Advanced Synthesis of Halogen-Substituted Phenylenedimethanols for Global Pharmaceutical Manufacturing
Advanced Synthesis of Halogen-Substituted Phenylenedimethanols for Global Pharmaceutical Manufacturing
The pharmaceutical and agrochemical industries constantly demand high-purity intermediates that can be produced reliably at scale. A significant breakthrough in this domain is detailed in patent CN101370759A, which discloses a highly efficient process for the preparation of halogen-substituted phenylenedimethanols. These compounds, represented by Formula (2), serve as critical building blocks for various active ingredients, including household insecticides and specialized pharmaceutical agents. The core innovation lies in a streamlined reduction strategy that transforms halogen-substituted terephthalic acids into their corresponding dimethanol derivatives with exceptional yield and purity. By utilizing a boron hydride compound in an organic solvent followed by a precise acidification step, this method overcomes the limitations of traditional synthetic routes. The technical robustness of this approach ensures that manufacturers can achieve consistent quality, making it a vital asset for any reliable agrochemical intermediate supplier seeking to optimize their production lines.
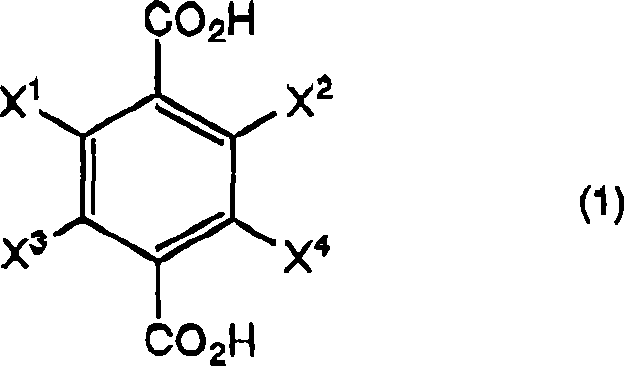
The structural versatility of this synthesis allows for a wide range of substituents, where X1, X2, X3, and X4 can independently represent hydrogen or halogen atoms, provided they are not all hydrogen simultaneously. This flexibility is crucial for generating diverse libraries of fluorinated or chlorinated intermediates required in modern drug discovery. For instance, the patent specifically highlights the successful synthesis of 2,3,5,6-tetrafluorobenzenedimethanol, a key precursor in the agrochemical sector. The ability to handle such highly halogenated substrates without dehalogenation or side reactions demonstrates the mild yet effective nature of the catalytic system employed. This level of chemical precision is what distinguishes a high-purity OLED material or pharmaceutical intermediate manufacturer from standard commodity chemical producers, ensuring that the final product meets the stringent specifications required for downstream biological applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of halogen-substituted benzenedimethanols was often plagued by inefficiencies and complex workup procedures. For example, US6759558 describes a method involving the reaction of tetrafluoroterephthalic acid with sodium borohydride, followed by treatment with alkylating agents, sulfuric acid, or sulfonic acids. These conventional pathways frequently suffer from poor atom economy and the generation of hazardous waste streams due to the use of strong mineral acids and alkylating reagents. Furthermore, comparative data within the field indicates that performing the acidification step at ambient temperatures, such as 25 to 30°C, leads to incomplete conversion. In such suboptimal conditions, significant amounts of starting material remain unreacted, and undesirable byproducts like 4-carboxy-2,3,5,6-tetrafluorobenzyl alcohol are formed in substantial quantities. This not only complicates the purification process but also drastically reduces the overall economic viability of the manufacturing route, creating bottlenecks for cost reduction in electronic chemical manufacturing and pharma sectors alike.
The Novel Approach
The methodology presented in CN101370759A fundamentally shifts the paradigm by introducing a controlled thermal activation during the acidification phase. Instead of relying on harsh alkylating agents or low-temperature acid treatments, the novel approach dictates contacting the reaction mixture with hydrogen chloride at a specific temperature range of 40 to 70°C. This seemingly simple adjustment has profound effects on the reaction kinetics and thermodynamics. Experimental results demonstrate that maintaining the temperature at 60°C during the addition of hydrochloric acid drives the reaction to near-completion, achieving yields as high as 95% with purities exceeding 95.1%. This stands in stark contrast to the 33% yield observed when the same reaction is conducted at 25 to 30°C. By eliminating the need for complex post-reduction modifications and optimizing the hydrolysis conditions, this process offers a direct, one-pot style efficiency that significantly simplifies the operational workflow. For a commercial scale-up of complex polymer additives or fine chemicals, such simplification translates directly into reduced processing time and lower energy consumption per unit of product.
