Advanced Synthesis of Iclaprim p-Toluenesulfonate for Commercial Antibiotic Production
Introduction to Advanced Iclaprim Intermediate Manufacturing
The pharmaceutical landscape for antibacterial agents continues to evolve, driven by the urgent need for effective treatments against resistant bacterial strains. Iclaprim, a potent dihydrofolate reductase inhibitor, represents a critical asset in this therapeutic area, specifically indicated for acute bacterial skin and skin structure infections. However, the commercial viability of such advanced therapeutics is often bottlenecked by the complexity and inefficiency of their synthetic routes. Patent CN110790753B discloses a groundbreaking preparation method for elaprine p-toluenesulfonate, a key intermediate, which addresses these historical manufacturing challenges. This technical insight report analyzes the novel elimination reaction strategy that transforms the chroman compound of Formula 6 directly into the target p-toluenesulfonate salt. By leveraging specific aprotic solvent systems and p-toluenesulfonic acid catalysis, this method achieves exceptional purity levels exceeding 99.9 percent without the need for labor-intensive column chromatography. For R&D directors and supply chain leaders, understanding this shift from multi-step, low-yield processes to a streamlined, high-efficiency route is paramount for securing a reliable supply of high-purity pharmaceutical intermediates.
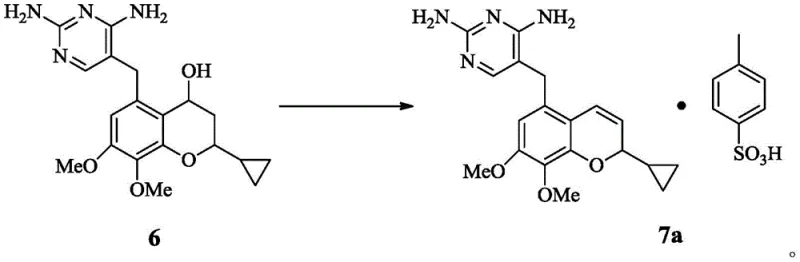
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Iclaprim and its intermediates has been plagued by inefficient reaction sequences that hinder industrial scalability. As illustrated in prior art such as CN 101115743A, traditional routes often involve protecting group strategies followed by Friedel-Crafts acetylation and selective demethylation. These pathways are notoriously lengthy, requiring multiple isolation steps where the overall yield can plummet to as low as 4 percent. Furthermore, the purification of intermediates in these legacy processes frequently necessitates column chromatography, a technique that is economically prohibitive and operationally cumbersome at the metric ton scale. Another existing method, described in CN 1092194C, relies on expensive starting materials and catalysts, such as cerium(III) chloride heptahydrate for carbonyl reduction. Additionally, the use of Mitsunobu reactions introduces severe operational hazards and difficult post-treatment requirements due to the generation of stoichiometric amounts of phosphine oxide byproducts. These factors collectively result in high production costs, significant environmental waste, and inconsistent supply continuity, rendering such methods unsuitable for modern commercial manufacturing demands.
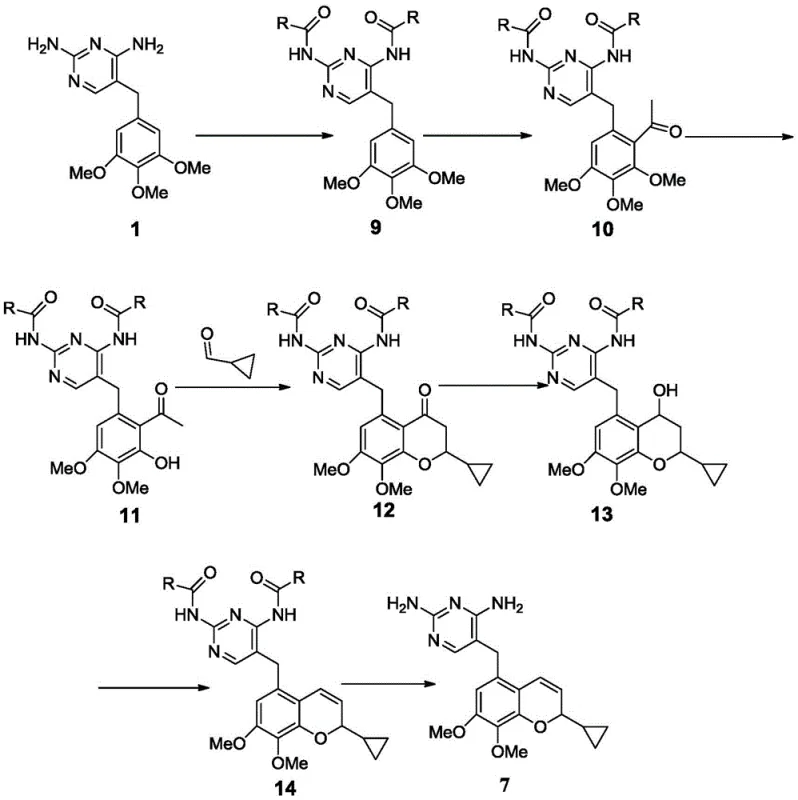
The Novel Approach
