Optimizing Ceftazidime Production: A High-Yield Route for Global API Manufacturers
Optimizing Ceftazidime Production: A High-Yield Route for Global API Manufacturers
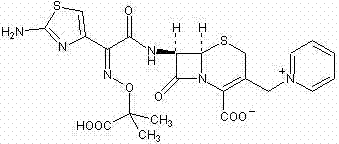
The pharmaceutical landscape demands increasingly efficient pathways for producing critical antibiotics, and the synthesis of ceftazidime stands as a prime example where process innovation drives commercial viability. Based on the technical disclosures in patent CN102286003A, a novel synthetic methodology has been established that fundamentally restructures the production of this third-generation cephalosporin. Unlike conventional routes that often suffer from prolonged reaction times and complex purification sequences, this approach leverages a streamlined silylation strategy starting from 7-aminocephalosporanic acid (7-ACA). The core breakthrough lies in the direct conversion of the starting material into 7-amino-3-(1-picolyl)-cephem acid (7-APCA) hydrochloride, bypassing the inefficient multi-step iodate preparations typical of legacy technologies. By integrating optimized acylation conditions and precise pH-controlled crystallization, this method delivers a robust framework for high-purity API intermediate manufacturing. For R&D directors and supply chain leaders, understanding this chemistry is essential for securing a reliable ceftazidime supplier capable of meeting global demand without compromising on quality or cost-efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of ceftazidime has been plagued by significant operational bottlenecks that inflate production costs and extend lead times. Traditional methodologies, such as those referenced in WO8504659, typically rely on a disjointed sequence where the introduction of the pyridine moiety at the C-3 position requires distinct, separate steps often involving unstable iodate intermediates. These legacy processes frequently necessitate rigorous nitrogen protection atmospheres to prevent oxidation or moisture interference, thereby increasing capital expenditure on specialized reactor equipment and inert gas consumption. Furthermore, the reliance on specific catalysts in the acylation phase can introduce heavy metal impurities that require costly downstream removal processes to meet pharmacopeial standards. The cumulative effect of these inefficiencies is a lower overall yield, often exacerbated by the instability of ceftazidime crystals which can degrade if not handled with extreme precision during isolation. Such complexities create substantial supply chain vulnerabilities, making it difficult for procurement managers to secure consistent volumes of high-purity material at competitive price points.
The Novel Approach
In stark contrast, the methodology outlined in CN102286003A introduces a paradigm shift by consolidating the synthesis of the critical 7-APCA intermediate into a single, highly efficient operation. This novel approach utilizes a mixed silylating reagent system comprising hexamethyldisilazane, trimethylchlorosilane, and iodotrimethylsilane to activate the 7-ACA nucleus, allowing for the direct introduction of the pyridinium ion without the need for prior iodate formation. A key differentiator is the elimination of nitrogen protection during this initial phase, which not only simplifies the engineering requirements but also facilitates the safe discharge of ammonia byproducts. The subsequent acylation step is equally innovative, operating effectively without additional catalysts by optimizing solvent ratios of dichloromethane and methanol alongside triethylamine. This "one-pot" philosophy reduces the number of unit operations, minimizes solvent switching, and significantly enhances the throughput capacity. For manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing, this route offers a tangible pathway to maximize asset utilization while maintaining exceptional control over the impurity profile.
Mechanistic Insights into Silylation-Mediated Cephalosporin Functionalization
The chemical elegance of this synthesis lies in the precise manipulation of the cephem nucleus through silylation protection and nucleophilic substitution. The process initiates with the silylation of the amino group at the C-7 position of 7-ACA using hexamethyldisilazane under reflux conditions, which temporarily masks the amine to prevent unwanted side reactions during the subsequent C-3 modification. The introduction of iodotrimethylsilane serves a dual purpose: it acts as a source of iodide for the displacement of the C-3 leaving group and generates reactive silyl species that enhance the electrophilicity of the methyl position. When pyridine is introduced, it undergoes a rapid nucleophilic attack on the activated C-3 methylene, forming the stable pyridinium salt structure characteristic of ceftazidime precursors. The use of xylene as a co-solvent or catalyst aid further stabilizes the transition state, ensuring that the reaction proceeds to completion with residual 7-ACA levels dropping below 0.5%. This mechanistic efficiency is critical for R&D teams focused on impurity control, as it minimizes the formation of regio-isomers or degradation products that often complicate purification in less optimized systems.
Following the formation of the 7-APCA hydrochloride, the acylation mechanism is driven by the high nucleophilicity of the deprotected C-7 amine towards the activated thiazole side chain ester. The patent specifies the use of alpha-(2-amino-4-thiazolyl)-alpha-[(1-tert-butoxycarbonyl-1-methylethoxy)imino]acetic acid benzothiazolyl ester (TAEM) as the acylating agent. In the absence of traditional Lewis acid catalysts, the reaction relies on the careful modulation of basicity using triethylamine within a dichloromethane and methanol solvent matrix. This specific solvent environment solubilizes both the zwitterionic cephalosporin intermediate and the lipophilic side chain, facilitating molecular collision and bond formation at mild temperatures between 10°C and 15°C. The resulting tert-butyl ester intermediate is then subjected to acidic hydrolysis, where the tert-butoxycarbonyl protecting group is cleaved to reveal the free carboxylic acid. The final crystallization step exploits the zwitterionic nature of ceftazidime, adjusting the pH to the isoelectric point range of 3.6 to 4.0 to precipitate the product in its purest form, effectively excluding ionic impurities and residual solvents.
