Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of 3-Arylquinolin-2(1H)one Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for active pharmaceutical ingredients (APIs). A significant breakthrough in this domain is detailed in patent CN113045489B, which discloses a novel preparation method for 3-arylquinolin-2(1H)one derivatives. These compounds are not merely academic curiosities; they represent a vital class of heterocycles found in numerous natural products and bioactive molecules, exhibiting potent activities as antibiotics, antiplatelet agents, and antitumor drugs. The structural versatility of the quinolin-2(1H)one core allows for extensive derivatization, making it a cornerstone in medicinal chemistry programs targeting various therapeutic areas including endothelin receptor antagonism. As a reliable pharmaceutical intermediate supplier, understanding the nuances of such patented methodologies is essential for delivering high-quality materials to global R&D teams.
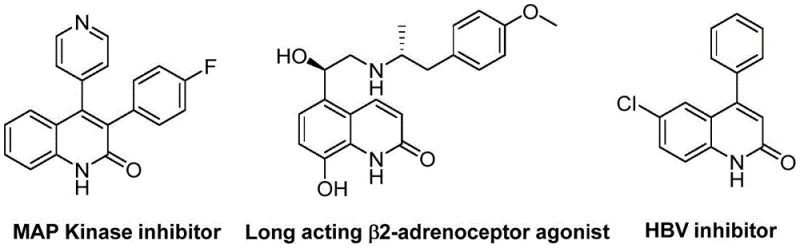
The significance of this technology extends beyond mere molecular construction; it addresses the pressing need for sustainable and cost-effective manufacturing processes in the fine chemical sector. Traditional syntheses of these scaffolds often rely on multi-step sequences involving hazardous reagents or expensive catalysts that complicate supply chains. In contrast, the method described in CN113045489B utilizes a palladium-catalyzed aminocarbonylation strategy that streamlines the assembly of the quinolinone ring system. By leveraging benzisoxazole as a unique dual-purpose reagent, the process achieves high atom economy while maintaining excellent functional group tolerance. This innovation positions manufacturers to offer cost reduction in API manufacturing by reducing the number of synthetic steps and minimizing waste generation, thereby aligning with modern green chemistry principles and regulatory expectations for cleaner production technologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the quinolin-2(1H)one skeleton has relied heavily on classical named reactions such as the Vilsmeier-Haack, Knorr, and Friedlander condensations. While these methods have served the industry for decades, they are increasingly viewed as suboptimal for modern large-scale production due to several inherent drawbacks. The Vilsmeier-Haack reaction, for instance, typically requires the use of phosphorus oxychloride and dimethylformamide, generating substantial amounts of acidic waste that necessitates complex neutralization and disposal procedures. Furthermore, these traditional routes often suffer from limited substrate scope, struggling to accommodate sensitive functional groups without requiring extensive protection-deprotection strategies. Such limitations not only increase the overall cost of goods but also extend the lead time for high-purity pharmaceutical intermediates, creating bottlenecks in drug development pipelines. Additionally, the reliance on stoichiometric amounts of harsh reagents poses significant safety risks in commercial scale-up of complex organic molecules, demanding specialized equipment and rigorous safety protocols that drive up capital expenditure.
The Novel Approach
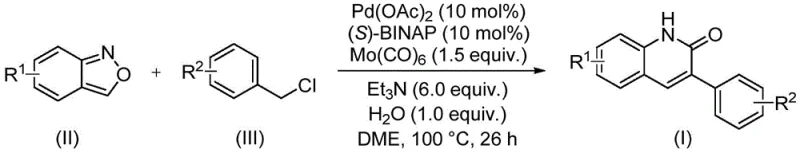
The methodology presented in patent CN113045489B offers a transformative alternative by employing a transition metal-catalyzed carbonylation coupling reaction. This approach fundamentally shifts the paradigm from stoichiometric reagent consumption to catalytic efficiency, utilizing a palladium catalyst system in conjunction with molybdenum hexacarbonyl as a solid carbon monoxide surrogate. The brilliance of this design lies in the utilization of benzisoxazole, which acts simultaneously as the nitrogen source and the formyl source, effectively bypassing the need for gaseous CO handling which is a major safety hazard in industrial settings. This novel pathway enables the direct coupling of readily available benzisoxazoles with benzyl chloride compounds under relatively mild thermal conditions. The result is a highly efficient one-pot transformation that constructs the target heterocycle with impressive regioselectivity and yield. For procurement managers, this translates to a simplified supply chain where fewer raw materials are required, and the operational complexity is drastically reduced, facilitating a more agile response to market demands for specialized chemical intermediates.
