Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
Advanced Carbonylation Strategy for Scalable Thioester Production in Pharmaceutical Manufacturing
The landscape of organic synthesis is constantly evolving to meet the rigorous demands of modern pharmaceutical manufacturing, particularly regarding the efficient production of key structural motifs like thioesters. Patent CN113004181B introduces a groundbreaking methodology for preparing thioester compounds through a transition metal-catalyzed carbonylation reaction. This innovation addresses long-standing challenges in the field by utilizing inexpensive and readily available benzyl chloride compounds alongside sulfonyl chlorides as starting materials. Unlike traditional routes that rely on hazardous reagents, this novel approach leverages a sophisticated palladium catalytic system supported by Xantphos ligands and tungsten carbonyl. For R&D directors and process chemists, this represents a significant leap forward in designing safer, more robust synthetic pathways that minimize operational complexity while maximizing yield and purity profiles essential for downstream drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thioester compounds has been fraught with significant operational and safety hurdles that complicate large-scale manufacturing. The conventional standard involves the direct acylation of thiols with carboxylic acids or their derivatives, a process heavily reliant on mercaptans as the sulfur source. Mercaptans are notoriously unpleasant due to their intense, repulsive odors, which pose severe health and safety risks in industrial settings and require specialized containment infrastructure. Furthermore, these sulfur-containing compounds are prone to oxidizing easily, leading to impurity profiles that are difficult to manage. Additionally, thiols act as potent catalyst poisons, often deactivating the very transition metal catalysts required for efficient coupling, thereby necessitating higher catalyst loadings or harsher reaction conditions that degrade overall process economics and environmental compliance.
The Novel Approach
The methodology disclosed in CN113004181B fundamentally reimagines the thioester synthesis landscape by replacing problematic thiols with stable, odorless sulfonyl chlorides. This strategic shift eliminates the safety hazards associated with volatile sulfur sources and prevents catalyst deactivation, allowing for smoother reaction kinetics. The process employs a dual-function reagent, tungsten hexacarbonyl, which acts simultaneously as the carbonyl source and the reducing agent, streamlining the reagent bill of materials. By operating under mild conditions in acetonitrile at 100°C, this route achieves high conversion rates with excellent functional group tolerance. This novel approach not only simplifies the post-treatment workflow but also opens up new possibilities for substrate design, enabling the synthesis of complex thioester architectures that were previously difficult to access reliably.
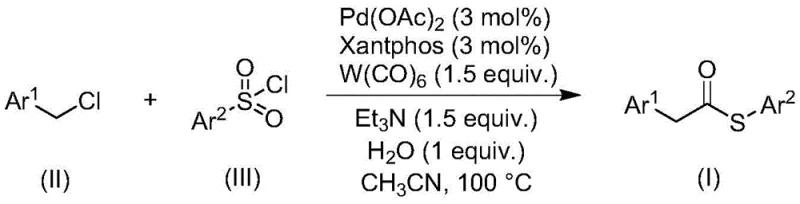
Mechanistic Insights into Palladium-Catalyzed Carbonylation
The core of this technological breakthrough lies in the intricate interplay between the palladium catalyst and the unique reagents employed. The reaction initiates with the oxidative addition of the benzyl chloride to the active palladium(0) species generated in situ from palladium acetate and the bulky bidentate phosphine ligand, Xantphos. The wide bite angle of Xantphos is critical here, as it stabilizes the palladium center and facilitates the subsequent insertion of carbon monoxide derived from the decomposition of tungsten hexacarbonyl. Unlike traditional carbonylations that require high-pressure CO gas cylinders, this system generates CO internally, enhancing safety and controllability. The sulfonyl chloride then participates in the cycle, likely undergoing reduction and sulfur transfer to form the thioester bond, a mechanism that bypasses the need for free thiol intermediates entirely. This elegant mechanistic pathway ensures that the sulfur atom is incorporated efficiently without generating malodorous byproducts.
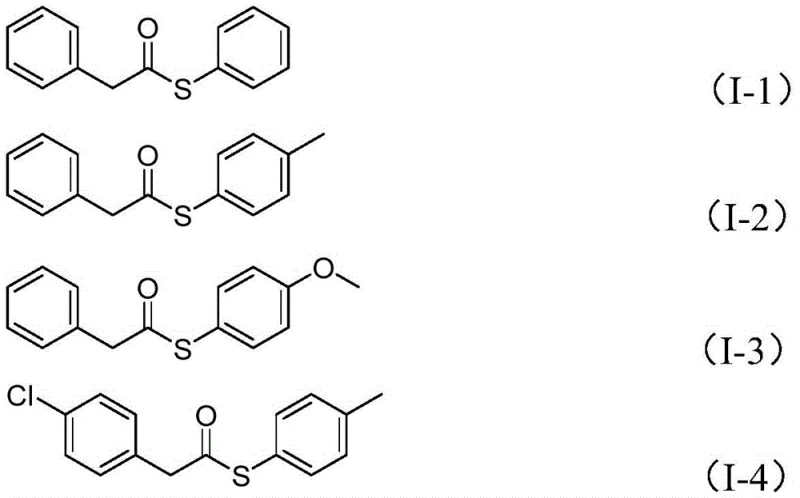
From an impurity control perspective, this mechanism offers distinct advantages for producing high-purity pharmaceutical intermediates. By avoiding free thiols, the formation of disulfide impurities—a common plague in traditional thioester synthesis—is effectively suppressed. The use of tungsten hexacarbonyl as a stoichiometric reductant ensures that the reaction environment remains sufficiently reducing to drive the transformation to completion without over-reducing sensitive functional groups on the aromatic rings. The compatibility with various substituents, such as halogens, methoxy, and alkyl groups, indicates a robust catalytic cycle that tolerates electronic diversity. This level of control is paramount for R&D teams aiming to synthesize libraries of analogs for structure-activity relationship studies, as it guarantees consistent quality and minimizes the burden on downstream purification processes like column chromatography.
