Optimizing Ioxilan Production: A Technical Breakthrough in Contrast Agent Manufacturing
Introduction to Advanced Ioxilan Manufacturing
The pharmaceutical landscape for non-ionic X-ray contrast agents demands increasingly rigorous standards for purity, safety, and manufacturing efficiency. A pivotal advancement in this domain is detailed in patent CN100344606C, which outlines a superior preparation method for Ioxilan, chemically known as 5-[acetyl(2,3-dihydroxypropyl)amino]-N-(2,3-dihydroxypropyl)-N'-(2-hydroxyethyl)-2,4,6-triiodo-1,3-benzenedicarboxamide. This compound represents a new generation of contrast media characterized by low osmotic pressure, low viscosity, and excellent biological tolerance, making it ideal for angiography, urography, and CT enhancement. The technical breakthrough lies in a re-engineered synthetic route that starts from 5-amino-N-(2-hydroxyethyl)-2,4,6-triiodoisophthalamic acid, diverging significantly from prior art to offer a more robust, cost-effective, and environmentally friendly pathway suitable for global supply chains.
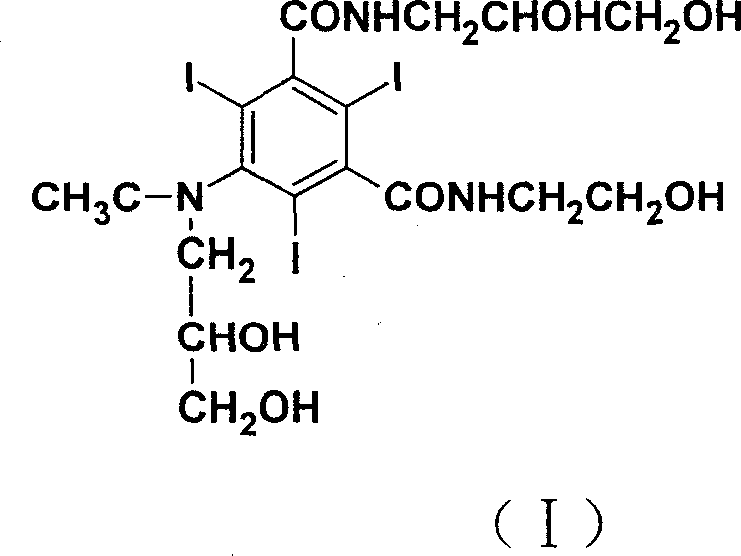
As a reliable pharmaceutical intermediate supplier, understanding the nuances of this synthesis is critical for ensuring the consistent delivery of high-quality active ingredients. The structural complexity of Ioxilan, featuring a heavily iodinated benzene ring and multiple hydrophilic side chains, presents unique challenges in regioselectivity and impurity control. The methodology described in CN100344606C addresses these challenges through a logical sequence of acylation, acyl chlorination, amidation, and a strategically placed final alkylation step. This approach not only enhances the final product's purity to greater than 99.0% but also streamlines the operational workflow, reducing the burden on downstream processing facilities.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the standard industrial synthesis, such as that disclosed in U.S. Patent 4954348, relied on a route that introduced significant inefficiencies and quality risks. The conventional method typically initiates with 5-acetamido-N-(2-hydroxyethyl)-2,4,6-triiodoisophthalamic acid and performs an alkylation reaction early in the sequence using 3-chloro-1,2-propylene glycol. A major drawback of this early alkylation is the competition between N-alkylation and O-alkylation reactions. Experimental data from legacy processes indicates a reaction conversion ratio of only roughly 88%, accompanied by the formation of oxygen alkylate impurities that possess chemical structures very similar to the desired product. These impurities are notoriously difficult to separate and remove, often carrying through subsequent reaction steps and compromising the purity of the final API.
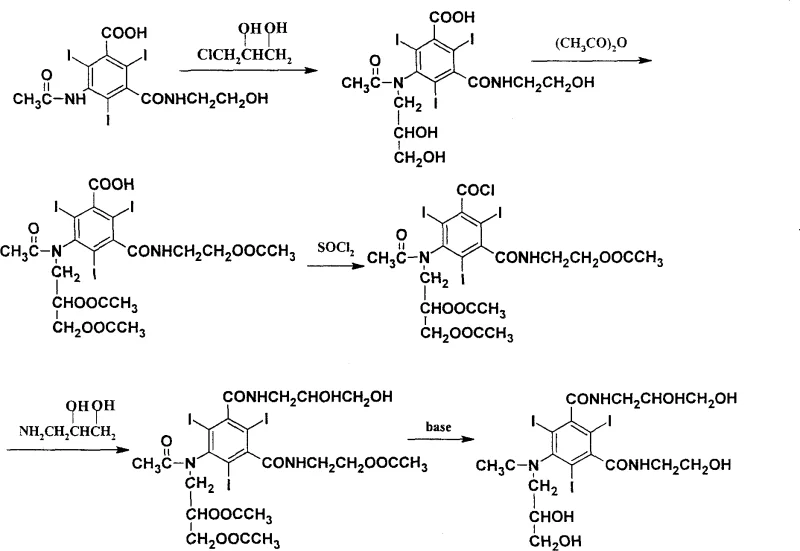
Furthermore, the conventional acyl chloride formation step utilized thionyl chloride in excess, serving simultaneously as both the reactant and the solvent. This practice resulted in excessive consumption of a hazardous reagent, necessitating complex removal procedures under reduced pressure to eliminate large volumes of residual thionyl chloride. The environmental and safety implications of handling such large quantities of corrosive and toxic materials are substantial, posing risks to operator health and increasing the cost of waste treatment. Additionally, the workup procedures in the older methods involved tedious steps such as azeotropic distillation with toluene and multiple extraction cycles, which are energy-intensive and ill-suited for modern, lean manufacturing environments focused on cost reduction in contrast agent manufacturing.
