Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
Advanced Palladium-Catalyzed Synthesis of Carbonyl-Bridged Biheterocyclic Compounds for Commercial Scale-Up
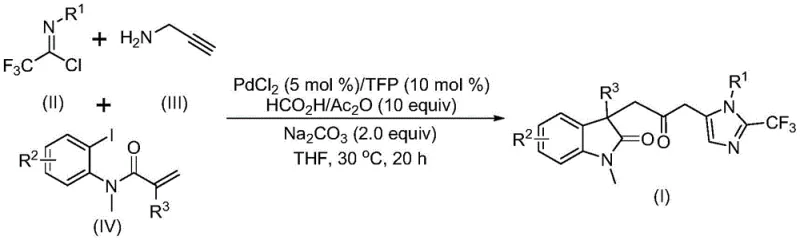
The landscape of organic synthesis for complex heterocyclic scaffolds is undergoing a significant transformation driven by the need for safer, more efficient, and scalable methodologies. A pivotal development in this domain is documented in patent CN115353511A, which discloses a novel multi-component method for synthesizing carbonyl-bridged biheterocyclic compounds. This technology represents a substantial leap forward for the production of high-purity pharmaceutical intermediates, addressing critical challenges associated with traditional carbonylation reactions. By leveraging a transition metal palladium-catalyzed cascade reaction, this approach eliminates the necessity for handling toxic carbon monoxide gas directly, instead utilizing a safe in-situ generation system. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages and commercial viability of this patent is essential for securing a competitive edge in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of carbonyl-bridged biheterocyclic frameworks has been fraught with significant technical and safety hurdles that impede efficient commercial scale-up of complex heterocycles. Traditional synthetic routes often rely on the direct coupling of two heterocyclic substrates or oxidative cyclization reactions that suffer from poor atom economy and limited substrate scope. More critically, conventional carbonylation strategies frequently mandate the use of pressurized carbon monoxide gas, which poses severe safety risks in a manufacturing environment and requires specialized, expensive high-pressure reactor equipment. Furthermore, these legacy methods often exhibit low reaction efficiency and struggle with functional group tolerance, leading to complex impurity profiles that necessitate costly and time-consuming purification processes. These factors collectively contribute to extended lead times and inflated production costs, creating bottlenecks for companies aiming for cost reduction in fine chemical manufacturing.
The Novel Approach
In stark contrast to these legacy limitations, the methodology outlined in CN115353511A introduces a streamlined, one-pot multicomponent reaction that fundamentally reshapes the production paradigm. This innovative route utilizes readily available starting materials, specifically trifluoroethylimidoyl chloride, propargylamine, and acrylamide derivatives, which are combined under mild catalytic conditions. The core breakthrough lies in the use of a formic acid and acetic anhydride mixture to generate carbon monoxide in situ, thereby circumventing the dangers associated with external CO gas sources. This not only enhances operational safety but also simplifies the reactor setup, allowing the reaction to proceed efficiently at a mild temperature of 30°C. The versatility of this system allows for the synthesis of diversified substituted double heterocyclic compounds with trifluoromethyl and carbonyl functionalities, providing a robust platform for drug discovery and process development.
Mechanistic Insights into Pd-Catalyzed Carbonylation Cascade
To fully appreciate the value proposition for R&D teams, one must delve into the intricate mechanistic pathway that drives this high-efficiency transformation. The reaction is initiated by the oxidative addition of a zero-valent palladium species into the carbon-iodine bond of the acrylamide substrate, followed by an intramolecular Heck reaction that generates a divalent alkyl palladium intermediate. This key intermediate then undergoes a carbonylation step facilitated by the carbon monoxide released from the formic acid-acetic anhydride system, resulting in the formation of a reactive acyl palladium species. Concurrently, a base-promoted intermolecular carbon-nitrogen bond formation occurs between the trifluoroethylimidoyl chloride and propargylamine, yielding a trifluoroacetamidine compound that subsequently isomerizes. The final stage involves the activation of this trifluoroacetamidine by the acyl palladium intermediate, catalyzing an intramolecular cyclization that constructs the final carbonyl-bridged biheterocyclic core with high precision.
Beyond mere bond formation, the mechanism offers profound implications for impurity control and product purity, which are paramount concerns for regulatory compliance. The mild reaction conditions of 30°C significantly reduce the thermal energy available for side reactions, such as polymerization of the acrylamide or decomposition of sensitive functional groups, which are common issues in high-temperature processes. The specific ligand system, utilizing trifurylphosphine alongside palladium chloride, stabilizes the catalytic cycle and ensures high turnover numbers, minimizing the residual metal content in the crude product. This inherent selectivity means that the resulting crude mixture contains fewer by-products, simplifying the downstream purification workflow. For manufacturers targeting high-purity biheterocyclic compounds, this mechanistic elegance translates directly into reduced solvent consumption during chromatography and higher overall yields, reinforcing the economic and technical superiority of this route.
