Advanced Spiro-Fluorene Derivatives for High-Performance OLED Manufacturing and Commercial Scale-Up
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that not only deliver exceptional luminous efficiency but also possess the thermal and morphological stability required for long-term device operation. Patent CN112409314A introduces a novel class of organic electroluminescent compounds, specifically designed as high-performance hole transport materials (HTMs). These compounds, characterized by a rigid spiro-fluorene core substituted with various aryl and heteroaryl amines, address critical failure modes in OLED devices such as crystallization and voltage drift. By strategically modifying the substituent positions and incorporating heterocyclic ligands, the invention achieves a balance between high hole mobility and low ionization potential. For R&D directors and procurement specialists seeking a reliable OLED material supplier, this technology represents a significant leap forward in stabilizing light-emitting performance while simplifying the manufacturing complexity associated with traditional small molecule HTMs.
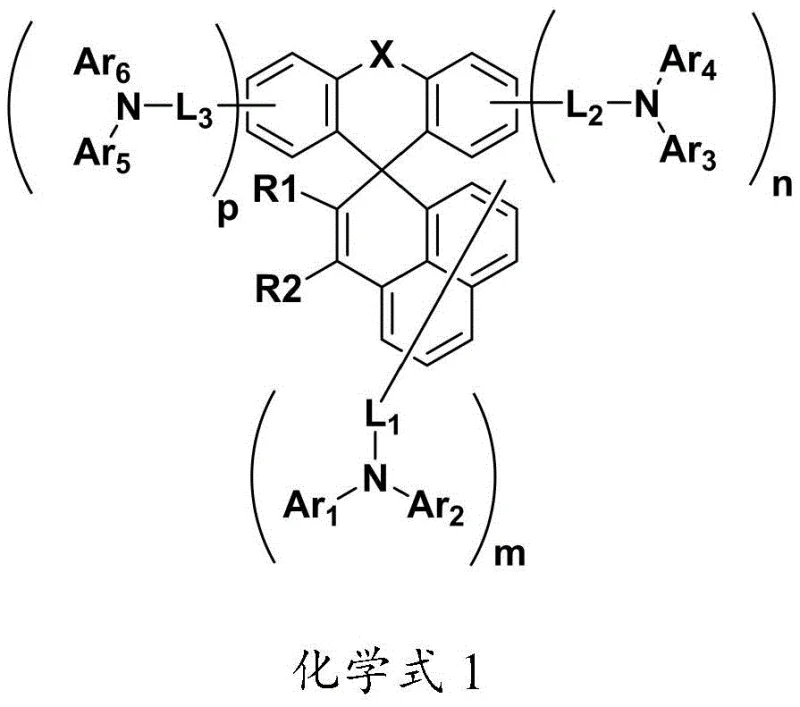
The development of efficient hole transport layers is pivotal for the commercial viability of next-generation displays. Conventional materials often suffer from low glass transition temperatures, leading to morphological instability under the heat generated during device operation. This patent discloses a sophisticated molecular architecture where the spiro-connection effectively inhibits molecular packing and crystallization. Unlike linear conjugated systems that tend to aggregate, the three-dimensional spiro-structure maintains an amorphous state, which is crucial for uniform film formation. Furthermore, the inclusion of electron-donating amine units enhances the hole injection capability from the anode, thereby reducing the overall driving voltage of the device. This structural innovation directly translates to lower power consumption and extended operational lifetimes, key metrics for any electronics manufacturer aiming to reduce cost in electronic chemical manufacturing through improved device yield and longevity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hole transport materials such as TPD (N,N'-diphenyl-N,N'-bis(3-methylphenyl)-(1,1'-biphenyl)-4,4'-diamine) and NPB (N,N'-di(1-naphthyl)-N,N'-diphenylbenzidine) have long served as industry standards, yet they exhibit inherent limitations that hinder the performance of high-resolution displays. These planar molecules possess relatively low glass transition temperatures, making them susceptible to crystallization during the thermal evaporation process or device operation, which creates dark spots and reduces luminous efficiency. Additionally, their oxidative stability is often compromised, leading to the formation of quenching sites that degrade the organic layers over time. From a supply chain perspective, the synthesis of some high-performance alternatives often involves complex multi-step routes with harsh conditions or expensive catalysts that are difficult to remove, posing challenges for achieving the ultra-high purity (>99.9%) required for commercial OLED panels. These factors collectively increase the total cost of ownership and limit the scalability of production.
