Advanced Pd-Catalyzed Synthesis of Carbonyl-bridged Biheterocyclic Compounds for Commercial API Production
Advanced Pd-Catalyzed Synthesis of Carbonyl-bridged Biheterocyclic Compounds for Commercial API Production
The pharmaceutical industry continuously seeks efficient pathways to construct complex heterocyclic scaffolds that serve as core structures for bioactive molecules. Patent CN115353511A introduces a groundbreaking multicomponent method for synthesizing carbonyl-bridged biheterocyclic compounds, specifically targeting the fusion of indolinone and imidazole motifs. This technology addresses the significant synthetic challenges associated with installing carbonyl bridges between distinct heterocyclic systems, a transformation that is often plagued by harsh conditions or poor atom economy in conventional literature. By leveraging a palladium-catalyzed cascade reaction, this invention enables the one-pot assembly of these valuable architectures from readily available starting materials, marking a substantial advancement for reliable pharmaceutical intermediate supplier networks seeking to optimize their production pipelines.
The strategic value of this patent lies in its ability to bypass the limitations of stepwise synthesis, which typically requires the isolation of unstable intermediates and multiple purification steps. The described methodology integrates carbon-nitrogen bond formation, carbonylation, and cyclization into a single operational sequence. This consolidation not only accelerates the timeline for process development but also inherently reduces the accumulation of waste solvents and reagents. For research and development teams focused on generating diverse libraries of drug candidates, this approach offers a robust platform for accessing novel chemical space with high structural complexity, ensuring that the resulting compounds possess the necessary physicochemical properties for further biological evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of biheterocyclic systems containing a carbonyl bridge has relied heavily on the direct coupling of two pre-functionalized heterocyclic substrates or the oxidative cyclization of substrates bearing dual nucleophilic sites. These traditional pathways often suffer from significant drawbacks, including the requirement for stoichiometric amounts of expensive oxidants or the necessity of using toxic carbon monoxide gas under high pressure. Furthermore, direct coupling reactions frequently exhibit poor regioselectivity and limited substrate compatibility, particularly when sensitive functional groups are present on the heterocyclic rings. The reliance on gaseous CO introduces severe safety hazards and necessitates specialized reactor infrastructure, creating a bottleneck for cost reduction in pharmaceutical intermediate manufacturing where safety and simplicity are paramount.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a transition metal palladium-catalyzed carbonylation cascade that operates under remarkably mild conditions. By employing a mixture of formic acid and acetic anhydride as a safe, liquid carbon monoxide surrogate, the process eliminates the risks associated with handling toxic gases while maintaining high reaction efficiency. The reaction proceeds at a moderate temperature of 30°C in common organic solvents like tetrahydrofuran, demonstrating exceptional functional group tolerance. This innovation allows for the seamless integration of trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives into a unified synthetic flow, effectively constructing multiple chemical bonds in a single step.
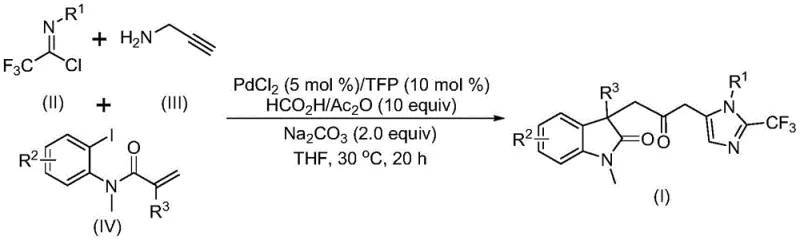
The versatility of this new route is further evidenced by its compatibility with a wide array of substituents, enabling the rapid generation of diversified analogues without the need for protecting group strategies. The use of cheap and commercially available starting materials ensures that the supply chain remains resilient and cost-effective. Moreover, the successful expansion of this reaction to the gram scale confirms its potential for industrial application, providing a scalable solution for the commercial scale-up of complex pharmaceutical intermediates. This represents a paradigm shift from hazardous, multi-step protocols to a streamlined, safe, and economically viable manufacturing process.
