Scalable Synthesis of 3-Arylquinolin-2(1H)one Derivatives via Novel Palladium Catalysis for Global Pharmaceutical Supply Chains
Scalable Synthesis of 3-Arylquinolin-2(1H)one Derivatives via Novel Palladium Catalysis for Global Pharmaceutical Supply Chains
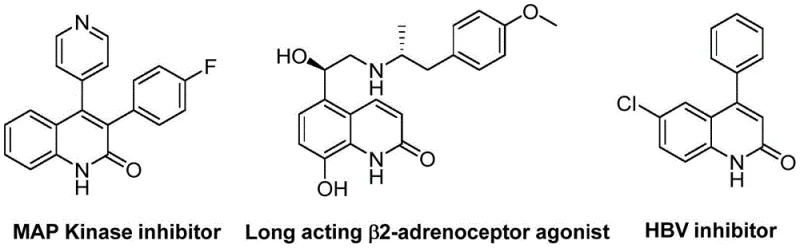
The pharmaceutical industry constantly seeks robust, scalable, and cost-effective methodologies for constructing privileged heterocyclic scaffolds, particularly quinolin-2(1H)one derivatives, which serve as critical cores in numerous bioactive molecules ranging from antibiotics to antitumor agents. A significant technological breakthrough in this domain is detailed in Chinese Patent CN113045489B, which discloses a highly efficient preparation method for 3-arylquinolin-2(1H)one derivatives. This innovation leverages a sophisticated palladium-catalyzed aminocarbonylation strategy that fundamentally alters the synthetic landscape by utilizing benzisoxazole not merely as a reactant, but as a dual-purpose reagent acting simultaneously as the nitrogen source and the formyl source. This approach addresses long-standing challenges in heterocycle synthesis, offering a pathway that is operationally simple, employs readily available starting materials, and exhibits remarkable tolerance for diverse functional groups. For R&D directors and procurement managers alike, this patent represents a pivotal shift towards more sustainable and economically viable manufacturing processes for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one core has relied heavily on classical methodologies such as the Vilsmeier-Haack reaction, the Knorr synthesis, or the Friedlander condensation. While these methods have served the industry for decades, they are increasingly viewed through a modern lens as suboptimal for large-scale commercial production due to several inherent drawbacks. Traditional routes often necessitate the use of harsh reaction conditions, including strong acids or bases and elevated temperatures that can degrade sensitive functional groups, thereby limiting the structural diversity of the final products. Furthermore, many conventional protocols involve multi-step sequences with poor atom economy, generating substantial quantities of chemical waste that complicate downstream purification and environmental compliance. The reliance on toxic reagents, such as phosphorus oxychloride in Vilsmeier-Haack reactions or unstable diazo compounds in other variations, poses significant safety risks and increases the operational costs associated with hazard management and waste disposal in a GMP manufacturing environment.