How to Synthesize Ceftazidime Efficiently
The execution of this synthesis requires strict adherence to the defined parameters to replicate the high yields and purity reported in the patent literature. The process is divided into four distinct operational phases: the preparation of the 7-APCA hydrochloride salt, the coupling of the side chain to form the tert-butyl ester, the hydrolytic deprotection to the dihydrochloride salt, and the final pH-adjusted crystallization of the active pharmaceutical ingredient. Each step builds upon the previous one, relying on the high purity of the intermediate to drive the efficiency of the subsequent reaction. Operators must pay particular attention to the temperature controls during the acylation phase and the precise pH ramping during the final isolation to ensure optimal crystal morphology and filterability. The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and processing times.
- Convert 7-ACA to 7-APCA hydrochloride using hexamethyldisilazane and iodotrimethylsilane under reflux without nitrogen protection.
- Perform acylation with the thiazole side chain ester in dichloromethane and methanol using triethylamine at 10-15°C.
- Hydrolyze the tert-butyl ester using hydrochloric and formic acid mixture, followed by acetone crystallization.
- Adjust pH using NaOH and sodium carbonate to isolate the final ceftazidime crystal with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates directly into enhanced operational resilience and significant cost optimization opportunities. By eliminating the need for nitrogen protection in the initial silylation step, manufacturers can reduce utility costs and simplify reactor maintenance schedules, leading to a more agile production environment. The removal of expensive catalysts in the acylation stage further strips away raw material costs and eliminates the need for specialized scavenging resins or filtration media typically required to remove metal residues. This streamlining of the bill of materials allows for a more predictable cost structure, shielding buyers from volatility associated with specialty reagents. Moreover, the use of common, recyclable solvents like isopropanol and dichloromethane aligns with modern environmental compliance standards, reducing the burden of hazardous waste disposal and lowering the overall environmental footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The consolidation of the 7-APCA preparation into a single step drastically reduces labor hours and energy consumption compared to multi-step legacy methods. By avoiding the formation of unstable iodate intermediates, the process minimizes material loss due to decomposition, thereby improving the effective mass balance. The ability to reuse siloxane byproducts further contributes to a circular economy within the plant, turning potential waste streams into valuable resources. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), enabling competitive pricing strategies for the final API without sacrificing margin.
- Enhanced Supply Chain Reliability: The robustness of this chemical route ensures consistent batch-to-batch quality, a critical factor for maintaining uninterrupted supply to downstream formulation partners. The high purity of the intermediates, consistently exceeding 98-99%, reduces the risk of batch rejection during quality control testing, which is a common cause of supply delays. Additionally, the simplified equipment requirements mean that production can be scaled across multiple facilities with minimal requalification effort, diversifying the supply base and mitigating the risk of single-source bottlenecks. This reliability is paramount for securing long-term contracts with major pharmaceutical companies that demand guaranteed delivery timelines.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as reflux, filtration, and crystallization that are easily replicated in large-scale reactors. The avoidance of cryogenic conditions or extreme pressures reduces safety risks and insurance costs associated with high-hazard operations. Furthermore, the generation of easily treatable "three wastes" (waste water, gas, and residue) simplifies regulatory compliance, ensuring that production can continue uninterrupted by environmental audits. This scalability supports the commercial expansion of complex pharmaceutical intermediates, allowing suppliers to rapidly respond to surges in market demand for critical antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ceftazidime synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this route for potential partners. Understanding these details is crucial for making informed decisions about sourcing strategies and technology transfer initiatives.
Q: How does this synthesis method improve upon traditional ceftazidime production?
A: This method simplifies the preparation of the key intermediate 7-APCA hydrochloride into a single step rather than two, eliminates the need for nitrogen protection, and avoids expensive catalysts in the acylation stage, resulting in higher overall yields and reduced operational complexity.
Q: What are the purity specifications achievable with this route?
A: The patented process demonstrates the ability to achieve intermediate purities exceeding 99% for 7-APCA hydrochloride and final product content greater than 98%, meeting stringent pharmaceutical standards through controlled crystallization and pH adjustment.
Q: Is this process scalable for industrial manufacturing?
A: Yes, the process is designed for industrial suitability by utilizing common solvents like dichloromethane and isopropanol, minimizing hazardous waste through reusable siloxane byproducts, and employing robust crystallization techniques that ensure consistent batch-to-batch quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ceftazidime Supplier
At NINGBO INNO PHARMCHEM, we recognize that the technical potential of a synthesis route is only as valuable as its execution at scale. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN102286003A are fully realized in our manufacturing suites. Our facility is equipped with state-of-the-art reactors capable of handling the specific solvent systems and temperature profiles required for this silylation-based chemistry. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC methods to verify that every batch of ceftazidime meets or exceeds the 98% content threshold required by global pharmacopeias. Our commitment to quality assurance ensures that the impurity profiles remain tightly controlled, providing our clients with the confidence needed for regulatory filings.
We invite forward-thinking pharmaceutical companies to collaborate with us on optimizing their cephalosporin supply chains. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can become the cornerstone of your ceftazidime sourcing strategy, delivering both value and reliability in a competitive market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