Mechanistic Insights into Palladium-Catalyzed Carbonylation Cascade
The mechanistic pathway of this transformation involves a sophisticated sequence of organometallic steps initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate. This is followed by an intramolecular Heck reaction that generates a divalent alkyl-palladium intermediate, setting the stage for the subsequent carbonylation event. The carbon monoxide, released in situ from the decomposition of the formic acid and acetic anhydride mixture, inserts into the palladium-carbon bond to form an acyl-palladium intermediate. This critical step installs the carbonyl bridge that defines the structural core of the final product, linking the indolinone and imidazole moieties with high precision.
Concurrently, the base-promoted reaction between trifluoroethylimidoyl chloride and propargylamine facilitates the formation of a trifluoroacetamidine compound, which subsequently undergoes isomerization. The acyl-palladium intermediate then activates this species, catalyzing an intramolecular cyclization that closes the imidazole ring and releases the final carbonyl-bridged biheterocyclic product. This intricate interplay of catalytic cycles ensures high selectivity and minimizes the formation of side products, which is crucial for maintaining a clean impurity profile. The mild reaction temperature of 30°C further suppresses thermal degradation pathways, ensuring that the integrity of sensitive functional groups is preserved throughout the synthesis.
Understanding this mechanism is vital for process optimization, as it highlights the importance of the ligand environment and the precise stoichiometry of the CO surrogate. The use of trifurylphosphine as a ligand stabilizes the palladium center, preventing premature catalyst deactivation and ensuring high turnover numbers. For quality control teams, recognizing the origin of potential impurities—such as unreacted starting materials or incomplete cyclization byproducts—allows for the design of targeted purification strategies. The robustness of this catalytic system against various electronic and steric variations in the substrates underscores its utility as a general platform for synthesizing high-purity OLED material precursors or pharmaceutical building blocks.
How to Synthesize Carbonyl-bridged Biheterocyclic Compounds Efficiently
The practical execution of this synthesis is designed to be straightforward, requiring standard laboratory equipment and avoiding exotic reagents. The protocol begins with the preparation of the catalytic system in an aprotic solvent, followed by the sequential addition of the three key components. The reaction is allowed to proceed for 12 to 20 hours, a timeframe that balances complete conversion with operational efficiency. Post-reaction processing involves simple filtration to remove inorganic salts, followed by silica gel treatment and column chromatography to isolate the pure product. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different manufacturing sites.
- Combine palladium chloride, trifurylphosphine ligand, sodium carbonate, and the formic acid/acetic anhydride mixture in an organic solvent such as THF.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the reaction mixture under inert atmosphere.
- Stir the reaction at 30°C for 12 to 20 hours, followed by filtration, silica gel treatment, and column chromatography purification to isolate the target biheterocycle.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this technology offers substantial benefits by relying on commodity chemicals that are widely available in the global market. The elimination of high-pressure carbon monoxide cylinders removes a significant logistical and safety burden, simplifying the regulatory compliance landscape for manufacturing facilities. The use of palladium chloride, a relatively inexpensive catalyst compared to specialized palladium complexes, contributes to a lower bill of materials. Furthermore, the high reaction efficiency and yield reported in the patent examples suggest that raw material consumption is optimized, leading to significant cost savings in pharmaceutical intermediate manufacturing through reduced waste and higher throughput.
- Cost Reduction in Manufacturing: The replacement of toxic gaseous carbon monoxide with a liquid formic acid/acetic anhydride mixture drastically simplifies the reactor requirements, removing the need for expensive high-pressure autoclaves and specialized gas handling systems. This capital expenditure saving is compounded by the operational savings derived from milder reaction conditions, which consume less energy for heating and cooling. Additionally, the high atom economy of the multicomponent reaction ensures that a greater proportion of the starting mass is converted into the desired product, minimizing the cost associated with waste disposal and raw material procurement.