The Novel Approach
The synthetic strategy outlined in CN112409314A overcomes these hurdles through a modular four-step sequence that balances yield, purity, and scalability. The process begins with a controlled lithiation reaction at cryogenic temperatures (-70°C to -78°C), ensuring precise regioselectivity during the formation of the carbon-carbon bond. This is followed by an acid-catalyzed cyclization step using concentrated sulfuric acid in glacial acetic acid at 120°C, which efficiently constructs the rigid spiro-fluorene skeleton in a short reaction time of merely 5 minutes. Subsequent functionalization via Suzuki-Miyaura cross-coupling and Buchwald-Hartwig amination allows for the diverse introduction of aryl and heteroaryl groups without compromising the integrity of the core structure. This approach not only streamlines the synthesis but also facilitates the removal of impurities through standard workup procedures like recrystallization and column chromatography, ensuring the final product meets the stringent quality specifications demanded by top-tier display manufacturers.
Mechanistic Insights into Acid-Catalyzed Cyclization and Pd-Catalyzed Coupling
The core of this technology lies in the efficient construction of the spiro-fluorene framework, achieved through a dehydration cyclization mechanism. In the second step of the synthesis, the hydroxy-intermediate undergoes protonation by sulfuric acid, generating a stable carbocation species that is immediately attacked by the adjacent aromatic ring. This intramolecular Friedel-Crafts-type alkylation closes the ring to form the spiro-center, a process that is thermodynamically driven by the formation of the stable aromatic system and the release of water. The use of glacial acetic acid as a solvent provides a polar medium that stabilizes the ionic intermediates while maintaining solubility of the organic substrates. Following the core formation, the attachment of functional arms is executed via palladium-catalyzed cross-coupling reactions. The Suzuki coupling utilizes Pd(PPh3)4 to link aryl boronic acids with halogenated intermediates, a reaction known for its tolerance to various functional groups and mild conditions. This is complemented by the Buchwald-Hartwig amination, employing Pd2(dba)3 and bulky phosphine ligands like P(t-Bu)3, which facilitate the formation of C-N bonds even with sterically hindered substrates, ensuring high conversion rates and minimal side products.
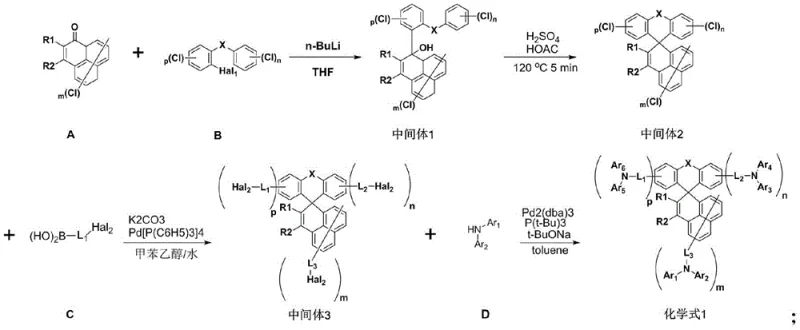
Impurity control is rigorously managed throughout the synthetic pathway to prevent the incorporation of trace metals or organic byproducts that could act as charge traps. The initial lithiation step is quenched carefully with saturated ammonium chloride to avoid exothermic runaways, and the subsequent extraction processes effectively remove inorganic salts. During the palladium-catalyzed steps, the choice of ligands and bases is critical; for instance, the use of potassium carbonate in the Suzuki step helps maintain a basic environment necessary for the transmetallation cycle, while sodium tert-butoxide in the amination step activates the amine nucleophile. Post-reaction purification involves filtration through diatomite to remove bulk catalyst residues, followed by multiple washing steps with water and organic solvents. The final purification via column chromatography using dichloromethane and petroleum ether ensures that isomeric impurities and unreacted starting materials are separated, resulting in a material with HPLC purity exceeding 99%, which is essential for preventing premature device failure.
How to Synthesize High-Purity Spiro-Fluorene Derivatives Efficiently
The preparation of these advanced OLED materials requires precise control over reaction parameters to maximize yield and minimize batch-to-batch variability. The protocol detailed in the patent provides a robust framework for synthesizing compounds like HT-9, HT-25, and HT-42, which have demonstrated superior performance in device testing. The process leverages widely available industrial chemicals and standard reactor setups, making it highly adaptable for both pilot-scale and full-scale commercial production. By adhering to the specified molar ratios and temperature profiles, manufacturers can consistently achieve yields above 80% for intermediates and over 83% for the final products. For a detailed breakdown of the specific operational parameters, stoichiometry, and workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Perform lithiation of aryl bromide precursors at -78°C followed by nucleophilic addition to ketone substrates to form hydroxy-intermediates.