How to Synthesize Carbonyl-Bridged Biheterocyclic Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific operational parameters to maximize yield and reproducibility. The process begins with the precise preparation of the catalytic system, where palladium chloride and trifurylphosphine are mixed with sodium carbonate and the CO-generating reagents in an aprotic solvent such as tetrahydrofuran. The choice of solvent is critical, as THF has been identified as optimal for dissolving the diverse range of substrates while maintaining high conversion rates. Once the catalyst system is established, the sequential or simultaneous addition of the trifluoroethylimidoyl chloride, propargylamine, and acrylamide substrates initiates the cascade. The detailed standardized synthesis steps see the guide below, which outlines the exact molar ratios and workup procedures validated in the patent examples to ensure consistent quality.
- Prepare the reaction mixture by combining palladium chloride, trifurylphosphine, sodium carbonate, and the formic acid-acetic anhydride CO source in THF solvent.
- Add trifluoroethylimidoyl chloride, propargylamine, and the acrylamide substrate to the catalyst system and stir at 30°C for 12 to 20 hours.
- Upon completion, filter the mixture, mix with silica gel, and purify via column chromatography to isolate the target carbonyl-bridged biheterocyclic compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic methodology offers tangible strategic benefits that extend beyond simple technical metrics. The primary advantage lies in the drastic simplification of the supply chain for raw materials; the key starting materials, including propargylamine and various acrylamides, are commercially available commodities that do not suffer from the geopolitical or logistical constraints often associated with specialized reagents. Furthermore, the elimination of toxic carbon monoxide gas cylinders removes a major regulatory and safety burden from the facility, reducing insurance costs and compliance overhead. This shift allows for more flexible manufacturing scheduling and reduces the lead time for high-purity intermediates, as the safety protocols required for high-pressure gas handling are no longer a bottleneck. Consequently, organizations can achieve substantial cost savings through both direct material optimization and indirect operational efficiencies.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the replacement of expensive and hazardous reagents with cheap, easy-to-obtain alternatives. By utilizing a formic acid-acetic anhydride system for carbonylation, the process avoids the capital expenditure associated with high-pressure autoclaves and gas monitoring systems required for traditional CO methods. Additionally, the high reaction efficiency and substrate compatibility mean that less material is wasted on failed batches or extensive purification, leading to a lower cost of goods sold (COGS). The ability to operate at ambient pressure and mild temperatures also results in significantly reduced energy consumption compared to high-thermal-demand processes, further enhancing the overall cost-effectiveness of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Supply continuity is critical for pharmaceutical production, and this method bolsters reliability by relying on a robust portfolio of starting materials. Since trifluoroethylimidoyl chloride and propargylamine are synthesized from widely available precursors like aromatic amines and acryloyl chloride, the risk of raw material shortage is minimized. The process tolerance for various functional groups also means that if a specific substrate variant is unavailable, alternative analogs can often be substituted without re-optimizing the entire process. This flexibility ensures that production schedules remain intact even when facing market volatility, providing a stable foundation for long-term supply agreements and reducing the risk of project delays due to material scarcity.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the plant floor often introduces unforeseen challenges, but this patent explicitly demonstrates successful expansion to the gram-scale, indicating strong potential for tonnage production. The simplified post-treatment process, involving filtration and standard column chromatography, is easily adaptable to industrial crystallization or extraction techniques, facilitating a smooth technology transfer. Moreover, the avoidance of toxic gases and the use of relatively benign solvents align with increasingly stringent environmental regulations, reducing the burden on waste treatment facilities. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in global B2B partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for stakeholders evaluating this technology for adoption. Understanding these nuances is crucial for making informed decisions about process integration and vendor selection.
Q: How does this method improve safety compared to traditional carbonylation?
A: Unlike conventional methods requiring toxic carbon monoxide gas cylinders, this patent utilizes a formic acid-acetic anhydride system to generate CO in situ, significantly enhancing operational safety and eliminating high-pressure gas handling requirements.
Q: What is the substrate compatibility for this synthesis route?
A: The methodology demonstrates broad functional group tolerance, accommodating various substituents such as halogens, alkyl groups, alkoxy, and trifluoromethyl groups on the aromatic rings without compromising reaction efficiency.
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly validates the method at the gram-scale level with simple post-treatment procedures, indicating strong potential for commercial scale-up of complex heterocycles in an industrial setting.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Carbonyl-Bridged Biheterocyclic Compounds Supplier
As the demand for complex heterocyclic scaffolds continues to rise in the pharmaceutical and agrochemical sectors, partnering with an experienced CDMO is vital for translating patent potential into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move seamlessly from development to full-scale manufacturing. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of carbonyl-bridged biheterocyclic compounds meets the highest international standards. We understand the critical nature of timeline and quality in drug development, and our team is dedicated to providing the technical support necessary to optimize this specific palladium-catalyzed route for your unique requirements.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your volume needs. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that you have all the necessary information to proceed with confidence. Let us help you leverage this advanced technology to accelerate your product development and secure a competitive position in the market.
