Optimizing Iloperidone Manufacturing: A Strategic Analysis of Patent CN102212063A for Commercial Scale-Up
The global demand for atypical antipsychotics continues to rise, driven by the increasing prevalence of schizophrenia and the need for medications with improved side-effect profiles. Within this critical therapeutic area, Iloperidone (marketed as Zomaril) stands out as a potent serotonin-dopamine antagonist. However, the commercial viability of such complex molecules often hinges on the efficiency of their synthetic routes. Patent CN102212063A, filed in 2011, introduces a transformative methodology for preparing Iloperidone that addresses longstanding challenges in impurity management and yield optimization. By fundamentally altering the sequence of bond formation, this intellectual property offers a robust pathway that is highly attractive for reliable pharmaceutical intermediate supplier networks seeking to enhance their portfolio. The technical breakthrough lies not merely in a new reagent, but in a strategic reordering of the synthetic logic that minimizes downstream purification burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iloperidone has relied on routes that prioritize the functionalization of the phenolic component early in the sequence. As illustrated in the prior art referenced by the patent, traditional methods often involve reacting 4-hydroxy-3-methoxyacetophenone directly with dihaloalkanes. While chemically intuitive, this approach suffers from significant drawbacks regarding selectivity. The phenolic oxygen and the ketone functionality can lead to competing side reactions, generating structural analogs that are chemically similar to the target molecule. These byproducts are notoriously difficult to separate using standard crystallization techniques, often requiring expensive and time-consuming chromatographic purification. This complexity not only drives up the cost of goods sold but also introduces variability in the final drug substance quality, posing risks for cost reduction in pharmaceutical intermediates manufacturing.
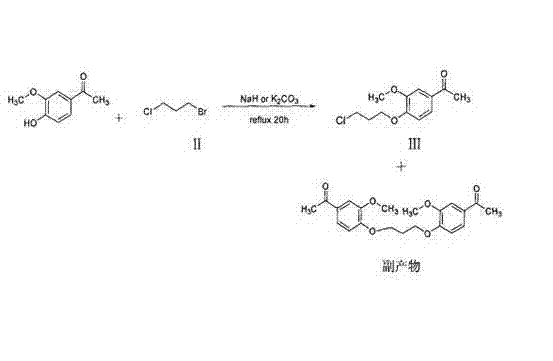
The Novel Approach
In stark contrast, the methodology disclosed in CN102212063A employs a "reverse alkylation" strategy that prioritizes the modification of the benzisoxazole-piperidine core. By first reacting 6-fluoro-3-(4-piperidyl)-1,2-benzisoxazole monohydrochloride with 1-bromo-3-chloropropane, the synthesis establishes the crucial propyl linker on the more nucleophilic piperidine nitrogen. This intermediate, 3-[1-(3-chloropropyl)piperidin-4-yl]-6-fluoro-1,2-benzisoxazole, is then coupled with the phenolic component in a subsequent step. This sequence ensures that the final etherification occurs under controlled conditions where the primary impurity profile is much cleaner. The patent data indicates that byproducts generated in this revised sequence are structurally distinct enough to be easily removed during the workup, thereby securing high-purity pharmaceutical intermediates without the need for exotic purification technologies.
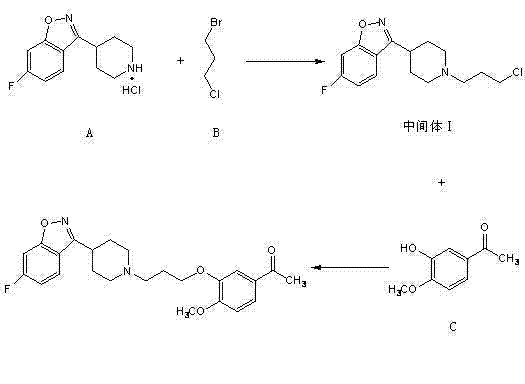
Mechanistic Insights into Nucleophilic Substitution and Etherification
The success of this synthetic route relies heavily on the precise control of nucleophilic substitution reactions (SN2). In the first step, the secondary amine of the piperidine ring acts as a potent nucleophile, attacking the terminal bromine of the 1-bromo-3-chloropropane. The choice of base is critical here; the patent explores a wide range including sodium hydroxide, potassium hydroxide, and organic bases like triethylamine. The use of polar aprotic solvents such as acetonitrile or DMF facilitates this displacement by stabilizing the transition state without solvating the nucleophile too strongly. Maintaining temperatures between 5-10°C during this addition is a key process parameter that suppresses potential elimination reactions or over-alkylation, ensuring the integrity of the chloropropyl tail which is essential for the next coupling step.