Mechanistic Insights into Palladium-Catalyzed Aminocarbonylation
To fully appreciate the technical robustness of this synthesis, one must delve into the mechanistic intricacies of the palladium-catalyzed cycle. The reaction initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species, generated in situ from palladium acetate and the chiral ligand (S)-BINAP. This step is critical as it activates the carbon-chlorine bond, setting the stage for subsequent migratory insertion. The presence of molybdenum hexacarbonyl is pivotal; upon heating, it releases carbon monoxide in a controlled manner, which then coordinates to the palladium center to form an acyl-palladium complex. This in situ generation of CO avoids the logistical nightmares associated with high-pressure gas cylinders, enhancing the safety profile of the process significantly. The benzisoxazole then participates in a nucleophilic attack or insertion sequence, facilitated by the base triethylamine, which helps to deprotonate intermediates and drive the equilibrium forward. The final reductive elimination step releases the 3-arylquinolin-2(1H)one product and regenerates the palladium catalyst, completing the cycle. This elegant choreography of organometallic steps ensures high turnover numbers and minimizes the formation of palladium black, a common deactivation pathway in such reactions.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-temperature thermal cyclizations. The specificity of the palladium catalyst, tuned by the bulky and electron-rich (S)-BINAP ligand, ensures that side reactions such as homocoupling of the benzyl chloride or polymerization of the isoxazole ring are suppressed. The use of water as a co-reagent, though seemingly counterintuitive in organometallic chemistry, plays a subtle yet crucial role in hydrolyzing intermediate species to yield the final ketone functionality without over-hydrolysis of the sensitive lactam ring. This precise control over the reaction trajectory results in a clean crude reaction profile, which significantly eases the burden on downstream purification units. For R&D directors focused on purity specifications, this means that the resulting intermediates possess a superior impurity profile, reducing the risk of genotoxic impurities or difficult-to-remove byproducts that could compromise the safety of the final drug substance. The broad functional group tolerance observed, accommodating substituents like cyano, trifluoromethyl, and halogens, further underscores the versatility of this catalytic system in synthesizing diverse analog libraries.
How to Synthesize 3-Arylquinolin-2(1H)one Derivatives Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The protocol outlined in the patent emphasizes the importance of reagent ratios and reaction duration. Typically, the reaction is conducted in ethylene glycol dimethyl ether (DME) as the solvent, which provides the necessary solubility for both organic substrates and the inorganic base. The molar ratio of benzisoxazole to benzyl chloride is optimized at approximately 1:2.5 to ensure complete consumption of the limiting reagent, while the catalyst loading is kept low at 10 mol% to maintain cost efficiency. The reaction temperature is maintained at 100°C for a duration of 26 hours, a timeframe determined to be optimal for balancing conversion rates against energy costs. Detailed standardized synthesis steps for this procedure are provided in the guide below, ensuring that technical teams can replicate the high yields reported in the patent data, which range from 74% to 97% depending on the substrate electronics.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders managing the bottom line and logistical flow of chemical production, the adoption of this patented methodology offers compelling economic and operational benefits. The shift towards catalytic processes inherently reduces the mass intensity of the synthesis, meaning less waste is generated per kilogram of product. This reduction in waste volume directly correlates to lower disposal costs and a smaller environmental footprint, which is increasingly a criterion for vendor selection by major pharmaceutical companies. Furthermore, the reliance on bench-stable solid reagents like molybdenum hexacarbonyl instead of gaseous carbon monoxide eliminates the need for specialized high-pressure reactors and gas handling infrastructure. This simplification of equipment requirements lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for faster technology transfer between sites. The robustness of the reaction conditions also implies a wider operating window, reducing the risk of batch failures due to minor fluctuations in temperature or mixing, thereby enhancing overall supply chain reliability and consistency of supply.