Advanced Decarboxylation Technology for Commercial Scale Fluoroalkyl Alcohol Manufacturing
The global demand for high-performance fluorinated solvents and intermediates continues to surge, driven by applications in pharmaceuticals, electronics, and advanced materials. A pivotal advancement in this sector is detailed in patent CN101679156A, which discloses a robust method for producing fluoroalkyl alcohols, such as hexafluoroisopropanol, through a catalytic decarboxylation process. This technology represents a significant departure from legacy synthesis routes, addressing critical pain points regarding toxicity, selectivity, and raw material costs. By leveraging specific amine compounds, quaternary ammonium salts, or quaternary phosphonium salts, manufacturers can now convert hydroxycarboxylic acid esters into target alcohols with exceptional efficiency. For R&D directors and procurement specialists seeking a reliable fluoroalkyl alcohol supplier, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a competitive supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of hexafluoroalkylisopropanol has relied heavily on the hydration of hexafluoroisobutene or the oxidation of ether derivatives. These traditional pathways are fraught with significant operational hazards and economic inefficiencies. Specifically, the direct use of hexafluoroisobutene involves handling highly toxic and reactive gases, necessitating expensive containment infrastructure and rigorous safety protocols that drive up capital expenditure. Furthermore, prior art methods, such as the decarboxylation of hydroxycarboxylates in protic solvents, often suffer from mediocre selectivity, typically hovering around 70%. This lack of precision results in complex impurity profiles that require energy-intensive downstream purification steps, thereby eroding profit margins and complicating the supply of high-purity fluoroalkyl alcohols required for sensitive electronic or pharmaceutical applications.
The Novel Approach
The innovative methodology described in the patent data introduces a transformative solution by utilizing hydroxycarboxylic acid esters or salts derived from industrial waste streams, specifically octafluoroisobutene by-products. This approach not only valorizes waste materials but also fundamentally alters the reaction landscape through the introduction of specific nitrogen-containing promoters. By conducting the decarboxylation in the presence of pyridines, amines, or quaternary salts, the reaction achieves markedly higher selectivity and conversion rates. The process allows for the use of inexpensive raw materials while mitigating the risks associated with toxic gaseous precursors. This shift enables a more sustainable and cost-effective manufacturing paradigm, positioning companies that adopt this technology as leaders in cost reduction in fluorine material manufacturing.
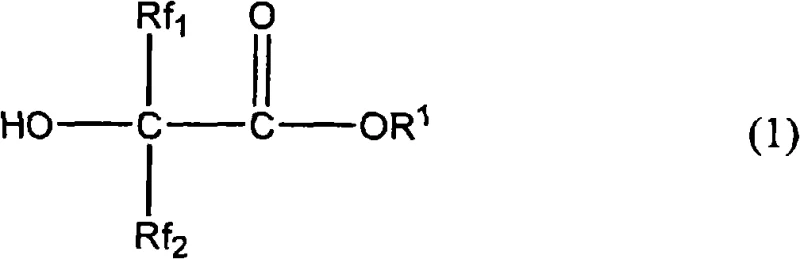
Mechanistic Insights into Amine-Catalyzed Decarboxylation
The core of this technological breakthrough lies in the specific interaction between the hydroxycarboxylic acid precursor and the nitrogen-based additives. The reaction mechanism involves the thermal decomposition of the carboxyl group, facilitated by the basic or nucleophilic nature of the added amines or quaternary salts. These additives likely stabilize the transition state or assist in the abstraction of the acidic proton, thereby lowering the activation energy required for carbon dioxide elimination. The patent specifies that the reaction can proceed effectively across a broad temperature range, from -20°C to 300°C, with optimal results observed between 5°C and 200°C. This flexibility suggests a robust catalytic cycle that is tolerant to varying thermal conditions, providing process engineers with significant leeway in reactor design and operation parameters.
Impurity control is another critical aspect where this mechanism excels. In conventional protic solvent systems, side reactions such as ether formation or polymerization can degrade product quality. However, the use of amide solvents like dimethylacetamide or dimethylformamide in conjunction with the specified catalysts creates a reaction environment that suppresses these competing pathways. The data indicates that selectivity can reach upwards of 80% under optimized conditions, significantly reducing the burden on purification units. For R&D teams focused on purity specifications, this means a cleaner crude product that requires fewer distillation cuts, directly translating to higher overall yields and reduced solvent consumption during the isolation phase.