The Novel Approach
The novel approach presented in CN100344606C fundamentally restructures the synthetic logic to overcome these bottlenecks. By shifting the starting material to the free amine (5-amino-N-(2-hydroxyethyl)-2,4,6-triiodoisophthalamic acid) and postponing the critical N-alkylation step to the very end of the synthesis, the process achieves remarkable selectivity. In this optimized route, the alkylation is performed on the fully formed amide backbone using sodium methylate as a base. High-performance liquid chromatography (HPLC) analysis of this final step reveals a reaction conversion ratio reaching 95%, with the problematic oxygen alkylate impurity reduced to less than 1%. This strategic sequencing effectively prevents the carryover of alkylation by-products into the final crystallization, ensuring a much cleaner product profile.
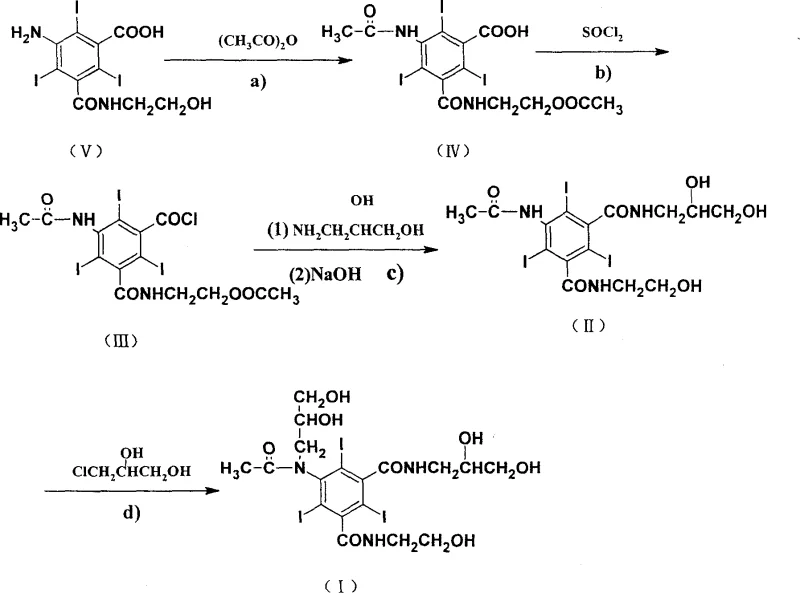
Moreover, the novel method introduces significant improvements in the acyl chloride formation step. Instead of using neat thionyl chloride, the reaction is conducted in ethyl acetate solvent with a controlled molar ratio of compound to thionyl chloride to ethyl acetate of approximately 1:3:19. This modification drastically reduces the absolute amount of thionyl chloride required, thereby mitigating environmental pollution and safety hazards. The workup is simplified to solvent evaporation under reduced pressure, eliminating the need for complex aqueous extractions and azeotropic drying. These refinements collectively result in a process that is not only chemically superior but also operationally simpler, aligning perfectly with the requirements for commercial scale-up of complex iodinated compounds.
Mechanistic Insights into the Optimized Synthetic Sequence
The core of this technological advancement lies in the precise control of reaction conditions across four distinct stages. The process begins with an acylation reaction where the starting amine reacts with acetic anhydride in the presence of a perchloric acid catalyst. The molar ratio is tightly controlled at approximately 1:24:0.03 (amine:anhydride:catalyst), and the reaction proceeds at temperatures between 50°C and 90°C for 10 to 25 hours. This step protects the amine functionality while acetylating the hydroxyl group on the side chain, setting the stage for subsequent transformations without requiring intermediate purification. The simplicity of this step, requiring only solvent evaporation to proceed, highlights the efficiency of the design.