- Execute acid-catalyzed cyclization using concentrated sulfuric acid in glacial acetic acid at 120°C to construct the rigid spiro-fluorene core.
- Conduct Suzuki-Miyaura cross-coupling reactions using palladium catalysts to attach aryl groups, followed by Buchwald-Hartwig amination for final functionalization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers distinct logistical and economic advantages over legacy methods. The reliance on commodity chemicals such as tetrahydrofuran, toluene, and common aryl halides reduces dependency on exotic or single-source reagents, thereby mitigating supply risk. Furthermore, the high yields reported in the examples (ranging from 80% to 87% for intermediates) indicate a material-efficient process that minimizes waste generation and raw material consumption. The use of standard palladium catalysts, while precious, is optimized through the use of efficient ligand systems that allow for effective turnover, and the catalysts can be removed via standard filtration techniques, avoiding the need for expensive scavenger resins. This streamlined workflow significantly reduces the operational complexity and associated costs in electronic chemical manufacturing, allowing for more competitive pricing structures without compromising on the quality of the final electronic material.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for cryogenic conditions beyond the initial lithiation step, with subsequent reactions proceeding at manageable temperatures like 90°C to 120°C. This reduction in energy intensity, combined with short reaction times (e.g., 5 minutes for cyclization), drastically lowers utility costs per kilogram of product. Additionally, the high selectivity of the reactions minimizes the formation of difficult-to-separate byproducts, reducing the load on downstream purification units and lowering solvent consumption. The overall process design prioritizes atom economy and operational simplicity, which translates into substantial cost savings when scaling from laboratory batches to multi-ton annual production capacities.
- Enhanced Supply Chain Reliability: The starting materials, including various brominated aromatics and boronic acids, are widely produced by the global fine chemical industry, ensuring a stable and redundant supply base. This diversity of suppliers prevents bottlenecks that often plague specialized OLED material chains. Moreover, the robustness of the chemistry means that slight variations in raw material quality can be accommodated without catastrophic batch failures, enhancing the reliability of delivery schedules. The ability to synthesize a wide library of derivatives (HT-1 to HT-88) using the same core platform allows manufacturers to pivot quickly between different product grades based on market demand, providing flexibility in inventory management and reducing lead time for high-purity organic electroluminescent compounds.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents or extreme pressures, aligning well with modern environmental, health, and safety (EHS) standards. Solvent systems like toluene/ethanol/water are easily separable and recyclable, minimizing hazardous waste disposal costs. The solid-state nature of the intermediates allows for easy isolation and storage, facilitating a campaign-based production mode that is ideal for large-scale manufacturing. The high thermal stability of the final products also ensures safe handling and transport, reducing the risk of degradation during logistics. These factors collectively support a sustainable and scalable supply chain capable of meeting the growing demands of the global display industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this technology. They are derived from the specific experimental data and performance metrics provided in the patent documentation, offering clarity on the practical aspects of utilizing these spiro-fluorene derivatives in OLED fabrication. Understanding these details is crucial for integrating new materials into existing production lines and ensuring optimal device performance.
Q: What are the thermal stability characteristics of these spiro-fluorene derivatives?
A: The compounds exhibit high glass transition temperatures (Tg) ranging from approximately 177°C to 182°C, ensuring excellent morphological stability during device operation and preventing crystallization.
Q: How does the synthetic route ensure high purity for electronic grade applications?
A: The process utilizes robust purification steps including recrystallization from ethanol and petroleum ether, followed by column chromatography, consistently achieving HPLC purity levels greater than 99%.
Q: Can this manufacturing process be scaled for industrial OLED production?
A: Yes, the synthesis relies on standard industrial reactions such as Suzuki coupling and acid cyclization using commercially available reagents like Pd(PPh3)4 and n-BuLi, facilitating straightforward scale-up from grams to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Organic Electroluminescent Compound Supplier
The technical potential of the spiro-fluorene derivatives described in CN112409314A is immense, offering a pathway to OLED devices with superior efficiency and lifespan. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to mass market deployment is seamless. Our facility is equipped with rigorous QC labs and stringent purity specifications capable of handling the sensitive nature of electronic chemicals. We understand that consistency is key in the display industry, and our dedicated process engineering team works tirelessly to optimize every step of the synthesis, from raw material sourcing to final packaging, guaranteeing a supply of high-purity OLED material that meets the exacting standards of leading panel manufacturers.
We invite you to collaborate with us to leverage this advanced chemistry for your next-generation products. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and application needs. Please contact us to request specific COA data for compounds like HT-9 or HT-25, and to discuss route feasibility assessments for custom derivatives. Let us be your partner in driving innovation and efficiency in the organic electronics sector.