In stark contrast to these convoluted legacy pathways, the novel approach disclosed in the patent utilizes a direct acid-catalyzed elimination reaction to construct the critical benzopyran core. This method involves treating the chroman compound of Formula 6 with p-toluenesulfonic acid in an aprotic solvent environment. The elegance of this chemistry lies in its simplicity and robustness; it bypasses the need for protecting groups and avoids the use of expensive transition metal catalysts. The reaction proceeds efficiently at temperatures between 60 and 100 degrees Celsius, typically reaching completion within 0.5 to 2 hours. By forming the p-toluenesulfonate salt directly during the elimination step, the process inherently drives the equilibrium towards the product and simplifies isolation through precipitation. This strategic shift not only drastically shortens the synthetic route but also ensures that the final intermediate is obtained with purity levels suitable for subsequent API synthesis without further chromatographic purification. The ability to produce the target molecule in yields approaching 90 percent marks a significant technological leap forward for cost reduction in antibiotic intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Elimination and Cyclization
The core of this innovative synthesis lies in the precise control of the elimination reaction mechanism, which converts the saturated chroman ring into the unsaturated benzopyran system found in Iclaprim. The use of p-toluenesulfonic acid serves a dual purpose: it acts as a proton source to facilitate the dehydration of the hydroxyl group at the 4-position of the chroman ring, and the tosylate anion acts as a counterion to stabilize the resulting cationic intermediate or the final alkene product as a salt. The choice of solvent is mechanistically critical; aprotic polar solvents like dimethyl sulfoxide (DMSO) mixed with tetrahydrofuran (THF) enhance the solubility of the polar intermediates while stabilizing the transition state. Comparative examples in the patent data highlight that using protic solvents like methanol leads to unwanted side reactions, such as etherification, where the solvent attacks the reactive intermediate instead of elimination occurring. This mechanistic understanding allows for rigorous impurity control, ensuring that the final product profile is clean and dominated by the desired E-isomer of the benzopyran ring. Such control is essential for meeting the stringent regulatory requirements for pharmaceutical intermediates.
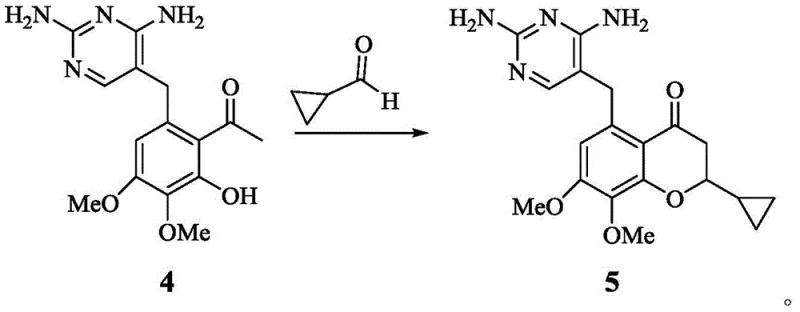
Furthermore, the construction of the precursor chromanone scaffold via aldol condensation and cyclization, as shown in the transformation of Formula 4 to Formula 5, demonstrates another layer of process optimization. This step utilizes cyclopropanecarboxaldehyde and a phenol derivative in the presence of a secondary amine base like pyrrolidine. The reaction conditions are mild, typically proceeding at room temperature, which minimizes thermal degradation of the sensitive diaminopyrimidine moiety. The subsequent reduction of the ketone to the alcohol (Formula 5 to Formula 6) using sodium borohydride is highly chemoselective, leaving the pyrimidine ring and methoxy groups intact. The integration of these steps into a cohesive linear sequence, culminating in the final elimination, showcases a deep understanding of functional group tolerance. By avoiding harsh Lewis acids like boron tribromide in the final steps and reserving them for earlier demethylation stages where they are manageable, the overall process safety and reliability are significantly enhanced. This holistic view of the reaction mechanism ensures that the process is not just theoretically sound but practically viable for large-scale operations.