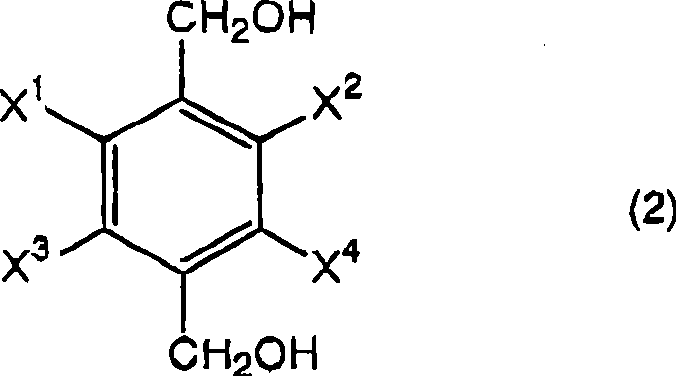
Mechanistic Insights into Borohydride-Catalyzed Reduction and Acidification
The core of this synthetic route relies on the selective reduction of carboxylic acid groups to primary alcohols using a borohydride species, typically sodium borohydride, in an ether solvent like dimethoxyethane. Mechanistically, the borohydride anion acts as a nucleophile, attacking the carbonyl carbon of the carboxylic acid. However, carboxylic acids are generally resistant to direct reduction by sodium borohydride under standard conditions due to the formation of unreactive carboxylate salts. The process described likely involves the formation of an acyloxyborohydride intermediate or utilizes the specific solvent environment to facilitate the reduction of the diacid to the dialkoxide species. The presence of halogen substituents, particularly fluorine, adds an layer of electronic complexity, as the electron-withdrawing nature of the halogens activates the aromatic ring and the carbonyl groups towards nucleophilic attack, potentially enhancing the reduction rate compared to unsubstituted terephthalic acid. This electronic activation is a key factor that allows the reaction to proceed efficiently at moderate temperatures ranging from 0 to 100°C, avoiding the extreme conditions often required for non-activated substrates.
Following the reduction, the critical step involves the protonation of the intermediate alkoxides to release the free alcohol. The patent specifies contacting the mixture with hydrogen chloride at 40 to 70°C. This thermal energy is likely required to overcome the activation barrier for the hydrolysis of stable boron-oxygen bonds formed during the reduction. At lower temperatures, these bonds may remain intact or hydrolyze too slowly, leading to the isolation of mono-reduced species or boron-containing impurities. The elevated temperature ensures rapid and complete cleavage of the B-O bonds, releasing the target halogen-substituted phenylenedimethanol. Furthermore, the use of concentrated hydrochloric acid or HCl gas provides a high concentration of protons, driving the equilibrium towards the product side. This mechanistic understanding underscores the importance of strict temperature control; deviating below 40°C risks incomplete reaction and impurity formation, while exceeding 70°C might pose unnecessary safety risks or promote decomposition, although the process remains robust within the specified window.
How to Synthesize 2,3,5,6-Tetrafluorobenzenedimethanol Efficiently
To implement this high-yielding synthesis in a laboratory or pilot plant setting, operators must adhere to precise stoichiometric and thermal parameters outlined in the patent examples. The process begins with the suspension of sodium borohydride in dimethoxyethane, followed by the controlled addition of the tetrafluoroterephthalic acid substrate. Maintaining the reaction temperature around 50 to 60°C during the addition and subsequent stirring period of approximately 7 hours is vital for complete conversion. Once the reduction is confirmed, typically via HPLC monitoring, the mixture is cooled slightly before the careful introduction of hydrochloric acid. It is imperative that this acidification step occurs within the 40 to 70°C window to maximize yield. Detailed standardized operating procedures regarding mixing rates, addition times, and quenching protocols are essential for reproducibility. For a comprehensive guide on the exact step-by-step execution, please refer to the technical documentation below.
- React halogen-substituted terephthalic acid with sodium borohydride in an ether solvent like dimethoxyethane at 0 to 100°C.
- Contact the resulting reaction mixture with hydrogen chloride or hydrochloric acid specifically at a temperature range of 40 to 70°C.
