Advanced Bromine-Free Synthesis of Naphthalene Derivatives for Commercial API Production
Advanced Bromine-Free Synthesis of Naphthalene Derivatives for Commercial API Production
The pharmaceutical industry constantly seeks more efficient and safer pathways for synthesizing complex active pharmaceutical ingredients (APIs) and their precursors. Patent CN1043932A introduces a groundbreaking methodology for the preparation of specific naphthalene derivatives that serve as potent hypolipidemic agents. This technology represents a significant departure from traditional synthetic routes by eliminating the need for hazardous halogenation steps, thereby addressing critical safety and environmental concerns inherent in large-scale chemical manufacturing. By utilizing a direct organolithium-mediated coupling strategy followed by an acid-catalyzed cyclization, this process offers a streamlined approach to constructing the substituted naphthalene core. For global procurement teams and R&D directors, understanding the nuances of this bromine-free pathway is essential for evaluating potential suppliers who can deliver high-purity pharmaceutical intermediates with improved cost structures and supply chain reliability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in U.S. Patent No. 4,771,072, rely heavily on the use of 2-bromo-3,4,5-tris(lower alkoxy)benzaldehyde dialkyl acetals as key starting materials. The synthesis of these brominated precursors necessitates a dedicated bromination step involving elemental bromine, a substance known for its high toxicity, corrosivity, and difficulty in handling within an industrial setting. This reliance on halogen chemistry introduces substantial operational risks, requiring specialized corrosion-resistant equipment and rigorous safety protocols to manage potential leaks or exposure. Furthermore, the generation of brominated waste streams creates a significant burden on environmental compliance departments, increasing the overall cost of goods sold through expensive waste treatment and disposal procedures. The multi-step nature of preparing the brominated acetal also extends the total production timeline, creating bottlenecks that can impact the continuity of supply for downstream API manufacturing processes.
The Novel Approach
In stark contrast, the methodology described in CN1043932A achieves the construction of the target naphthalene framework through a more direct and atom-economical route that completely circumvents the use of bromine. By reacting a non-brominated 3,4,5-tris(lower alkoxy)benzaldehyde dialkyl acetal directly with a 3,4-di(lower alkoxy)benzaldehyde, the process forms the critical carbon-carbon bond in a single addition step. This strategic simplification reduces the total number of unit operations required, effectively shortening the manufacturing cycle time and minimizing the accumulation of intermediate impurities. The elimination of the bromination step not only enhances operator safety but also simplifies the reactor train requirements, allowing for the use of standard stainless steel equipment rather than exotic alloys. This technological shift provides a robust foundation for commercial scale-up, offering a reliable pharmaceutical intermediates supplier the ability to produce materials with greater consistency and lower operational overhead.
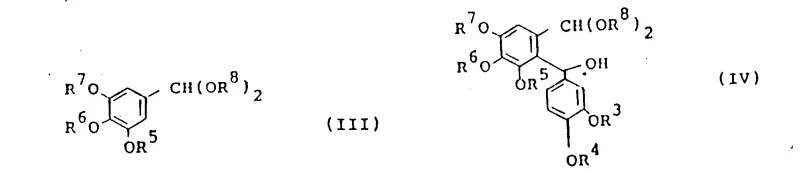
Mechanistic Insights into Organolithium-Mediated Coupling and Cyclization
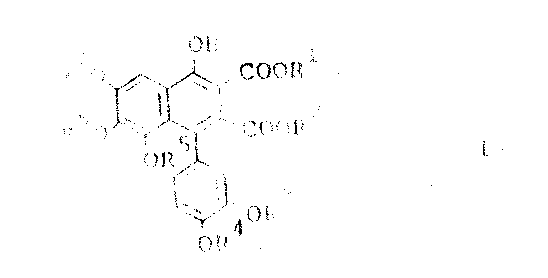
The core of this innovative synthesis lies in the precise execution of an organolithium-mediated nucleophilic addition. In the first stage, a tris(lower alkoxy)benzaldehyde dialkyl acetal is treated with a strong base, typically an organolithium compound such as n-butyllithium, phenyllithium, or lithium diisopropylamide. This deprotonation generates a reactive carbanion species at the position ortho to the acetal group, which then attacks the carbonyl carbon of the 3,4-di(lower alkoxy)benzaldehyde. The reaction conditions are critically controlled, with temperatures maintained preferably between -20 and 20°C, although the process remains viable within a broader range of -70 to 40°C. This thermal regulation is vital for suppressing competing side reactions, such as self-condensation of the aldehyde or decomposition of the organometallic species, ensuring that the formation of the 2-[3,4-di(lower alkoxy)-alpha-hydroxybenzyl] intermediate proceeds with high chemoselectivity. The choice of solvent, ranging from tetrahydrofuran to hydrocarbons like hexane or toluene, further modulates the reactivity of the lithiated species, allowing fine-tuning of the reaction kinetics for optimal yield.
Following the formation of the alpha-hydroxybenzyl intermediate, the synthesis proceeds to a sophisticated acid-catalyzed cyclization step that constructs the naphthalene ring system. The intermediate is reacted with a dialkyl acetylenedicarboxylate in the presence of a catalytic amount of acid, such as p-toluenesulfonic acid, methanesulfonic acid, or mineral acids like hydrochloric acid. This transformation likely proceeds through a mechanism involving the activation of the hydroxyl group or the acetal functionality, facilitating an intramolecular electrophilic aromatic substitution or a similar cyclization pathway that closes the ring. The reaction is remarkably robust, capable of proceeding at temperatures between 0 and 150°C, with a preferred range of 50 to 100°C to balance reaction rate and product stability. This step effectively installs the ester functionalities at the 2 and 3 positions of the naphthalene ring while establishing the aromatic character of the core structure. The ability to perform this cyclization with or without additional solvent adds further flexibility to the process, enabling concentration adjustments that can drive equilibrium towards the desired high-purity naphthalene derivative.