How to Synthesize Iclaprim p-Toluenesulfonate Efficiently
The practical execution of this synthesis requires careful attention to solvent quality and reaction monitoring to ensure consistent results. The process begins with the dissolution of the chroman precursor in a pre-mixed solvent system, followed by the controlled addition of the acid catalyst. Maintaining the reaction temperature within the optimal window is crucial to prevent decomposition while ensuring complete conversion. Real-time monitoring via HPLC or TLC is recommended to determine the exact endpoint, preventing over-reaction which could lead to polymerization or degradation. Once the reaction is complete, the workup is remarkably straightforward, involving cooling to induce crystallization of the salt, followed by simple filtration. This operational simplicity is a key driver for its adoption in commercial settings, as it reduces the burden on equipment and personnel. For detailed standard operating procedures and specific parameter ranges, please refer to the standardized guide below.
- Prepare the reaction system by dissolving the chroman compound (Formula 6) in an aprotic solvent mixture such as tetrahydrofuran and dimethyl sulfoxide.
- Add p-toluenesulfonic acid monohydrate to the solution and heat the mixture to a temperature range of 60 to 100 degrees Celsius to initiate the elimination reaction.
- Monitor the reaction progress via HPLC until the starting material disappears, then cool the mixture to precipitate the product, followed by filtration and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits that extend beyond mere technical feasibility. The elimination of column chromatography is perhaps the most significant economic driver, as this unit operation is a major bottleneck in fine chemical manufacturing, consuming vast amounts of silica gel and organic solvents while limiting batch sizes. By replacing this with a crystallization-based purification, the process becomes inherently more scalable, allowing for the production of larger batches in standard reactor vessels without specialized packing columns. This directly translates to substantial cost savings in terms of both raw materials and waste disposal. Furthermore, the reagents employed, such as p-toluenesulfonic acid and common aprotic solvents, are commodity chemicals with stable global supply chains, reducing the risk of raw material shortages that often plague processes relying on exotic catalysts. The high yield and purity achieved in this route also mean that less starting material is required to produce the same amount of final product, optimizing the overall material balance and reducing the carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The removal of chromatographic purification steps significantly lowers the operational expenditure associated with solvent consumption and stationary phase materials. Additionally, the high yield of the elimination step minimizes the loss of valuable intermediates, leading to a more efficient use of capital-intensive starting materials. The simplified workup procedure reduces labor hours and energy consumption associated with solvent recovery, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: By utilizing widely available reagents and avoiding supply-constrained catalysts, the risk of production delays due to raw material scarcity is drastically mitigated. The robustness of the reaction conditions allows for flexible scheduling and easier technology transfer between different manufacturing sites, ensuring business continuity. The ability to produce high-purity intermediates consistently strengthens the supply chain resilience against market fluctuations and regulatory audits.
- Scalability and Environmental Compliance: The process is designed for industrial scale-up, moving away from batch-limited purification techniques to continuous or large-batch crystallization. This scalability is accompanied by a reduction in hazardous waste generation, particularly phosphine oxides and heavy metal residues found in alternative routes. The use of greener solvent systems and the potential for solvent recycling align with modern environmental, social, and governance (ESG) goals, facilitating smoother regulatory approvals and community relations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and product quality. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their own production pipelines. The data supports the conclusion that this method offers a superior balance of efficiency, cost, and quality compared to historical precedents.
Q: What are the primary advantages of this new synthesis route over conventional methods?
A: The novel route eliminates the need for column chromatography purification, which is required in older methods, significantly reducing processing time and solvent waste while improving overall yield from approximately 4 percent to over 89 percent.
Q: Which solvents are preferred for the elimination reaction step?
A: Aprotic solvents are essential for this transformation, with a mixed system of tetrahydrofuran and dimethyl sulfoxide in a volume ratio of approximately 3:1 being particularly effective for maximizing yield and purity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available reagents and simple workup procedures like filtration and crystallization, making it highly scalable for industrial production without the need for complex separation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iclaprim p-Toluenesulfonate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced antibiotics like Iclaprim depends on a partner who can deliver both technical excellence and supply security. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to full market launch. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Iclaprim p-toluenesulfonate meets the highest international standards. Our facility is equipped to handle the specific solvent systems and reaction conditions required for this elimination chemistry, providing a safe and compliant environment for manufacturing.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can support your long-term strategic goals in the pharmaceutical sector.
