Revolutionizing Nitrogen Heterocycle Alkylation: A Cost-Effective Copper-Catalyzed Strategy for Commercial Scale-up
The landscape of medicinal chemistry is constantly evolving, driven by the relentless demand for novel scaffolds that can modulate biological targets with high specificity. Among the myriad of transformations available to synthetic chemists, the direct functionalization of nitrogen-containing heterocycles stands out as a cornerstone for constructing drug-like molecules. A significant breakthrough in this domain is detailed in patent CN112961105A, which discloses a highly efficient alkylation method for nitrogen-containing heterocyclic compounds. This technology leverages a robust copper-catalyzed system combined with Selectfluor and peroxides to achieve direct C-H alkylation under remarkably mild conditions. For R&D directors and procurement specialists alike, this represents a paradigm shift away from precious metal dependency towards a more sustainable and economically viable manufacturing model. The ability to introduce alkyl groups directly onto heteroarenes using simple alkanes not only streamlines the synthetic route but also opens new avenues for late-stage functionalization of complex drug candidates, thereby accelerating the timeline from bench to bedside.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the alkylation of nitrogen-containing heterocycles, often referred to as Minisci-type reactions, has been plagued by significant operational and economic hurdles that hinder large-scale adoption. Traditional approaches frequently rely on photocatalysis utilizing expensive Iridium or Ruthenium complexes, which not only inflate the raw material costs but also necessitate specialized photoreactors that are difficult to scale beyond gram quantities. Alternatively, silver-catalyzed methods have been employed, yet these often suffer from inconsistent yields and the generation of heavy metal waste that requires rigorous and costly removal processes to meet pharmaceutical purity standards. Furthermore, many conventional protocols demand harsh reaction conditions, including high temperatures, strong oxidants, or strictly anaerobic environments, which complicate the engineering controls required for safe industrial production. These limitations collectively create a bottleneck in the supply chain, leading to extended lead times and unpredictable batch-to-batch variability that procurement managers find increasingly unacceptable in a competitive market.
The Novel Approach
In stark contrast to these legacy methods, the novel approach outlined in the patent data introduces a streamlined catalytic cycle centered around earth-abundant copper species. By utilizing a combination of a copper catalyst, Selectfluor, and a peroxide oxidant, this method activates inert sp3 C-H bonds in simple alkanes to couple efficiently with electron-deficient nitrogen heterocycles. The reaction proceeds smoothly at moderate temperatures ranging from 40 to 60 ℃, eliminating the need for energy-intensive heating or cryogenic cooling. Crucially, the system tolerates a broad spectrum of substrates, including substituted pyrazines, quinolines, and pyridines, as demonstrated by the diverse array of successful transformations. 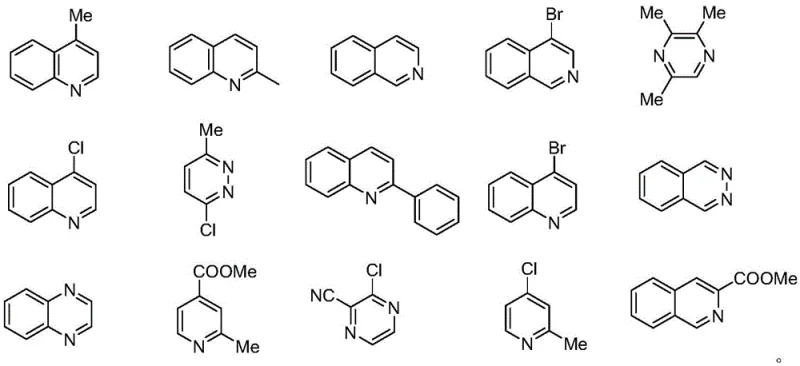
This versatility ensures that the process is not limited to a single niche application but serves as a general platform for synthesizing a wide library of alkylated derivatives. The operational simplicity is further enhanced by the fact that the reaction does not require dehydration or strict exclusion of oxygen, drastically reducing the complexity of the reactor setup and making it highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Copper-Catalyzed C-H Alkylation
To fully appreciate the technical sophistication of this methodology, one must delve into the mechanistic underpinnings that drive the transformation. The reaction initiates with the activation of the alkyl reagent, where the copper catalyst interacts with the peroxide and Selectfluor to generate reactive radical species. These radicals, derived from the sp3 C-H bonds of the alkane, are sufficiently electrophilic to attack the electron-deficient positions on the nitrogen-containing heterocyclic ring. The presence of the acid component plays a critical role in protonating the heterocycle, thereby increasing its electrophilicity and facilitating the radical addition step. Following the addition, the intermediate undergoes oxidation and deprotonation to restore aromaticity, yielding the final alkylated product. This catalytic cycle is highly efficient, with the copper species being regenerated to sustain the reaction turnover, ensuring that only catalytic amounts (5-10 mol%) are required to drive the process to completion.
