production schedules and inflated costs. By contrast, this novel approach stabilizes the supply chain by ensuring that the critical intermediate is consistently produced to a high specification, reducing the risk of batch failures and the need for reprocessing. The elimination of complex chromatographic steps simplifies the manufacturing workflow, allowing for faster turnover times and more efficient utilization of production assets. This reliability is paramount for maintaining continuous supply to downstream formulation partners and meeting the rigorous quality standards demanded by global regulatory agencies.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of expensive and low-throughput purification techniques with a highly efficient crystallization step. Chromatographic separation of closely related halogenated impurities typically requires large volumes of specialized solvents and silica gel, generating significant waste and operational expense. By removing the bromo impurity through crystallization, the process eliminates the need for these costly separation technologies entirely. Furthermore, the higher purity of the intermediate leads to improved yields in the subsequent coupling reaction, as fewer side reactions occur, maximizing the conversion of raw materials into the final valuable product and reducing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this purification method. Since the process effectively handles variations in the quality of the commercially available starting material by scrubbing out the bromo impurity early, manufacturers are less vulnerable to fluctuations in raw material specifications from different suppliers. This flexibility allows procurement teams to source starting materials from a broader range of vendors without compromising the quality of the final API. Additionally, the simplified process flow reduces the number of potential failure points in the manufacturing line, ensuring more predictable lead times and consistent delivery schedules for customers relying on this critical gastrointestinal therapeutic.
- Scalability and Environmental Compliance: The transition from chromatography to crystallization represents a major advancement in process scalability and environmental sustainability. Crystallization is a unit operation that scales linearly and predictably from laboratory to multi-ton production, whereas chromatography often faces significant engineering challenges when scaled up. Moreover, the reduction in solvent usage and the avoidance of silica waste align with green chemistry principles, lowering the environmental footprint of the manufacturing process. This compliance with environmental standards not only reduces waste disposal costs but also future-proofs the supply chain against increasingly stringent regulatory requirements regarding industrial emissions and solvent residues in pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and purification of Prucalopride. These insights are derived directly from the experimental data and beneficial effects reported in the underlying patent documentation. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this improved manufacturing route. The answers provided highlight the specific advantages of the recrystallization technique and its impact on the final quality attributes of the drug substance.
Q: Why is recrystallization critical for Prucalopride intermediates?
A: Commercial starting materials often contain bromo analogues that are chemically similar to the chloro substrate. Without a specific recrystallization step after hydrolysis, these bromo impurities carry through the synthesis, creating difficult-to-remove byproducts in the final API.
Q: What is the optimal solvent system for purifying the key intermediate?
A: The patent data indicates that a mixed solvent system of DMF/Water or DMA/Water provides the optimal balance of yield and purity, effectively keeping the bromo impurity in the mother liquor while crystallizing the desired chloro-acid intermediate.
Q: How does this process impact commercial scalability?
A: By replacing complex chromatographic purification with a robust crystallization step, the process significantly reduces processing time and solvent consumption, making it highly suitable for large-scale commercial production of gastrointestinal agents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prucalopride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the synthesis of complex gastrointestinal agents like Prucalopride requires not just chemical expertise, but a deep understanding of process optimization and impurity control. Our technical team has extensively analyzed advanced purification strategies, such as the recrystallization methods detailed in recent patent literature, to ensure our manufacturing processes deliver the highest possible quality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of laboratory-scale innovations are fully realized in large-scale manufacturing. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Prucalopride or its intermediates meets the exacting standards required for global pharmaceutical markets.
We invite procurement directors and supply chain managers to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized synthesis routes, we can offer competitive pricing structures without compromising on quality or compliance. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us partner with you to secure a stable, high-quality supply of Prucalopride that supports your long-term commercial goals.