- Isolate the final halogen-substituted benzenedimethanol product through extraction, washing, and recrystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented process offers substantial strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the raw material portfolio. By replacing expensive and hazardous alkylating agents and sulfonic acids with commodity chemicals like sodium borohydride and hydrochloric acid, manufacturers can significantly reduce their raw material costs and mitigate supply chain risks associated with specialty reagents. Sodium borohydride is widely available globally, ensuring a stable supply base, while hydrochloric acid is a ubiquitous industrial chemical. This shift not only lowers the direct cost of goods sold but also simplifies inventory management and regulatory compliance, as fewer controlled or hazardous substances need to be stored and handled on-site. For a reliable agrochemical intermediate supplier, this translates into a more resilient and cost-effective operation capable of weathering market fluctuations in specialty chemical pricing.
- Cost Reduction in Manufacturing: The elimination of multi-step workups and expensive reagents directly impacts the bottom line. Traditional methods often require neutralization of large volumes of sulfuric acid or disposal of alkylating byproducts, which incurs significant waste treatment costs. In contrast, the novel process generates simpler waste streams primarily consisting of boron salts and aqueous washes, which are easier and cheaper to treat. The high yield of 95% means that less starting material is wasted, maximizing the value extracted from every kilogram of tetrafluoroterephthalic acid purchased. This efficiency gain allows for competitive pricing strategies in the global market, making the final intermediate more attractive to cost-sensitive buyers in the generic pharmaceutical and crop protection sectors without compromising on quality standards.
- Enhanced Supply Chain Reliability: Dependence on niche reagents can create single points of failure in a supply chain. By utilizing universally available reagents, the risk of production stoppages due to raw material shortages is minimized. The robustness of the reaction conditions, which tolerate a range of solvent ratios and do not require cryogenic cooling or high-pressure equipment, further enhances operational reliability. This stability is crucial for long-term contracts with multinational corporations that demand consistent delivery schedules. The ability to produce high-purity intermediates consistently ensures that downstream customers do not face delays in their own API synthesis, fostering stronger, trust-based partnerships between the chemical manufacturer and the end-user.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory experiments to ton-scale commercial production. The use of dimethoxyethane and toluene, while requiring standard solvent recovery systems, is well-understood in the industry. More importantly, the reduction in hazardous waste generation aligns with increasingly stringent environmental regulations worldwide. Avoiding the use of heavy metal catalysts or persistent organic pollutants simplifies the permitting process for new manufacturing lines. This environmental stewardship is not just a regulatory requirement but a market differentiator, appealing to green chemistry initiatives and sustainability goals of major pharmaceutical clients who prioritize eco-friendly supply chains in their vendor selection criteria.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is critical for stakeholders evaluating its implementation. The following questions address common concerns regarding process safety, scalability, and product quality. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making. Whether you are a process chemist optimizing a route or a procurement officer assessing vendor capabilities, clarity on these points ensures alignment on project expectations and deliverables.
Q: Why is the temperature control critical in the acidification step?
A: Maintaining the temperature between 40 to 70°C during HCl contact is essential to prevent the formation of mono-alcohol byproducts and ensure complete conversion, boosting yield from 33% to over 95%.
Q: What are the advantages of using sodium borohydride over other reducing agents?
A: Sodium borohydride offers a safer, more cost-effective profile compared to lithium aluminum hydride, while the novel process eliminates the need for complex alkylating agents used in prior art.
Q: Can this process be scaled for industrial production?
A: Yes, the use of common solvents like dimethoxyethane and standard reagents like hydrochloric acid makes the process highly scalable and suitable for commercial manufacturing without specialized equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrafluorobenzenedimethanol Supplier
The technical potential of the borohydride reduction pathway described in CN101370759A represents a significant opportunity for optimizing the production of halogenated intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facility is equipped with state-of-the-art reactors capable of handling exothermic reductions and corrosive acidifications safely and efficiently. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2,3,5,6-tetrafluorobenzenedimethanol meets the exacting standards of the global pharmaceutical industry. Our commitment to quality assurance means that we can deliver intermediates with consistent impurity profiles, facilitating smoother regulatory filings for our clients' final drug products.
We invite potential partners to engage with our technical team to explore how this optimized synthesis can benefit your specific supply chain. By leveraging our expertise, you can achieve a Customized Cost-Saving Analysis tailored to your volume requirements and logistical needs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your projects. Together, we can drive innovation and efficiency in the production of high-value fine chemical intermediates, ensuring a secure and sustainable supply for the future of medicine and agriculture.