From an impurity control perspective, the mechanism offers distinct advantages over radical chain reactions that often lead to poly-alkylation or uncontrolled polymerization. The specific interplay between the Selectfluor reagent and the copper catalyst creates a controlled environment where the concentration of free radicals is managed effectively. This minimizes the formation of over-alkylated byproducts and ensures a clean reaction profile. The use of mild oxidants like sodium persulfate further contributes to the selectivity, preventing the degradation of sensitive functional groups that might be present on the heterocyclic scaffold. Consequently, the resulting crude reaction mixtures are cleaner, which simplifies the downstream purification workup and reduces the overall solvent consumption associated with extensive chromatographic separations. 
How to Synthesize Alkylated Nitrogen Heterocycles Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires adherence to specific procedural parameters to maximize yield and reproducibility. The patent data provides a clear roadmap for executing the reaction, emphasizing the importance of reagent ratios and solvent selection. While the general procedure is robust, fine-tuning the equivalents of the oxidant and fluorinating reagent can push yields to near-quantitative levels for certain substrates. The following guide summarizes the critical operational steps derived from the experimental examples, serving as a foundational protocol for process chemists looking to adopt this technology.
- Mix the nitrogen-containing heterocyclic compound, alkyl reagent, acid, peroxide, Selectfluor fluorine reagent, and Cu catalyst in an organic solvent such as acetonitrile.
- Heat the reaction mixture to a mild temperature range of 40-60 ℃ and stir for approximately 16 hours to ensure complete conversion.
- Extract the reaction liquid with ethyl acetate, remove the solvent via rotary evaporation, and purify the crude product using column chromatography to obtain high-purity derivatives.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders responsible for the bottom line and supply continuity, the transition to this copper-catalyzed alkylation method offers compelling strategic benefits that extend far beyond mere technical feasibility. The elimination of precious metal catalysts like Iridium immediately translates to a drastic reduction in raw material costs, which is a primary driver for margin improvement in high-volume manufacturing. Moreover, the simplified operational requirements—specifically the absence of anaerobic conditions and the use of ambient pressure—lower the barrier to entry for contract manufacturing organizations, thereby expanding the pool of potential suppliers and enhancing supply chain resilience. This flexibility is crucial for mitigating risks associated with single-source dependencies and ensures a steady flow of critical intermediates even during periods of market volatility.
- Cost Reduction in Manufacturing: The substitution of expensive photocatalysts with inexpensive copper salts represents a fundamental shift in the cost structure of the synthesis. Copper is orders of magnitude cheaper than Iridium or Silver, and its usage in catalytic amounts means the metal cost per kilogram of product is negligible. Additionally, the mild reaction temperatures reduce energy consumption for heating and cooling, while the simplified workup procedures decrease labor hours and solvent usage. These cumulative efficiencies result in substantial cost savings that can be passed down the supply chain, making the final API more competitive in the global marketplace without compromising on quality.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as cyclohexane, acetonitrile, and sodium persulfate ensures that the raw material supply is stable and不受 geopolitical disruptions that often affect specialty reagents. Since the reaction does not require specialized equipment like photoreactors or gloveboxes, it can be performed in standard stainless steel reactors available in most multipurpose chemical plants. This compatibility with existing infrastructure accelerates the technology transfer process and reduces the lead time for scaling up from pilot to commercial production, ensuring that project timelines are met consistently.
- Scalability and Environmental Compliance: The green chemistry metrics of this process are superior to traditional methods, aligning with the increasing regulatory pressure for sustainable manufacturing. The use of less toxic reagents and the generation of reduced waste streams simplify the environmental permitting process and lower waste disposal costs. The high atom economy of directly functionalizing C-H bonds means fewer steps are required to build molecular complexity, which inherently reduces the overall environmental footprint of the synthesis. This sustainability profile is increasingly becoming a key differentiator for suppliers seeking to partner with major pharmaceutical companies that have aggressive carbon reduction goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the process capabilities. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary advantages of this copper-catalyzed method over traditional photocatalysis?
A: Unlike traditional photocatalysis which often relies on expensive Iridium complexes and requires specialized light equipment, this method utilizes inexpensive and readily available copper catalysts. Furthermore, it operates under mild thermal conditions (40-60 ℃) without the need for strict anaerobic environments, significantly simplifying the operational complexity and reducing capital expenditure for industrial reactors.
Q: What types of alkyl reagents are compatible with this synthesis route?
A: The process demonstrates excellent substrate universality, accommodating a wide range of sp3 C-H bond-containing reagents. Compatible alkyl sources include saturated alkanes like cyclohexane and cyclododecane, as well as ether-containing compounds such as tetrahydrofuran, dioxane, and tetrahydropyran, allowing for diverse functionalization of the heterocyclic core.
Q: How does this method impact the purity profile of the final pharmaceutical intermediate?
A: By optimizing the ratio of Selectfluor and sodium persulfate, the reaction achieves high conversion rates with yields reaching up to 97% for specific substrates. The mild reaction conditions minimize side reactions and decomposition, facilitating easier downstream purification via standard column chromatography to meet stringent purity specifications required for API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nitrogen-containing Heterocyclic Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of new therapeutic agents depends heavily on the robustness and scalability of the underlying synthetic routes. Our team of expert process chemists has thoroughly evaluated the copper-catalyzed alkylation methodology described in patent CN112961105A and confirmed its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of alkylated heterocycle we deliver meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of this advanced catalytic technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this route can improve your margins. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for complex intermediate synthesis.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