- Enhanced Supply Chain Reliability: The starting materials, including trifluoroethylimidoyl chloride and propargylamine, are either commercially available or easily synthesized from bulk chemicals, reducing the risk of supply chain disruptions. The robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in temperature or reagent quality, ensuring consistent batch-to-batch performance. This reliability is critical for reducing lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond quickly to market demands without compromising on quality or delivery schedules.
- Scalability and Environmental Compliance: The successful demonstration of gram-scale synthesis indicates a clear path towards kilogram and ton-scale production, supported by the use of common solvents like THF which are easily recovered and recycled. The avoidance of toxic gases and the use of a heterogeneous workup (filtration) simplify the environmental management of the process, aligning with increasingly stringent green chemistry regulations. This scalability ensures that the technology can meet the growing demand for complex biheterocyclic compounds in the agrochemical and pharmaceutical sectors without encountering the bottlenecks typical of more hazardous synthetic routes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the operational parameters and scope of the technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios.
Q: What is the primary safety advantage of this carbonylation method?
A: Unlike traditional carbonylation reactions that require high-pressure cylinders of toxic carbon monoxide gas, this method utilizes a formic acid and acetic anhydride mixture to generate carbon monoxide in-situ, significantly enhancing operational safety and eliminating the need for specialized high-pressure equipment.
Q: What is the substrate scope for the R1 and R3 groups in this synthesis?
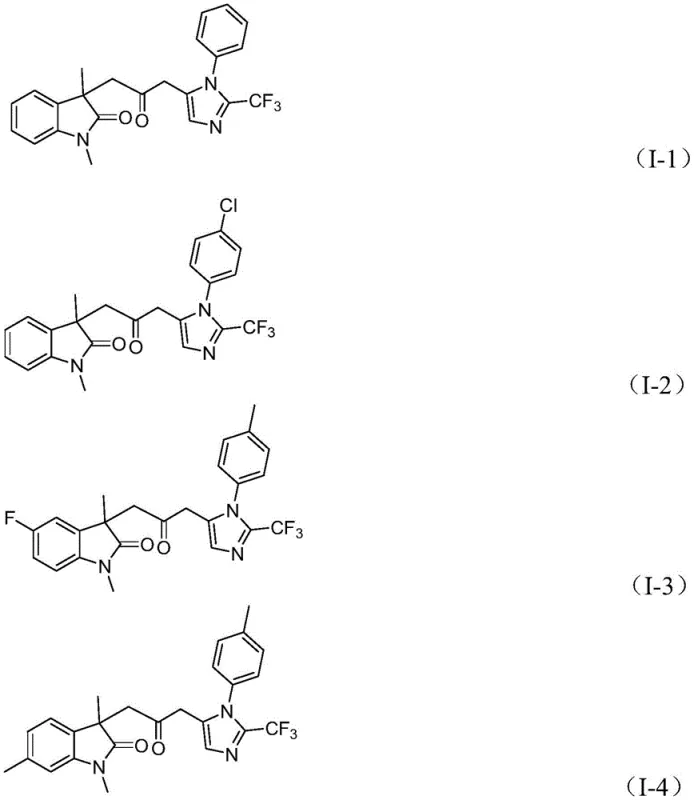
A: The methodology demonstrates excellent functional group tolerance, accommodating various substituents on the aryl rings including alkyl, alkoxy, halogen, trifluoromethyl, and nitro groups at ortho, meta, or para positions, allowing for the diverse synthesis of drug-like scaffolds.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent explicitly demonstrates the scalability of the reaction to the gram level with high efficiency. The use of cheap, commercially available starting materials and mild reaction conditions (30°C) supports feasible commercial scale-up for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-bridged Biheterocyclic Compounds Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like this palladium-catalyzed carbonylation can be seamlessly transferred to industrial manufacturing. Our commitment to quality is backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of carbonyl-bridged biheterocyclic compounds meets the exacting standards required for pharmaceutical applications. We understand the critical nature of supply continuity and are equipped to manage the complexities of multicomponent reactions to deliver consistent, high-quality intermediates to our global partners.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can accelerate your development timeline while optimizing your overall production costs.