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and commercially abundant starting materials. Benzisoxazoles and benzyl chlorides are commodity chemicals available from multiple global suppliers, mitigating the risk of single-source dependency and price volatility. By eliminating the need for pre-functionalized precursors or exotic reagents, the raw material cost per kilogram of the final intermediate is significantly optimized. Additionally, the high reaction efficiency and yields reported, often exceeding 90% for unsubstituted or electron-neutral substrates, mean that less starting material is wasted, further driving down the effective cost of production. The simplified workup procedure, involving basic filtration and standard chromatography, reduces the consumption of solvents and silica gel compared to more tedious multi-step syntheses, contributing to substantial cost savings in the overall manufacturing budget without compromising on quality.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the simplicity and modularity of this synthetic route. Since the key reagents are stable solids that do not require cold chain logistics or special hazmat shipping classifications beyond standard organic chemicals, procurement lead times are minimized. The ability to synthesize a wide variety of derivatives by simply swapping the R-groups on the benzyl chloride or benzisoxazole allows manufacturers to maintain a flexible inventory strategy. Instead of stocking dozens of different finished intermediates, a facility can stock the common core reagents and produce specific derivatives on demand, responding rapidly to client requests for custom analogs. This agility is crucial in the fast-paced environment of drug discovery where timelines are tight and the ability to deliver gram-to-kilogram quantities of novel structures quickly can accelerate a client's project milestones significantly.
- Scalability and Environmental Compliance: Scaling this reaction from gram to multi-kilogram levels is straightforward due to the absence of exothermic hazards associated with gas evolution or highly reactive intermediates. The use of DME as a solvent, while requiring appropriate recovery systems, is well-established in the industry, and the reaction mixture remains homogeneous or easily manageable throughout the process. From an environmental compliance standpoint, the atom economy is superior to traditional condensation reactions, generating fewer byproducts that require treatment. The catalytic nature of the palladium system means that heavy metal usage is minimized, and established scavenging technologies can effectively reduce residual palladium levels in the final product to meet stringent ICH Q3D guidelines. This alignment with green chemistry metrics makes the process attractive for companies aiming to improve their sustainability scores and meet corporate social responsibility goals regarding chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and background analysis of patent CN113045489B, providing clarity for technical teams evaluating this route for their specific projects. Understanding these nuances helps in making informed decisions about process adoption and resource allocation.
Q: What are the primary advantages of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual function as both the nitrogen source and the formyl source, eliminating the need for separate carbon monoxide gas sources and simplifying the reaction setup significantly.
Q: How does this method compare to traditional Vilsmeier-Haack reactions?
A: Unlike traditional methods that often require harsh conditions and generate significant waste, this palladium-catalyzed approach operates under milder conditions with broader functional group tolerance and higher atom economy.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of commercially available starting materials, simple post-processing involving filtration and chromatography, and high reaction efficiency make it highly scalable for industrial API intermediate production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic methodologies like the palladium-catalyzed carbonylation described in CN113045489B. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering high-purity 3-arylquinolin-2(1H)one derivatives that meet the rigorous quality standards demanded by the global pharmaceutical industry. Our state-of-the-art facilities are equipped with rigorous QC labs capable of performing comprehensive impurity profiling and structural confirmation, guaranteeing that every batch shipped adheres to stringent purity specifications. Whether you require custom synthesis of novel analogs or reliable supply of known intermediates, our infrastructure is designed to support your long-term development goals with consistency and excellence.
We invite you to collaborate with us to leverage this cutting-edge technology for your next drug development program. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific molecule, identifying opportunities to optimize your supply chain further. We encourage potential clients to contact our technical procurement team to request specific COA data for our catalog items or to discuss route feasibility assessments for your proprietary compounds. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing, delivering value through innovation, quality, and unwavering reliability.
