Advanced Manufacturing of Ioxilan: A Strategic Route for High-Purity Contrast Agents
Advanced Manufacturing of Ioxilan: A Strategic Route for High-Purity Contrast Agents
The pharmaceutical landscape for non-ionic X-ray contrast agents has evolved significantly with the introduction of Ioxilan, a compound renowned for its low osmotic pressure and superior biological tolerance. As detailed in patent CN1800149A, a novel preparation method has emerged that fundamentally restructures the synthetic pathway to enhance efficiency and product quality. This technical insight report analyzes the strategic shift from traditional alkylation-first approaches to a more robust acylation-first methodology, offering critical value for R&D directors seeking purity and procurement managers aiming for cost optimization. The chemical structure of Ioxilan, characterized by its triiodinated benzene ring and asymmetric side chains, presents unique synthetic challenges that this new process elegantly resolves.
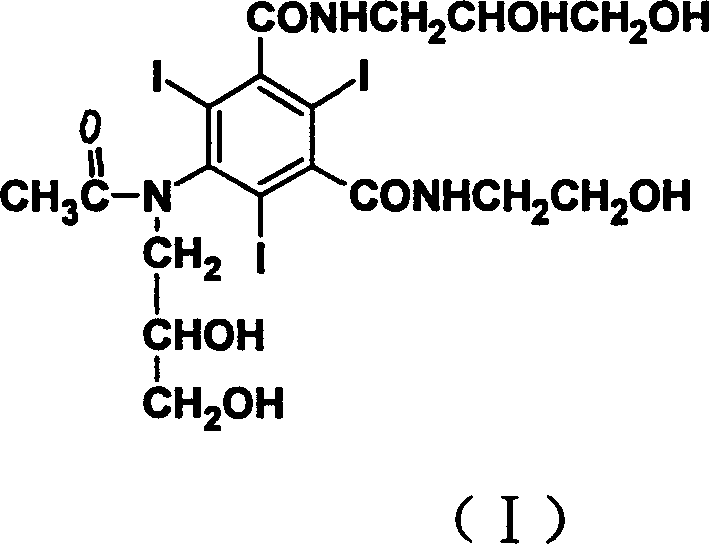
Ioxilan represents a significant advancement over previous generations of contrast media due to its reduced viscosity and minimized side effects such as ventricular fibrillation. The core innovation lies in the sequence of functional group transformations, specifically delaying the critical N-alkylation step to the final stage of synthesis. By starting with 5-amino-N-(2-hydroxyethyl)-2,4,6-triiodo-isophthalic acid monoamide, the process avoids the competitive O-alkylation issues that plague earlier methods. This strategic reordering not only simplifies purification but also drastically improves the overall yield and consistency of the final active pharmaceutical ingredient (API), positioning it as a prime candidate for reliable pharmaceutical intermediates supplier networks globally.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ioxilan, as disclosed in US Patent 4954348, relied on an initial alkylation step that introduced significant inefficiencies into the manufacturing workflow. In this conventional route, the alkylation of the amino group competes fiercely with the alkylation of the hydroxyl groups, leading to a reaction conversion ratio of merely 88%. This competition generates O-alkylated impurities that possess chemical structures strikingly similar to the desired product, making their separation and removal extremely difficult and costly. Furthermore, the subsequent protection of hydroxyl groups involves complex workup procedures, including multiple azeotropic distillations and extractions, which are unfavorable for industrial suitability. The excessive use of thionyl chloride as both a reagent and a solvent in the acyl chloride formation step further exacerbates safety concerns and environmental pollution, creating a bottleneck for cost reduction in contrast agent manufacturing.
The Novel Approach
The methodology presented in CN1800149A offers a transformative solution by reversing the synthetic order, prioritizing acylation and amidation before the final alkylation. This approach ensures that the N-alkylation occurs on a fully protected and structured intermediate, effectively eliminating the competitive O-alkylation pathway. Experimental data indicates that this late-stage alkylation achieves a reaction conversion ratio reaching 95%, with O-alkyl impurities reduced to less than 1%. The process utilizes milder conditions and safer solvents, such as ethyl acetate, to mediate the acyl chloride formation, significantly reducing the consumption of hazardous thionyl chloride. By streamlining the workup procedures to simple solvent evaporation and recrystallization, this novel route enhances the commercial scale-up of complex pharmaceutical intermediates while ensuring a cleaner impurity profile.
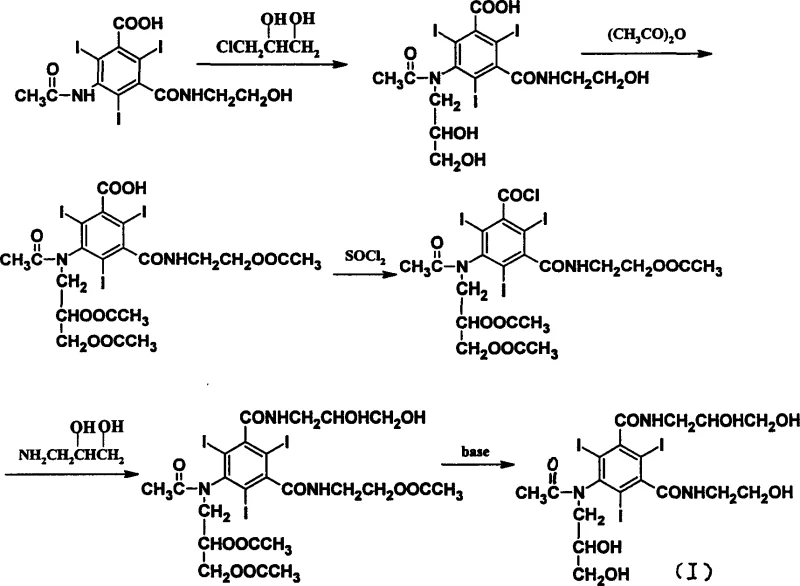
Mechanistic Insights into Optimized Acylation and Alkylation Sequence
The mechanistic superiority of this new route begins with the acylation step, where 5-amino-N-(2-hydroxyethyl)-2,4,6-triiodo-isophthalic acid monoamide reacts with acetic anhydride under perchloric acid catalysis. This step effectively protects the amino group, preventing unwanted side reactions in subsequent stages. The reaction is conducted at moderate temperatures between 50°C and 90°C, ensuring complete conversion without degrading the sensitive iodine-substituted aromatic core. Following acylation, the carboxylic acid moiety is activated to an acyl chloride using thionyl chloride in an ethyl acetate medium. The choice of ethyl acetate is critical; it acts as a diluent that moderates the exothermic nature of the reaction and allows for the use of stoichiometric rather than excess thionyl chloride, thereby minimizing toxic waste generation and simplifying the removal of byproducts through vacuum distillation.