The Novel Approach
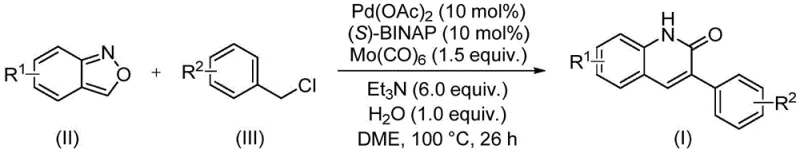
In stark contrast to these legacy methods, the technology described in patent CN113045489B introduces a streamlined, transition-metal-catalyzed pathway that elegantly bypasses many of these bottlenecks. The core of this innovation lies in the palladium-catalyzed coupling of benzisoxazole derivatives with benzyl chloride compounds. By employing benzisoxazole as a unique synthon that provides both the nitrogen atom and the carbonyl carbon required for the lactam ring formation, the process eliminates the need for external carbon monoxide gas or hazardous formylating agents. The reaction proceeds smoothly in ethylene glycol dimethyl ether (DME) at a moderate temperature of 100°C over a period of 26 hours, utilizing a catalytic system composed of palladium acetate and the chiral ligand (S)-BINAP, with molybdenum hexacarbonyl serving as the solid CO source. This methodology not only simplifies the operational procedure but also significantly enhances the safety profile of the synthesis, making it an attractive candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
From a mechanistic perspective, this transformation represents a complex yet highly controlled orchestration of organometallic steps that ensure high fidelity and yield. The catalytic cycle likely initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from the Pd(OAc)2 precursor and the (S)-BINAP ligand. This step forms a key organopalladium(II) intermediate, which subsequently undergoes migratory insertion of carbon monoxide released from the thermal decomposition of molybdenum hexacarbonyl. The resulting acyl-palladium species then engages in a nucleophilic attack by the nitrogen atom of the benzisoxazole ring, followed by ring-opening and cyclization events that construct the quinolinone framework. The presence of water and triethylamine plays a crucial role in facilitating proton transfer and neutralizing the hydrochloric acid byproduct, driving the equilibrium towards the desired product. Understanding these mechanistic nuances is vital for R&D teams aiming to optimize reaction parameters or adapt the protocol for novel substrates, as it highlights the delicate balance between catalyst activity and substrate stability.
Furthermore, the impurity profile of this reaction is exceptionally clean, a feature attributable to the high chemoselectivity of the palladium catalyst system. The use of benzisoxazole as a masked formamide equivalent prevents the formation of common side products associated with direct formylation, such as over-acylated species or polymeric tars often seen in Friedel-Crafts type reactions. The broad functional group tolerance observed across various examples, including those with electron-withdrawing groups like cyano and trifluoromethyl, as well as electron-donating groups like methoxy and tert-butyl, suggests that the catalytic cycle is robust against competitive coordination or deactivation. This level of control over the reaction pathway ensures that the crude reaction mixture contains minimal structurally related impurities, thereby reducing the burden on downstream purification processes and enhancing the overall purity of the final API intermediate.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
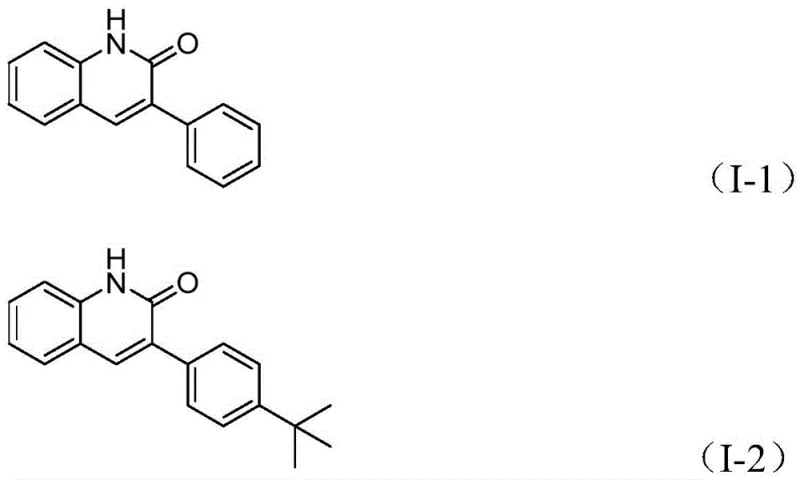
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings, requiring standard equipment and commercially sourced reagents. The protocol dictates a precise stoichiometric balance, typically employing a molar ratio of benzisoxazole to benzyl chloride of approximately 1:2.5, ensuring that the electrophile is in excess to drive the reaction to completion without necessitating complex recycling strategies. The reaction is conducted in a sealed vessel to maintain the integrity of the volatile solvent and the carbon monoxide generated in situ, heated to 100°C for a duration of 26 hours to maximize conversion. Following the reaction, the workup procedure is remarkably straightforward, involving simple filtration to remove insoluble metal residues and salts, followed by adsorption onto silica gel and purification via column chromatography. Detailed standardized synthesis steps for specific derivatives are outlined in the guide below.