The second mechanistic phase involves the formation of the aryl alkyl ether. Here, the phenolic hydroxyl group of 4-hydroxy-3-methoxyacetophenone must displace the terminal chloride of the intermediate. Since chlorides are poorer leaving groups than bromides, the patent wisely incorporates catalytic amounts of potassium iodide or sodium iodide. This invokes a Finkelstein-type mechanism in situ, where the chloride is exchanged for a more reactive iodide, accelerating the etherification rate significantly. The reaction is conducted at moderate temperatures (40-60°C) in solvents like isopropanol or dioxane. This careful balancing of reactivity prevents the degradation of the sensitive benzisoxazole ring while driving the reaction to completion, a vital consideration for the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Iloperidone Efficiently
Implementing this patented process requires strict adherence to the specified stoichiometry and thermal profiles to maximize the benefits of the reverse alkylation strategy. The procedure begins with the preparation of the chloropropyl intermediate under basic conditions, followed by isolation and drying. The subsequent coupling with the acetophenone derivative utilizes iodide catalysis to ensure high conversion. Finally, the crude product undergoes a simplified purification regimen involving hot filtration and activated carbon treatment. For detailed operational parameters, safety data, and exact stoichiometric ratios required for GMP manufacturing, please refer to the standardized protocol below.
- React 6-fluoro-3-(4-piperidyl)-1,2-benzoisoxazole hydrochloride with 1-bromo-3-chloropropane in the presence of a base to form the chloropropyl intermediate.
- Condense the resulting intermediate with 4-hydroxy-3-methoxyacetophenone using an alkali metal carbonate and iodide catalyst.
- Purify the crude product via recrystallization and activated carbon treatment to achieve pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthesis route offers tangible benefits that extend beyond mere chemical yield. The ability to generate high-purity material through simple crystallization and carbon treatment drastically reduces the dependency on specialized purification resins or preparative HPLC, which are often bottlenecks in large-scale production. This simplification translates directly into reduced processing time and lower solvent consumption, addressing the critical need for reducing lead time for high-purity pharmaceutical intermediates. Furthermore, the robustness of the reaction conditions allows for flexibility in raw material sourcing, as the process tolerates a variety of common bases and solvents, mitigating supply risk.
- Cost Reduction in Manufacturing: The elimination of difficult-to-remove byproducts means that the purification stage is significantly streamlined. By avoiding complex chromatographic separations, manufacturers can achieve substantial cost savings in both consumables and labor. The use of commodity chemicals like potassium carbonate and isopropanol further ensures that the raw material costs remain stable and predictable, enhancing the overall economic viability of the production line.
- Enhanced Supply Chain Reliability: The synthetic route utilizes widely available starting materials and avoids exotic reagents that might be subject to supply constraints. The high yields reported in the patent embodiments (often exceeding 85% for the intermediate and nearly 90% for the final purification) ensure that material throughput is maximized. This efficiency supports a more reliable supply schedule, allowing procurement teams to plan inventory levels with greater confidence and reduce safety stock requirements.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents that are manageable in large reactor systems. The purification method, which relies on activated carbon decolorization, generates less hazardous waste compared to silica-based chromatography. This aligns with modern environmental, health, and safety (EHS) standards, facilitating easier regulatory approval and reducing the environmental footprint associated with the commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. They are derived from the specific experimental data and claims found within the patent documentation, providing clarity on reaction conditions and purification strategies. Understanding these nuances is essential for technology transfer teams evaluating the feasibility of adopting this route for commercial production.
Q: Why is the reaction order reversed in this patent compared to conventional methods?
A: Conventional methods react the phenol first, generating stubborn byproducts. This patent alkylates the piperidine ring first, ensuring byproducts are easily removed during purification, significantly boosting final purity.
Q: What specific purification advantages does this method offer?
A: The process utilizes simple recrystallization and activated carbon decolorization, avoiding complex chromatographic separations and reducing solvent consumption and processing time.
Q: Can this process be scaled for industrial production?
A: Yes, the patent demonstrates scalability from gram to kilogram levels using common solvents like acetonitrile and isopropanol, with yields consistently exceeding 85% for intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iloperidone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and rigorous quality control. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN102212063A are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Iloperidone intermediate meets the exacting standards required by global regulatory bodies. Our commitment to process excellence ensures that our clients receive materials that are not only chemically pure but also consistent in their physical properties.
We invite you to discuss how our advanced manufacturing capabilities can support your supply chain goals. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest level of technical and commercial confidence.