The subsequent amidation with 3-amino-1,2-propanediol introduces the second side chain, followed by a hydrolysis step that removes the acetyl protecting groups to reveal the free hydroxyls necessary for the final structure. The culmination of the synthesis is the N-alkylation step, performed in ethylene glycol monomethyl ether with sodium methylate as the base. Performing this step last is the key to high purity; since the molecule is already largely assembled, the steric and electronic environment favors N-alkylation over O-alkylation. The final product is purified through ion-exchange resin treatment and recrystallization from aqueous ethanol, yielding a white solid with a purity greater than 99.0%. This rigorous control over the reaction mechanism ensures that the high-purity Ioxilan meets the stringent specifications required for injectable contrast media.
How to Synthesize Ioxilan Efficiently
The synthesis of Ioxilan via this optimized route involves four distinct chemical transformations that can be executed sequentially with minimal intermediate isolation. The process begins with the acylation of the starting amine, followed by conversion to the acid chloride, amidation with the diol amine, and concludes with the final N-alkylation. Each step is designed to maximize atom economy and minimize waste, utilizing standard industrial equipment such as three-necked flasks equipped with agitators and reflux condensers. The detailed standardized synthetic steps, including specific molar ratios, temperature profiles, and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency protocol.
- Perform acylation of 5-amino-N-(2-hydroxyethyl)-2,4,6-triiodo-isophthalic acid monoamide using acetic anhydride and perchloric acid catalyst.
- Convert the acylated intermediate to the corresponding acyl chloride using thionyl chloride in ethyl acetate solvent.
- Execute amidation with 3-amino-1,2-propanediol followed by hydrolysis to remove acetyl protecting groups.
- Conduct the final N-alkylation with 3-chloro-1,2-propanediol in the presence of sodium methylate to yield Ioxilan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers substantial strategic benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification process; by avoiding the formation of hard-to-separate O-alkyl impurities, the need for expensive and time-consuming chromatographic separations is eliminated. This directly translates to significant cost savings in manufacturing, as fewer solvents and stationary phases are consumed, and batch cycle times are reduced. Furthermore, the high conversion rates in the final steps mean that raw material utilization is maximized, reducing the overall cost of goods sold (COGS) and enhancing the margin potential for the final API. The ability to produce high-purity material consistently also reduces the risk of batch failures, ensuring a more reliable supply chain for downstream drug formulation.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the reduction in hazardous reagent usage significantly lower operational expenditures. By using ethyl acetate instead of excess thionyl chloride, the process reduces the burden on waste treatment facilities and lowers the cost associated with handling toxic materials. The high yield of the final alkylation step ensures that expensive iodinated precursors are not wasted, providing a clear economic advantage over legacy methods that suffer from lower conversion efficiencies and higher impurity loads.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route, characterized by mild reaction conditions and tolerant catalysts, makes it highly suitable for large-scale production. The reliance on readily available starting materials and common industrial solvents ensures that supply disruptions are minimized. Additionally, the simplified workup procedures allow for faster turnaround times between batches, enabling manufacturers to respond more agilely to market demand fluctuations and reducing lead time for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in thionyl chloride usage and the avoidance of chlorinated solvents in favor of ethyl acetate align with modern green chemistry principles. This facilitates easier regulatory compliance and reduces the environmental footprint of the manufacturing facility. The process is inherently scalable, as demonstrated by the successful transition from laboratory to pilot scales without loss of yield or purity, making it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates to meet global healthcare needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Ioxilan using the patented CN1800149A methodology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield, purity, and process safety. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the new synthesis route improve purity compared to US Patent 4954348?
A: The new route delays the N-alkylation step until the end, avoiding the formation of difficult-to-remove O-alkyl impurities common in earlier methods, resulting in purity greater than 99.0%.
Q: What are the safety advantages of using ethyl acetate in the acyl chloride step?
A: Using ethyl acetate as a solvent significantly reduces the required amount of thionyl chloride, minimizing toxic exposure risks and environmental pollution compared to using thionyl chloride as both reagent and solvent.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions (50-90°C), simplified workup procedures without complex chromatography, and high conversion rates, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ioxilan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality contrast agents in modern diagnostic imaging. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated chemistry required for Ioxilan is executed with precision and consistency. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch meets the >99.0% purity benchmark essential for injectable applications. Our commitment to process optimization allows us to deliver a reliable Ioxilan supplier experience that balances technical excellence with commercial viability.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthetic route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a seamless integration of high-performance contrast media intermediates into your production pipeline.
