Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)ones for Commercial API Manufacturing
Advanced Pd-Catalyzed Synthesis of 3-Arylquinolin-2(1H)ones for Commercial API Manufacturing
The pharmaceutical industry continuously seeks robust and efficient synthetic routes for heterocyclic scaffolds that serve as critical building blocks for drug discovery and development. Patent CN113045489B introduces a groundbreaking preparation method for 3-arylquinolin-2(1H) ketone derivatives, a class of compounds renowned for their presence in numerous natural products and biologically active molecules. These derivatives function as potent antibiotics, antiplatelet agents, antitumor drugs, and receptor antagonists, making them indispensable in modern medicinal chemistry. The disclosed technology leverages a palladium-catalyzed aminocarbonylation strategy that fundamentally shifts the paradigm of quinolinone synthesis by utilizing benzisoxazole as a dual-purpose reagent. This innovation not only streamlines the synthetic pathway but also addresses key safety and cost concerns associated with traditional carbonylation methods. As a leading entity in fine chemical manufacturing, we recognize the immense potential of this methodology to enhance the production of high-purity pharmaceutical intermediates.
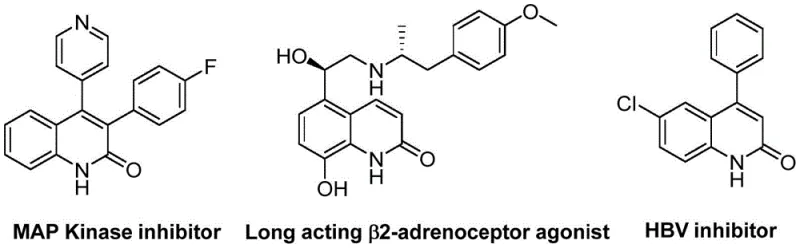
The significance of this technological advancement cannot be overstated when considering the broader landscape of organic synthesis. Quinolin-2(1H)one derivatives have historically been synthesized via classical methods such as Vilsmeier-Haack, Knorr, and Friedlander reactions, which often suffer from limitations regarding regioselectivity, harsh reaction conditions, or the requirement for pre-functionalized starting materials. Furthermore, transition metal-catalyzed improvements and ring-closing metathesis have been explored, yet challenges remain in terms of atom economy and operational simplicity. The approach detailed in CN113045489B overcomes these hurdles by employing a catalytic system that operates under relatively mild thermal conditions while maintaining exceptional substrate compatibility. This ensures that complex molecular architectures can be accessed with high fidelity, providing R&D teams with a reliable tool for generating diverse chemical libraries for biological screening.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to quinolinone cores frequently rely on the use of toxic carbon monoxide gas under high pressure or involve multi-step sequences that degrade overall yield and increase waste generation. For instance, classic carbonylation reactions often necessitate specialized high-pressure autoclaves, posing significant safety risks and capital expenditure barriers for manufacturing facilities. Additionally, methods relying on pre-formed anilines or complex precursors limit the structural diversity accessible to chemists, as the introduction of sensitive functional groups may be incompatible with the harsh acidic or basic conditions typically employed. These constraints result in prolonged development timelines and inflated costs, particularly when scaling up processes for commercial API manufacturing. The reliance on hazardous reagents also complicates regulatory compliance and environmental safety protocols, creating bottlenecks in the supply chain for critical pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel method described in the patent utilizes benzisoxazole as both a nitrogen source and a formyl source, effectively acting as a solid surrogate for carbon monoxide and ammonia equivalents. This strategic design eliminates the need for handling gaseous CO, thereby drastically improving operational safety and simplifying the reactor setup to standard sealed tubes or vessels. The reaction proceeds efficiently at 100°C using a palladium catalyst system comprising Pd(OAc)2, (S)-BINAP, and Mo(CO)6 in the presence of triethylamine and water. This one-pot transformation allows for the direct coupling of readily available benzyl chloride compounds with benzisoxazoles, delivering 3-arylquinolin-2(1H)ones in high yields. The simplicity of the protocol, combined with the commercial availability of starting materials, positions this method as a superior alternative for cost reduction in pharmaceutical intermediate manufacturing.
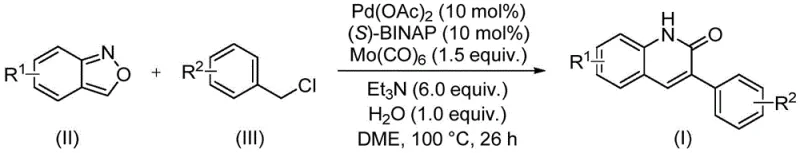
Mechanistic Insights into Pd-Catalyzed Aminocarbonylation
The core of this synthetic breakthrough lies in the intricate palladium-catalyzed aminocarbonylation mechanism that facilitates the construction of the quinolinone ring system. The catalytic cycle initiates with the oxidative addition of the benzyl chloride substrate to the active palladium(0) species generated in situ from Pd(OAc)2 and the chiral ligand (S)-BINAP. Subsequently, the insertion of carbon monoxide, released from the decomposition of molybdenum hexacarbonyl (Mo(CO)6), forms an acyl-palladium intermediate. Crucially, the benzisoxazole molecule undergoes ring opening or activation to provide the necessary nitrogen nucleophile and the carbonyl carbon equivalent, which then engages with the acyl-palladium species. This sequence is followed by intramolecular cyclization and reductive elimination to release the final 3-arylquinolin-2(1H)one product while regenerating the palladium catalyst. The inclusion of water and triethylamine plays a vital role in facilitating proton transfer and neutralizing acid byproducts, ensuring the catalytic turnover remains high throughout the 26-hour reaction period.
