Revolutionizing Lactam Production: A Chemo-Enzymatic Approach for Commercial Scale-up
Introduction to Advanced Lactam Manufacturing Technologies
The pharmaceutical and agrochemical industries constantly seek robust methodologies for synthesizing nitrogen-containing heterocycles, particularly lactams, which serve as critical scaffolds in active pharmaceutical ingredients. Patent CN1554646A introduces a groundbreaking chemo-enzymatic process that fundamentally alters the production landscape for five-membered and six-membered ring lactams derived from aliphatic alpha,omega-dinitriles. This technology integrates the high specificity of biocatalysis with the efficiency of chemical hydrogenation, offering a streamlined pathway that bypasses the harsh conditions traditionally associated with lactam cyclization. By leveraging specific microbial strains such as Acidovorax facilis and Comamonas testosteroni, the process achieves exceptional regioselectivity during the initial hydrolysis phase, setting the stage for a highly efficient downstream transformation.
Unlike conventional synthetic routes that often rely on stoichiometric reagents and generate substantial inorganic waste, this patented method operates primarily in an aqueous environment, aligning with modern green chemistry principles. The core innovation lies in the seamless transition from the enzymatic generation of an omega-nitrilecarboxylic acid ammonium salt directly to the final lactam product via hydrogenation. This eliminates the need for intermediate isolation, acidification, or solvent exchanges that typically inflate manufacturing costs and extend lead times. For R&D directors and process chemists, this represents a significant opportunity to enhance purity profiles while simplifying the overall unit operations required for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chemical methods for converting nitriles to carboxylic acids and subsequently to lactams are fraught with significant operational challenges and environmental drawbacks. Historically, these transformations necessitate the use of strong acidic or alkaline reaction conditions coupled with very high temperatures, often exceeding 250 degrees Celsius, to drive the cyclization equilibrium towards the lactam product. Such extreme thermal requirements not only demand specialized high-pressure equipment but also increase the risk of thermal degradation of sensitive functional groups, leading to complex impurity profiles that are difficult to purge. Furthermore, chemical hydrolysis frequently lacks regioselectivity when applied to asymmetric dinitriles, resulting in mixtures of isomeric products that require costly and yield-reducing separation processes.
In addition to selectivity issues, conventional routes typically involve the isolation of intermediate omega-amino-carboxylic acids as free acids prior to cyclization. This necessitates multiple pH adjustments, extraction steps, and drying operations, each introducing potential points of failure and material loss. The reliance on organic solvents for these isolation steps further complicates waste management and increases the carbon footprint of the manufacturing process. For supply chain managers, these inefficiencies translate into longer production cycles and higher vulnerability to raw material price fluctuations, as the overall process mass intensity is significantly elevated compared to more direct catalytic approaches.
The Novel Approach
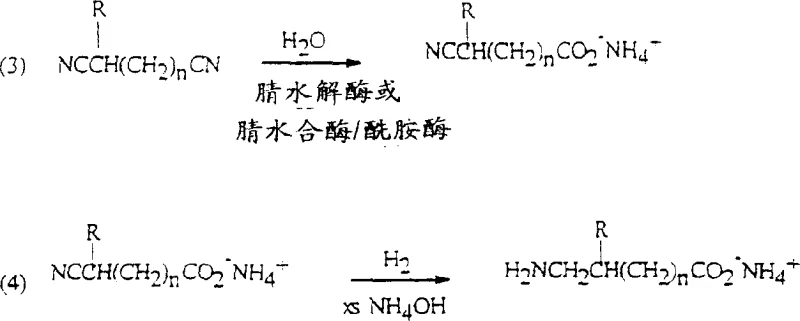
The novel approach detailed in the patent data revolutionizes this workflow by combining enzymatic hydrolysis with direct reductive cyclization in a single aqueous phase. By utilizing enzymes with nitrilase or nitrile hydratase activity, the process achieves greater than 98 percent regioselectivity in the hydrolysis of the dinitrile substrate, ensuring that only the desired omega-nitrilecarboxylic acid ammonium salt is produced. This biological precision eliminates the formation of unwanted dicarboxylic acid byproducts that plague non-enzymatic methods. Following hydrolysis, the reaction mixture is subjected directly to hydrogenation in the presence of a hydrogenation catalyst and excess ammonia, facilitating the conversion of the nitrile group to an amine and its immediate cyclization to the lactam without isolating the intermediate amino acid.