Following acylation, the carboxylic acid moiety is activated via conversion to an acyl chloride. This is achieved using thionyl chloride in ethyl acetate at 50°C to 90°C for 1 to 6 hours. The use of ethyl acetate as a solvent is a critical mechanistic enabler, allowing for better heat transfer and reagent distribution while minimizing the aggressive nature of the reaction medium. The subsequent amidation step involves reacting the acyl chloride with 3-amino-1,2-propanediol in N,N-dimethylacetamide, catalyzed by triethylamine. The reaction temperature is carefully managed between 10°C and 50°C over 8 to 17 hours. Crucially, the deprotection of the acetyl groups occurs concurrently or immediately following this step via the addition of sodium hydroxide, revealing the essential diol functionalities required for the contrast agent's water solubility and low osmolarity.
How to Synthesize Ioxilan Efficiently
The synthesis of Ioxilan via this patented route offers a clear pathway for manufacturers seeking to optimize their production lines. The protocol emphasizes the importance of maintaining strict stoichiometric controls, particularly during the acylation and alkylation phases, to maximize yield and minimize impurity formation. The use of common organic solvents like ethyl acetate and methanol, combined with standard unit operations such as filtration and vacuum drying, ensures that the process is accessible to most fine chemical facilities. For detailed operational parameters, including specific charging orders and temperature ramping profiles, operators should refer to the standardized synthesis guidelines derived from the patent embodiments.
- Perform acylation of the starting amine with acetic anhydride using perchloric acid catalysis at 50-90°C.
- Convert the resulting carboxylic acid to acyl chloride using thionyl chloride in ethyl acetate solvent at 50-90°C.
- Execute amidation with 3-amino-1,2-propanediol followed by hydrolysis to form the diol side chains.
- Conduct final N-alkylation with 3-chloro-1,2-propanediol using sodium methylate to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthesis route translates into tangible strategic benefits beyond mere chemical elegance. The primary advantage is the significant simplification of the manufacturing workflow, which directly impacts operational expenditures. By eliminating complex purification steps such as multiple extractions and azeotropic distillations, the process reduces energy consumption and labor hours per batch. Furthermore, the reduction in thionyl chloride usage lowers the cost of raw materials and decreases the financial liability associated with hazardous waste disposal. These factors combine to create a more economically viable production model that can withstand market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive and hazardous reagents in excess quantities. Specifically, the substitution of neat thionyl chloride with a diluted solution in ethyl acetate reduces reagent costs and the associated costs of neutralizing and disposing of acidic waste streams. Additionally, the high conversion rates in the final alkylation step (>95%) mean that less starting material is wasted, improving the overall atom economy of the synthesis. This efficiency allows for a more competitive pricing structure for the final high-purity Ioxilan intermediate, providing a buffer against margin compression in the generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The robustness of the new synthetic route enhances supply chain stability by reducing the risk of batch failures due to impurity buildup. In the legacy process, the accumulation of O-alkylated impurities often necessitated re-processing or scrapping of batches, leading to unpredictable lead times. With the new method's ability to consistently achieve purity levels greater than 99.0%, manufacturers can offer more reliable delivery schedules. The use of readily available solvents and reagents further insulates the supply chain from shortages of specialized chemicals, ensuring reducing lead time for high-purity contrast media projects.
- Scalability and Environmental Compliance: The mild reaction conditions (mostly below 90°C) and the absence of extreme pressure requirements make this process highly scalable from pilot plant to multi-ton commercial production. The reduced environmental footprint, driven by lower thionyl chloride emissions and simplified waste streams, facilitates easier compliance with increasingly stringent environmental regulations (EHS). This compliance is a critical factor for long-term supply continuity, as it minimizes the risk of production shutdowns due to regulatory violations, thereby securing the supply line for downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Ioxilan using the methodology described in CN100344606C. These insights are derived directly from the patent's experimental data and comparative analysis, providing a factual basis for decision-making. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing portfolios.
Q: How does the new Ioxilan synthesis method improve purity compared to US Pat 4954348?
A: The new method delays the N-alkylation step to the final stage, achieving a conversion ratio of over 95% with less than 1% O-alkylation impurity, whereas the older method produced difficult-to-remove oxygen alkylate impurities early in the sequence.
Q: What are the environmental benefits of the acyl chloride reaction in this process?
A: By using ethyl acetate as a solvent rather than excess thionyl chloride acting as both reactant and solvent, the process significantly reduces the consumption of thionyl chloride, minimizing hazardous waste and environmental pollution.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process features mild reaction conditions (50-90°C), simplified workup procedures involving solvent evaporation rather than complex extractions, and high yields (85-86%), making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ioxilan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of safe and effective diagnostic agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to industrial reality is seamless. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to verify that every batch of Ioxilan meets the exacting standards required for injectable pharmaceutical applications. Our infrastructure is designed to handle complex iodinated chemistry safely and efficiently.
We invite global pharmaceutical partners to collaborate with us to leverage these advanced manufacturing capabilities. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how our optimized Ioxilan synthesis can enhance your supply chain resilience and product quality. Let us be your trusted partner in delivering the next generation of contrast media to the market.