How to Synthesize Naphthalene Derivatives Efficiently
The practical implementation of this synthesis route requires careful attention to reagent stoichiometry and thermal management to maximize throughput and purity. The process begins with the generation of the lithiated acetal species under inert atmosphere conditions to prevent quenching by moisture or oxygen, followed by the controlled addition of the second aldehyde component. Once the intermediate is isolated or telescoped, the subsequent cyclization with the acetylenedicarboxylate serves as the key ring-forming event that defines the molecular architecture. Detailed standardized operating procedures regarding quenching, workup, and crystallization are essential to ensure that the final product meets the stringent specifications required for pharmaceutical applications. For a comprehensive breakdown of the specific experimental parameters, reagent grades, and isolation techniques validated in the patent examples, please refer to the technical guide below.
- React bis(lower alkoxy)benzaldehyde with tris(lower alkoxy)benzaldehyde dialkyl acetal using an organolithium reagent like n-butyllithium at -70 to 40°C to form the alpha-hydroxybenzyl intermediate.
- Subject the resulting intermediate to reaction with dialkyl acetylenedicarboxylate in the presence of an acid catalyst at 0-150°C to effect cyclization into the naphthalene core.
- Optionally convert the final naphthalene derivative into its pharmaceutically acceptable salt using bases such as sodium hydroxide or sodium hydride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this bromine-free synthesis route translates into tangible strategic advantages regarding cost structure and risk mitigation. By removing the requirement for elemental bromine and the associated bromination infrastructure, manufacturers can significantly reduce capital expenditure on specialized corrosion-resistant reactors and scrubbing systems. This simplification of the plant footprint allows for more flexible production scheduling and the potential to utilize existing multipurpose facilities without extensive retrofitting. Furthermore, the reduction in the number of synthetic steps directly correlates with lower consumption of utilities, solvents, and labor hours, driving down the variable cost per kilogram of the intermediate. These efficiencies enable a reliable pharmaceutical intermediates supplier to offer more competitive pricing models while maintaining healthy margins, ultimately supporting cost reduction in API manufacturing for the end client.
- Cost Reduction in Manufacturing: The elimination of the bromination step removes the need for expensive halogen handling protocols and the subsequent neutralization of acidic bromine waste, which are major cost drivers in traditional fine chemical production. Additionally, the shortened synthetic sequence reduces the cumulative loss of material at each isolation stage, leading to improved overall mass balance and higher effective yield from raw materials. The use of readily available starting materials, such as simple alkoxybenzaldehydes and acetylene dicarboxylates, ensures that raw material costs remain stable and predictable, avoiding the volatility often associated with specialized halogenated building blocks. This streamlined approach allows for substantial cost savings that can be passed down the supply chain, enhancing the economic viability of the final hypolipidemic therapeutic.
- Enhanced Supply Chain Reliability: Dependence on hazardous reagents like bromine often introduces supply chain fragility due to strict transportation regulations and limited supplier availability. By shifting to a chemistry based on common organolithium reagents and standard aldehydes, the production process becomes more resilient to external disruptions and regulatory changes. The robustness of the reaction conditions, which tolerate a range of solvents and temperatures, further ensures consistent batch-to-batch performance even when scaling from pilot plants to commercial tonnage. This stability is crucial for securing long-term supply agreements and reducing lead time for high-purity pharmaceutical intermediates, ensuring that downstream API production schedules are met without interruption.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, the absence of bromine significantly lowers the toxicological profile of the manufacturing process, simplifying permitting and compliance reporting. The waste streams generated are primarily organic in nature and easier to treat compared to halogenated effluents, reducing the burden on wastewater treatment facilities. The process demonstrates excellent scalability, as evidenced by the successful execution of reactions on multi-gram scales in the patent examples, suggesting a clear path to commercial scale-up of complex pharmaceutical intermediates. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturing partner.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this naphthalene derivative synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction scope and operational parameters. Understanding these details is vital for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the elimination of bromine significant in this synthesis route?
A: Conventional methods require the preparation of 2-bromo-3,4,5-tris(lower alkoxy)benzaldehyde acetals, necessitating the use of elemental bromine which is hazardous, corrosive, and difficult to handle on an industrial scale. This novel process bypasses the bromination step entirely, significantly improving operational safety and reducing environmental waste treatment costs associated with halogenated byproducts.
Q: What are the critical temperature controls for the organolithium reaction step?
A: The reaction between the aldehyde and the acetal in the presence of organolithium compounds is highly exothermic and sensitive. The patent specifies a preferred temperature range of -20 to 20°C, although the broader operable range is -70 to 40°C. Maintaining strict temperature control within this window is essential to prevent side reactions and ensure high selectivity for the desired alpha-hydroxybenzyl intermediate.
Q: Which solvents are compatible with the cyclization step?
A: The cyclization reaction involving the intermediate and acetylene dicarboxylate can be performed in various organic solvents including toluene, benzene, or tetrahydrofuran. The process is flexible enough to proceed even without a solvent if desired, provided an acid catalyst such as p-toluenesulfonic acid or methanesulfonic acid is present to drive the formation of the naphthalene ring system.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Naphthalene Derivative Supplier
The technological advancements detailed in CN1043932A highlight the potential for producing high-value hypolipidemic intermediates with superior efficiency and safety. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such innovative laboratory methodologies into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and compliant with international quality standards. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of naphthalene derivative meets the exacting requirements of global pharmaceutical clients.
We invite you to collaborate with us to leverage this bromine-free technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data for our available intermediates and to discuss route feasibility assessments that can optimize your supply chain for the future.