How to Synthesize Hexafluoroisopropanol Efficiently
Implementing this synthesis route requires careful attention to reagent selection and reaction conditions to maximize the benefits outlined in the patent. The process begins with the preparation of the hydroxycarboxylic acid ester, which can be sourced from the oxidation of hexafluoroisobutenyl alkyl ethers. Once the precursor is secured, the decarboxylation step is initiated by mixing the ester with a catalytic amount of an amine hydrochloride or quaternary ammonium salt in a polar aprotic solvent. The detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below.
- Prepare the reaction mixture by combining the hydroxycarboxylic acid ester precursor with a selected amine, pyridine, or quaternary salt catalyst in an amide solvent.
- Heat the mixture to a temperature range of 5°C to 200°C, preferably around 120°C, to initiate the decarboxylation reaction under atmospheric pressure.
- Monitor the evolution of carbon dioxide gas and maintain reflux conditions until conversion is complete, followed by standard purification via distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this decarboxylation technology offers profound strategic advantages beyond mere technical performance. The ability to utilize industrial waste derivatives as feedstocks fundamentally reshapes the cost structure of fluoroalkyl alcohol production. By bypassing the need for fresh, high-purity hexafluoroisobutene gas, manufacturers can secure a more stable and predictable supply of raw materials, insulating the production process from volatile market fluctuations associated with primary fluorocarbon feedstocks. This stability is crucial for maintaining long-term contracts with downstream users in the semiconductor and pharmaceutical industries who demand uninterrupted supply continuity.
- Cost Reduction in Manufacturing: The elimination of toxic gas handling infrastructure and the use of waste-derived precursors lead to substantial capital and operational expenditure savings. The process operates under atmospheric pressure, removing the need for high-pressure reactors and the associated maintenance and safety certification costs. Furthermore, the high selectivity reduces the volume of waste solvents and by-products that require disposal, contributing to a leaner and more economically efficient production model without compromising on output quality.
- Enhanced Supply Chain Reliability: Sourcing raw materials from established industrial waste streams ensures a consistent feedstock supply that is less susceptible to the bottlenecks often seen in specialty chemical markets. The robustness of the reaction conditions, which tolerate a wide range of temperatures and pressures, means that production can be maintained even if minor utility fluctuations occur. This resilience is vital for reducing lead time for high-purity fluoroalkyl alcohols, ensuring that customers receive their orders on schedule regardless of external logistical challenges.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, requiring only standard glass-lined or metal reactors, facilitates easy scale-up from pilot batches to multi-ton commercial production. Additionally, the process aligns with increasingly stringent environmental regulations by converting hazardous waste precursors into valuable products, thereby minimizing the overall environmental footprint. This compliance advantage simplifies permitting processes and enhances the corporate sustainability profile, which is becoming a key differentiator in global B2B procurement decisions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this decarboxylation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for evaluating the process viability. Understanding these details is critical for stakeholders assessing the risk and reward profile of adopting this new synthetic route.
Q: What are the primary advantages of this decarboxylation method over traditional hexafluoroisobutene routes?
A: This method utilizes stable hydroxycarboxylic acid esters derived from industrial waste octafluoroisobutene, avoiding the direct handling of highly toxic hexafluoroisobutene gas and achieving significantly higher selectivity rates compared to conventional protic solvent methods.
Q: Which catalysts are most effective for maximizing selectivity in this process?
A: Experimental data indicates that amine hydrochlorides, such as diethylamine hydrochloride or pyridine hydrochloride, as well as quaternary ammonium salts like tetra-n-butylammonium chloride, provide superior selectivity and conversion rates when used in amide solvents.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the reaction operates under atmospheric pressure and moderate temperatures (5°C to 200°C) using inexpensive and readily available raw materials, making it highly scalable and economically viable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexafluoroisopropanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the decarboxylation technology described in CN101679156A for the production of high-value fluorinated intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients benefit from both laboratory-grade precision and industrial-scale efficiency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of fluoroalkyl alcohol meets the exacting standards required for pharmaceutical and electronic applications.
We invite you to collaborate with us to leverage this advanced chemistry for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your bill of materials. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of critical fluorine materials.