This telescoped strategy drastically reduces the number of unit operations, effectively merging hydrolysis, reduction, and cyclization into a cohesive flow. The ability to perform the cyclization directly from the ammonium salt in aqueous solution at temperatures significantly lower than the traditional 250 degrees Celsius threshold is a major technical breakthrough. It allows for the use of standard stainless steel reactors rather than exotic alloys required for high-temperature acidic conditions. For procurement teams, this translates to a more resilient supply chain with reduced dependency on aggressive reagents and specialized infrastructure, ultimately driving down the cost of goods sold for high-value lactam intermediates.
Mechanistic Insights into Chemo-Enzymatic Cyclization
The mechanistic elegance of this process begins with the enzymatic hydrolysis step, where specific microbial catalysts distinguish between the two nitrile groups of the alpha,omega-dinitrile. Enzymes such as those derived from Acidovorax facilis 72W exhibit a profound preference for hydrolyzing the omega-nitrile group while leaving the alpha-nitrile group intact, a feat difficult to achieve with chemical catalysts. This regioselectivity is critical because it dictates the ring size of the final lactam product. The enzyme converts the nitrile directly to the carboxylic acid ammonium salt without forming a stable amide intermediate, or via a transient amide that is rapidly hydrolyzed by amidase activity. This ensures that the reaction mixture entering the hydrogenation stage contains predominantly the linear omega-nitrilecarboxylic acid ammonium salt, minimizing the load on the subsequent chemical catalyst.
Following the biocatalytic step, the mechanism shifts to heterogeneous catalysis. The addition of excess ammonia or methylamine during hydrogenation serves a dual purpose: it suppresses the reductive alkylation of the newly formed amine with the carbonyl of another molecule, which would otherwise lead to dimerization and polymer formation, and it maintains the species in the ammonium salt form. Surprisingly, the patent reveals that under these hydrogenation conditions, typically ranging from 70 degrees Celsius to 180 degrees Celsius, the omega-amino-ammonium carboxylate salt undergoes direct cyclization to the lactam. This contradicts prior art which suggested that cyclization required the free acid form and high temperatures. The presence of the hydrogenation catalyst, likely Raney nickel or palladium on carbon, appears to facilitate this intramolecular condensation even in the basic aqueous environment, allowing for high yields of five and six-membered rings without the need for acidification.
How to Synthesize Lactams Efficiently
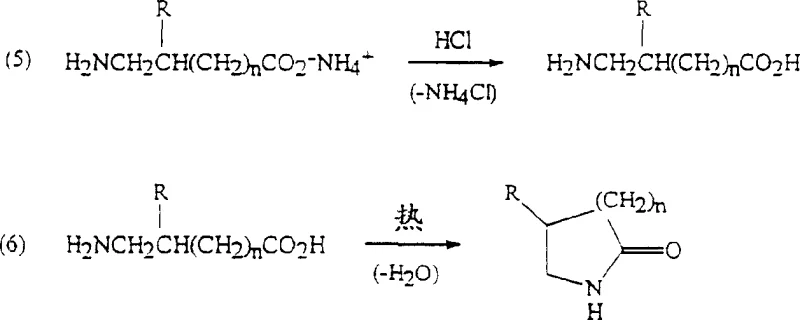
The synthesis of lactams via this chemo-enzymatic route is designed for operational simplicity and high throughput, making it ideal for both laboratory optimization and industrial manufacturing. The process begins with the preparation of the biocatalyst, either as whole cells or immobilized enzymes, which are contacted with the dinitrile substrate in a buffered aqueous solution. Careful control of temperature during the enzymatic step, often involving a heat treatment to deactivate undesirable side activities, ensures maximum regioselectivity. Once the hydrolysis is complete, the enzyme is removed by filtration or centrifugation, and the clarified aqueous solution is transferred to a hydrogenation vessel. Here, the addition of a hydrogenation catalyst and stoichiometric excess of ammonia sets the stage for the reductive cyclization.