From an impurity control perspective, this mechanism offers distinct advantages over conventional pathways. The mild reaction temperature of 100°C minimizes thermal degradation of sensitive functional groups, thereby reducing the formation of tar-like byproducts often seen in high-temperature condensations. Furthermore, the use of Mo(CO)6 as a controlled CO source prevents the local excess of carbon monoxide that can lead to over-carbonylation or side reactions. The high functional group tolerance observed in the patent data, accommodating substituents such as cyano, trifluoromethyl, and various halogens, indicates that the catalytic system is robust against electronic variations in the substrate. This stability ensures a cleaner crude reaction profile, which simplifies downstream purification processes like column chromatography and enhances the overall purity of the isolated API intermediate, meeting the stringent quality specifications required by global regulatory bodies.
How to Synthesize 3-Arylquinolin-2(1H)one Efficiently
The practical implementation of this synthesis is designed for ease of execution in both laboratory and pilot plant settings. The protocol involves charging a reaction vessel with the palladium catalyst, ligand, carbonyl source, base, and substrates in a suitable solvent such as dimethoxyethane (DME). The mixture is then heated to promote the transformation, after which standard workup procedures involving filtration and silica gel treatment are employed. This straightforward workflow minimizes the need for specialized equipment or exotic reagents, making it accessible for widespread adoption. For detailed operational parameters and specific stoichiometric ratios optimized for maximum yield, please refer to the standardized synthesis guide below.
- Combine palladium acetate, (S)-BINAP, molybdenum hexacarbonyl, triethylamine, water, benzisoxazole, and benzyl chloride compound in a sealed tube with DME solvent.
- Heat the reaction mixture to 100°C and maintain stirring for approximately 26 hours to ensure complete conversion.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target 3-arylquinolin-2(1H)one derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift towards using benzisoxazole and benzyl chlorides as primary feedstocks leverages a supply chain built on commodity chemicals that are widely produced and globally available. This abundance mitigates the risk of raw material shortages that often plague specialized synthetic routes dependent on custom-made precursors. Moreover, the elimination of high-pressure carbon monoxide gas removes a significant safety hazard and the associated costs of specialized containment infrastructure, leading to substantial cost savings in facility operations and insurance. The robustness of the reaction conditions also implies a lower rate of batch failures, ensuring consistent output and reliable delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the use of inexpensive and commercially available starting materials such as benzisoxazole and substituted benzyl chlorides. By avoiding the need for high-pressure reactors and toxic gas handling systems, capital expenditure for manufacturing setup is significantly reduced. Additionally, the high reaction efficiency and yield minimize raw material waste, directly lowering the cost of goods sold (COGS) for the final pharmaceutical intermediate. The simplified workup procedure further reduces labor and solvent consumption, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical factor for long-term project success, and this method excels by utilizing bench-stable reagents that do not require cold chain logistics or special permits for transport. The reliance on standard organic solvents like DME and common bases like triethylamine ensures that supply disruptions are unlikely. This reliability allows for better inventory planning and reduces the lead time for high-purity pharmaceutical intermediates, enabling faster time-to-market for new drug candidates. The scalability of the process from gram to kilogram scales ensures that supply can easily ramp up to meet commercial demand without process re-engineering.
- Scalability and Environmental Compliance: The environmental footprint of chemical manufacturing is increasingly scrutinized, and this green chemistry approach offers a cleaner alternative to traditional methods. The absence of gaseous CO emissions and the use of a closed system for carbonylation align with strict environmental regulations. The high atom economy of the reaction means less waste generation per unit of product, simplifying waste treatment protocols. Furthermore, the ability to run the reaction in standard sealed tubes at moderate temperatures demonstrates excellent scalability, allowing for seamless transition from R&D to commercial scale-up of complex pharmaceutical intermediates without compromising safety or quality.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is essential for stakeholders evaluating its integration into their production pipelines. The following questions address common inquiries regarding the catalyst system, substrate scope, and operational safety, derived directly from the technical disclosures in the patent literature. These insights are intended to clarify the feasibility of implementing this route for specific project requirements and to highlight the versatility of the methodology across different chemical contexts.
Q: What is the primary advantage of using benzisoxazole in this synthesis?
A: Benzisoxazole serves a dual role as both the nitrogen source and the formyl (C1) source, eliminating the need for external carbon monoxide gas and simplifying the reaction setup significantly.
Q: Is this method suitable for large-scale production of pharmaceutical intermediates?
A: Yes, the method utilizes commercially available starting materials and standard heating conditions (100°C) without high-pressure equipment, making it highly scalable for industrial manufacturing.
Q: What is the functional group tolerance of this Pd-catalyzed reaction?
A: The reaction exhibits excellent tolerance for various functional groups including halogens, alkoxy groups, cyano, and trifluoromethyl groups, allowing for the synthesis of diverse derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Arylquinolin-2(1H)one Supplier
At NINGBO INNO PHARMCHEM, we are committed to translating cutting-edge academic research into commercial reality for our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature are faithfully reproduced at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-arylquinolin-2(1H)one derivative meets the highest standards required for pharmaceutical applications. Our expertise in palladium-catalyzed transformations allows us to optimize this specific route for maximum efficiency and cost-effectiveness, providing you with a competitive edge in your drug development programs.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your supply chain goals. Let us be your trusted partner in delivering high-quality chemical solutions that drive innovation in the healthcare sector.