How to Synthesize Thioester Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires precise adherence to the optimized parameters established in the patent data. The procedure involves charging a reaction vessel with the specific molar ratios of palladium acetate, Xantphos, tungsten carbonyl, triethylamine, and water, followed by the addition of the benzyl chloride and sulfonyl chloride substrates in acetonitrile. The mixture is then heated to 100°C for approximately 24 hours to ensure full conversion. The detailed standardized synthesis steps, including specific workup procedures and purification protocols, are outlined below to guide technical teams in replicating these high-yield results consistently.
- Charge a reaction vessel with palladium acetate, Xantphos ligand, tungsten carbonyl, triethylamine, water, benzyl chloride, and sulfonyl chloride in acetonitrile.
- Heat the reaction mixture to 100°C and maintain stirring for 24 hours to ensure complete conversion.
- Filter the mixture, mix with silica gel, and purify via column chromatography to isolate the high-purity thioester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this carbonylation technology translates into tangible strategic benefits that extend beyond mere chemical efficiency. The shift from volatile, hazardous thiols to stable, solid sulfonyl chlorides drastically simplifies raw material handling and storage logistics. This change reduces the need for specialized scrubbing systems and personal protective equipment associated with noxious gases, thereby lowering the total cost of ownership for the manufacturing facility. Furthermore, the elimination of external high-pressure carbon monoxide sources removes a significant safety bottleneck, allowing for more flexible reactor scheduling and reduced regulatory compliance burdens related to toxic gas management.
- Cost Reduction in Manufacturing: The economic viability of this process is significantly enhanced by the dual role of tungsten hexacarbonyl, which serves as both the carbonyl source and the reducing agent. This consolidation of reagent functions eliminates the need for purchasing and handling separate reducing agents, directly reducing the raw material costs per kilogram of product. Additionally, the use of inexpensive benzyl chlorides and sulfonyl chlorides, which are commodity chemicals with stable global supply chains, ensures that input costs remain predictable and low. The high reaction efficiency and simplified post-treatment, which avoids complex extraction sequences, further contribute to substantial cost savings by reducing solvent consumption and labor hours required for isolation.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available, non-hazardous starting materials that are not subject to the same strict transportation regulations as thiols or pressurized gases. The stability of sulfonyl chlorides allows for bulk purchasing and long-term storage without degradation, mitigating the risk of production stoppages due to raw material spoilage. Moreover, the robustness of the catalytic system means that the process is less sensitive to minor variations in feedstock quality, ensuring consistent output even when sourcing from different suppliers. This reliability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of gaseous reagents that require specialized high-pressure equipment. The reaction operates at atmospheric pressure in standard glass-lined or stainless steel reactors, making it highly adaptable to existing manufacturing infrastructure. From an environmental standpoint, the avoidance of malodorous thiols significantly improves the workplace environment and reduces the risk of community complaints, a key factor in maintaining social license to operate. The simplified waste stream, devoid of sulfur-heavy byproducts, eases the burden on wastewater treatment facilities and aligns with increasingly stringent green chemistry mandates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding this carbonylation technology, derived directly from the patent specifications and experimental data. These insights are intended to clarify the practical implications of adopting this method for industrial applications, focusing on safety, efficiency, and product quality. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this route into their current manufacturing portfolios.
Q: Why is sulfonyl chloride preferred over thiols for thioester synthesis?
A: Sulfonyl chlorides are odorless, stable solids that eliminate the safety hazards and catalyst poisoning issues associated with volatile, foul-smelling thiol compounds, ensuring a cleaner reaction profile.
Q: What is the role of Tungsten Carbonyl in this reaction?
A: Tungsten carbonyl serves a dual function as both the carbonyl source (CO donor) and the reducing agent, removing the need for additional external reductants and simplifying the reagent load.
Q: Does this method tolerate diverse functional groups?
A: Yes, the protocol demonstrates excellent tolerance for various substituents including halogens, alkyl groups, and alkoxy groups on both the benzyl and aryl rings, yielding products with 55-78% efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thioester Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic methodologies in the development of next-generation pharmaceutical intermediates. Our team of expert process chemists has thoroughly analyzed the potential of this palladium-catalyzed carbonylation route and is fully equipped to translate these laboratory findings into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from benchtop to plant floor is seamless and efficient. Our state-of-the-art facilities are designed to handle complex organometallic reactions with precision, adhering to stringent purity specifications and utilizing rigorous QC labs to guarantee that every batch meets the highest international standards.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. By partnering with our technical procurement team, you can gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and timeline. We encourage you to reach out today to request specific COA data for our thioester portfolio and to discuss route feasibility assessments that could optimize your supply chain. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