- Contact aliphatic alpha,omega-dinitrile with an enzyme catalyst (nitrilase or nitrile hydratase/amidase combination) in aqueous solution to form omega-nitrilecarboxylic acid ammonium salt.
- Subject the aqueous product mixture directly to hydrogenation with a hydrogenation catalyst in the presence of excess ammonia or methylamine.
- Recover the corresponding five-membered or six-membered ring lactam directly from the aqueous product mixture without isolating intermediate acids or amines.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the pain points of cost and reliability in fine chemical manufacturing. The elimination of intermediate isolation steps is perhaps the most significant driver of cost reduction. By avoiding the need to acidify, extract, dry, and redissolve the omega-amino-carboxylic acid intermediate, manufacturers can save substantially on solvent consumption, utility costs for evaporation, and labor hours. Furthermore, the aqueous nature of the reaction medium reduces the fire hazard and environmental burden associated with large volumes of flammable organic solvents, simplifying regulatory compliance and waste disposal logistics.
- Cost Reduction in Manufacturing: The integration of enzymatic selectivity with chemical hydrogenation removes the need for expensive protecting groups or complex separation technologies required to isolate specific isomers in traditional synthesis. The ability to run the cyclization at lower temperatures also reduces energy consumption significantly compared to thermal cyclization methods requiring over 250 degrees Celsius. Additionally, the high conversion rates and minimal byproduct formation mean that raw material utilization is maximized, reducing the effective cost per kilogram of the final lactam product.
- Enhanced Supply Chain Reliability: The robustness of the microbial catalysts described, such as Comamonas testosteroni, ensures consistent batch-to-batch performance, which is critical for maintaining supply continuity for downstream API production. The process tolerates a wide range of substrate concentrations and does not require ultra-high purity starting materials, allowing for flexibility in sourcing aliphatic dinitriles. This resilience minimizes the risk of production delays caused by raw material variability, providing procurement managers with greater confidence in long-term supply agreements.
- Scalability and Environmental Compliance: Scaling this process is straightforward as it relies on standard fermentation and hydrogenation equipment already present in most multipurpose chemical plants. The aqueous waste streams generated are generally less toxic and easier to treat than those containing heavy metals or strong mineral acids typical of purely chemical routes. This alignment with green chemistry metrics not only improves the sustainability profile of the product but also future-proofs the manufacturing site against increasingly stringent environmental regulations regarding solvent emissions and hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this chemo-enzymatic lactam synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these nuances is essential for process engineers evaluating the feasibility of adopting this route for specific target molecules.
Q: What is the primary advantage of the enzymatic hydrolysis step in this lactam synthesis?
A: The enzymatic step provides greater than 98% regioselectivity when hydrolyzing asymmetrically substituted dinitriles, ensuring only one specific lactam product is formed, unlike chemical methods which produce mixtures.
Q: Does this process require isolation of the intermediate omega-amino-carboxylic acid?
A: No, a key innovation of this patent is the direct conversion of the omega-nitrilecarboxylic acid ammonium salt to the lactam via hydrogenation without isolating the intermediate acid or amine salts.
Q: What are the typical reaction conditions for the hydrogenation step?
A: The hydrogenation is conducted in aqueous solution at temperatures between 70°C and 180°C under hydrogen pressure, typically using Raney nickel or palladium catalysts in the presence of excess ammonia.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Lactam Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of integrating biocatalysis into traditional chemical synthesis to produce high-value intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT annual commercial production, ensuring that innovative laboratory processes like the one described in CN1554646A can be successfully translated into robust manufacturing operations. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of lactam intermediate meets the exacting standards required by global pharmaceutical and agrochemical clients.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Our experts are ready to provide a Customized Cost-Saving Analysis to demonstrate how this chemo-enzymatic route can optimize your budget without compromising quality. Please contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your target molecules, and let us help you secure a competitive advantage in the market.