- Charge a sealed tube with palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in DME solvent.
- Heat the reaction mixture to 100°C and stir continuously for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling strategic advantages that extend beyond mere chemical efficiency. The primary value proposition lies in the significant simplification of the raw material supply chain; benzisoxazoles and substituted benzyl chlorides are commodity chemicals available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. This abundance of starting materials translates directly into enhanced supply chain reliability and reduced lead times for high-purity pharmaceutical intermediates, as there is no reliance on exotic or custom-synthesized precursors that often bottleneck production schedules. Moreover, the elimination of gaseous carbon monoxide cylinders from the process inventory removes a major safety hazard and regulatory hurdle, streamlining facility compliance and reducing insurance and storage costs associated with toxic gas handling.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the high atom economy and the use of inexpensive, earth-abundant catalysts relative to the value of the product. By utilizing benzisoxazole as a dual nitrogen and carbon source, the process effectively consolidates two reagent costs into one, while the high yields reported (often exceeding 90% for optimized substrates) minimize raw material waste. The avoidance of cryogenic conditions or high-pressure reactors further reduces capital expenditure and energy consumption, leading to substantial cost savings in the overall manufacturing budget without compromising on quality or throughput.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, a critical factor for maintaining uninterrupted supply to downstream API manufacturers. The tolerance for a wide range of substituents allows for the flexible production of diverse analog libraries from a common set of building blocks, enabling rapid response to changing market demands or clinical trial requirements. This flexibility, combined with the use of stable solid reagents like Mo(CO)6 instead of pressurized gas, significantly de-risks the logistics of raw material transport and storage, ensuring a steady flow of materials even in volatile geopolitical environments.
- Scalability and Environmental Compliance: Scaling this process from gram to kilogram and eventually to metric ton levels is facilitated by the homogeneous nature of the reaction and the absence of exothermic hazards typical of traditional nitration or oxidation steps. The simplified workup, which avoids extensive aqueous extractions and reduces solvent usage through efficient chromatography, aligns with green chemistry principles by minimizing the generation of hazardous liquid waste. This environmental compatibility not only lowers waste disposal costs but also future-proofs the manufacturing process against increasingly stringent global environmental regulations, securing the long-term viability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology, providing clarity for stakeholders evaluating its potential integration into their existing portfolios. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the primary advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (carbonyl) source, eliminating the need for separate toxic carbonylation reagents like carbon monoxide gas and simplifying the reaction setup significantly.
Q: How does this method compare to traditional Friedlander or Vilsmeier-Haack reactions?
A: Unlike traditional methods that often require harsh acidic conditions or generate significant waste, this palladium-catalyzed approach operates under milder conditions (100°C) with excellent functional group tolerance and higher atom economy.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes commercially available and inexpensive starting materials like benzyl chlorides and benzisoxazoles, and the workup involves standard filtration and chromatography, making it highly amenable to scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
As the global demand for complex heterocyclic intermediates continues to surge, partnering with a CDMO that possesses deep technical expertise in advanced catalytic methodologies is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver exceptional results for our clients. Our state-of-the-art facilities are equipped to handle the specific requirements of palladium-catalyzed reactions, including rigorous QC labs that enforce stringent purity specifications to ensure every batch meets the highest international standards. We understand that the transition from bench-scale discovery to commercial manufacturing requires not just chemical knowledge, but a holistic approach to process safety, cost optimization, and regulatory compliance.
We invite pharmaceutical companies and research institutions to collaborate with us to unlock the full potential of this innovative synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific project needs, along with specific COA data and route feasibility assessments for your target molecules. Let us help you accelerate your drug development timeline and secure a reliable, cost-effective supply of high-quality 3-arylquinolin-2(1H)one derivatives for your next breakthrough therapy.
