Advanced Clean Production Technology for High-Purity Testosterone Propionate Manufacturing
Advanced Clean Production Technology for High-Purity Testosterone Propionate Manufacturing
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally sustainable pathways for the synthesis of critical hormonal active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN110563789B, which outlines a novel clean production preparation method for testosterone propionate. This technology represents a paradigm shift from traditional acylation techniques, addressing long-standing issues regarding impurity profiles and waste management. By utilizing testosterone as the primary raw material and employing a specific esterification protocol followed by rigorous refining, the process guarantees a final product purity of ≥99.5%. Furthermore, the innovation extends beyond mere synthesis; it incorporates a comprehensive waste treatment strategy that converts hazardous acid water into a commercially viable sodium propionate byproduct. For global procurement teams and R&D directors, this patent offers a compelling blueprint for cost reduction in pharmaceutical intermediates manufacturing while adhering to increasingly strict environmental regulations.
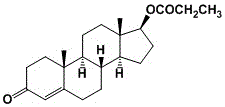
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of testosterone propionate has relied heavily on acylation reactions using propionic anhydride. While chemically straightforward, this conventional approach suffers from significant drawbacks that impact both product quality and operational expenditure. The primary technical challenge lies in the lack of selectivity; during the reaction with propionic anhydride, both the 3-position ketone group and the 17-position hydroxyl group of the testosterone molecule can simultaneously react. This non-specific reactivity leads to the formation of diester impurities, which are notoriously difficult to separate and can compromise the safety and efficacy of the final drug product. Additionally, the hydrolysis of excess propionic anhydride generates substantial volumes of highly acidic wastewater. Treating this waste acid water is not only environmentally burdensome but also incurs high disposal costs, creating a financial drag on the overall production economics. These factors combined make the traditional anhydride route less attractive for modern, green chemistry-focused manufacturing facilities.
The Novel Approach
In stark contrast to the anhydride-based legacy methods, the technology disclosed in patent CN110563789B introduces a refined esterification strategy that utilizes a combination of propionic acid and propionyl chloride. This dual-reagent system allows for precise control over the reaction kinetics, effectively suppressing the formation of unwanted diester byproducts and ensuring high regioselectivity at the 17-hydroxyl position. The process is designed with a closed-loop mindset regarding waste; rather than viewing the acidic effluent as a liability, the method captures the waste acid water generated during the workup phase. Through a subsequent neutralization step using sodium carbonate, this waste stream is transformed into sodium propionate, a useful chemical commodity. This approach not only simplifies the purification workflow but also turns a cost center (waste treatment) into a potential revenue stream. For a reliable testosterone propionate supplier, adopting this methodology signifies a commitment to both superior product quality and sustainable operational practices.
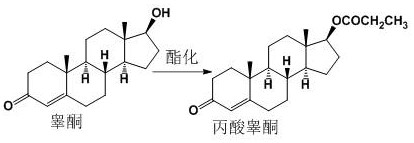
Mechanistic Insights into Propionyl Chloride Catalyzed Esterification
The core of this synthetic advancement lies in the mechanistic interplay between propionic acid and propionyl chloride in the presence of the steroid substrate. The reaction initiates with the activation of the 17-hydroxyl group of testosterone by the highly reactive propionyl chloride, forming an intermediate ester linkage rapidly. The presence of propionic acid serves multiple critical functions: it acts as a solvent to ensure homogeneity of the steroid substrate, buffers the reaction environment to prevent excessive acidity that could degrade the sensitive steroid backbone, and participates in the equilibrium to drive the reaction to completion. The patent specifies a reflux condition, typically maintained for 2 to 3 hours, which provides the necessary thermal energy to overcome activation barriers without inducing thermal decomposition. Monitoring via Thin Layer Chromatography (TLC) is employed to determine the exact endpoint, ensuring that the conversion is complete before proceeding to workup, thereby maximizing the yield and minimizing the presence of unreacted starting material in the crude mixture.
Impurity control is further enhanced during the downstream processing stages, specifically the recrystallization and decolorization steps. The crude product, obtained after water elutriation and filtration, contains trace organic impurities and colored bodies that must be removed to meet pharmacopeial standards. The process utilizes methanol as a recrystallization solvent in a specific weight ratio (10-12:1 relative to the crude product), combined with activated carbon treatment under reflux. This step is crucial for adsorbing polar impurities and chromophores. The subsequent concentration of the filtrate to a specific residual methanol ratio, followed by controlled cooling crystallization at 0-5°C, ensures the formation of large, pure crystals while excluding soluble impurities from the lattice. This rigorous control over thermodynamic parameters during crystallization is what enables the consistent achievement of ≥99.5% purity, a critical metric for high-purity testosterone propionate intended for clinical applications.
How to Synthesize Testosterone Propionate Efficiently
The implementation of this clean production method requires adherence to specific operational parameters to ensure reproducibility and safety. The synthesis is divided into three distinct phases: the primary esterification reaction, the purification of the API, and the valorization of the waste stream. Each phase relies on precise stoichiometric ratios and temperature controls as defined in the patent examples. For R&D teams looking to replicate or scale this process, it is essential to note the specific weight ratios of reagents, such as the 6-8:1 ratio of propionic acid to testosterone, which is vital for maintaining reaction homogeneity. The following guide summarizes the standardized operational flow derived from the patent data, serving as a foundational reference for process engineers aiming to establish commercial scale-up of complex steroid intermediates.
- Step A: Esterification. React testosterone with propionic acid and propionyl chloride under reflux. Monitor via TLC, then precipitate in water to isolate crude product and collect waste acid water.
- Step B: Purification. Recrystallize the crude product using methanol and activated carbon for decolorization. Filter, concentrate, cool to crystallize, and dry to obtain high-purity testosterone propionate.
- Step C: Waste Treatment. Neutralize the collected waste acid water with sodium carbonate, decolorize with activated carbon, filter, and concentrate to crystallize and recover sodium propionate byproduct.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic advantages that extend beyond simple chemical yield. The most significant value proposition is the drastic simplification of waste management protocols. In traditional steroid manufacturing, the disposal of acidic wastewater represents a significant operational expense and regulatory risk. By integrating a neutralization and recovery step that produces sodium propionate, this process effectively eliminates the environmental liability associated with acid waste. This transformation of waste into a byproduct not only reduces the direct costs of wastewater treatment but also mitigates the risk of production stoppages due to environmental compliance issues. Consequently, the overall cost of goods sold (COGS) is optimized, providing a competitive edge in pricing negotiations for bulk contracts without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the elimination of expensive purification steps often required to remove diester impurities found in anhydride-based routes. By preventing the formation of these impurities at the source through selective esterification, the need for complex chromatographic separations or multiple recrystallizations is minimized. Furthermore, the recovery of sodium propionate adds a secondary revenue stream that offsets the cost of raw materials. The use of commodity reagents like propionic acid and propionyl chloride, which are widely available and cost-effective, ensures that the raw material supply remains stable and affordable. This structural efficiency translates into substantial cost savings over the lifecycle of the product, making it an ideal candidate for long-term supply agreements.
- Enhanced Supply Chain Reliability: Supply continuity is paramount for pharmaceutical manufacturers, and this process enhances reliability by relying on robust, non-proprietary raw materials. Unlike methods that depend on specialized catalysts or exotic reagents that may face supply bottlenecks, the reagents used here are standard industrial chemicals with mature global supply chains. The simplicity of the unit operations—reflux, filtration, and crystallization—means that the process can be easily transferred between different manufacturing sites or scaled up rapidly to meet surges in demand. This flexibility ensures that a reliable testosterone propionate supplier can maintain consistent delivery schedules even in volatile market conditions, reducing the lead time risks often associated with complex hormone synthesis.
- Scalability and Environmental Compliance: From a facility perspective, the process is designed for seamless scalability from pilot plant to multi-ton commercial production. The reaction conditions (reflux temperatures) and workup procedures (water elutriation) are standard in chemical engineering, requiring no specialized high-pressure or cryogenic equipment. Moreover, the 'clean production' aspect aligns perfectly with modern ESG (Environmental, Social, and Governance) goals. By neutralizing waste acid and recovering byproducts, the facility reduces its environmental footprint, simplifying the permitting process and fostering better relationships with local regulatory bodies. This proactive approach to environmental stewardship future-proofs the supply chain against tightening environmental regulations, ensuring long-term operational viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this clean production technology. These insights are derived directly from the experimental data and beneficial effects described in patent CN110563789B. Understanding these details is crucial for technical evaluators assessing the feasibility of integrating this route into existing manufacturing portfolios. The answers highlight the balance between high-purity output and process efficiency that defines this innovation.
Q: What is the achieved purity of testosterone propionate using this method?
A: According to patent CN110563789B, this clean production method consistently achieves a product purity of greater than or equal to 99.5%, meeting stringent pharmaceutical standards through optimized esterification and recrystallization.
Q: How does this process handle environmental waste compared to traditional methods?
A: Unlike traditional methods that generate difficult-to-treat acid water, this process neutralizes waste acid with sodium carbonate to produce sodium propionate, a valuable byproduct, thereby eliminating pollution liabilities and reducing disposal costs.
Q: What are the key advantages regarding yield and impurity control?
A: The method avoids the formation of diester impurities common with propionic anhydride routes. It reports a yield of over 110% (relative to theoretical mass increase) and effectively controls impurities through specific solvent ratios and temperature controls during crystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Testosterone Propionate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and rigorous process control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN110563789B are fully realized in industrial practice. Our facilities are equipped with state-of-the-art reactors and purification systems capable of handling the specific reflux and crystallization requirements of steroid chemistry. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of testosterone propionate meets or exceeds the ≥99.5% purity benchmark, providing our partners with absolute confidence in the quality of their supply chain.
We invite global pharmaceutical companies and chemical distributors to collaborate with us to optimize their sourcing strategies. By leveraging our technical capabilities, you can achieve significant efficiencies in your hormone supply chain. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our team is ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing processes can enhance your product portfolio while reducing overall operational costs. Let us be your partner in delivering high-quality hormonal intermediates to the global market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
