Advanced Industrial Synthesis of High-Purity Beta-Ionone for Global Supply Chains
Advanced Industrial Synthesis of High-Purity Beta-Ionone for Global Supply Chains
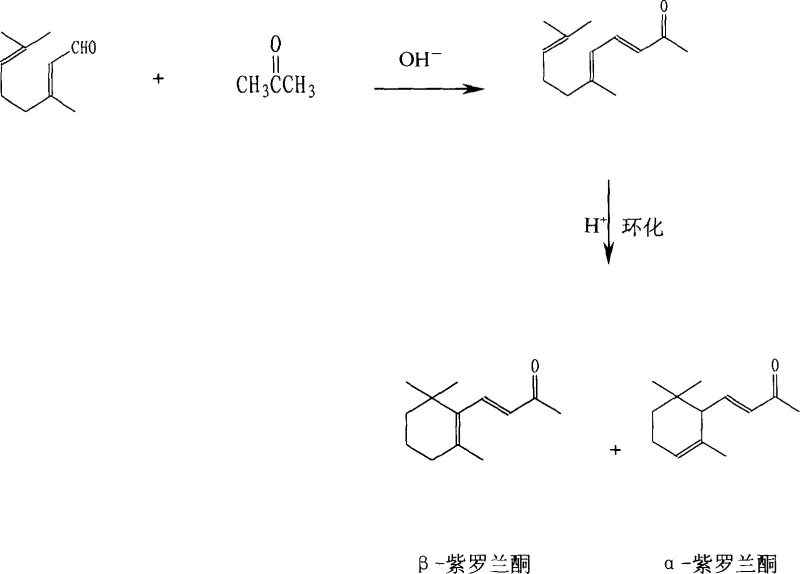
The global demand for high-purity fragrance intermediates and vitamin precursors necessitates robust, scalable synthetic routes that prioritize both yield and isomeric purity. Patent CN1508113A introduces a transformative industrial preparation method for beta-ionone that addresses critical bottlenecks in the traditional citral-acetone condensation and subsequent cyclization processes. By fundamentally altering the reaction environment from a heterogeneous two-phase system to a near-homogeneous phase through precise adjustment of acetone, water, and alkali ratios, this technology achieves reaction conditions that are significantly milder, typically between 18°C and 28°C, compared to the harsh 60°C to 70°C required by conventional methods. Furthermore, the innovation incorporates a novel thermal management strategy during the cyclization step, utilizing dry ice to absorb exothermic heat and prevent local overheating, thereby driving the selectivity of beta-ionone to exceed 99.0 percent while suppressing the formation of the less desirable alpha-isomer to below 1.0 percent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of ionones has been plagued by inefficiencies inherent in traditional base-catalyzed condensation and acid-catalyzed cyclization steps. Conventional protocols typically employ a 10 percent aqueous sodium hydroxide solution as a catalyst, necessitating reaction temperatures of 60°C to 70°C over extended periods of 5 to 6 hours to drive the conversion of citral to pseudoionone. These elevated temperatures are detrimental because citral is highly sensitive to thermal stress; under alkaline conditions with heat, citral is prone to self-condensation and polymerization, which drastically reduces the overall yield to a mediocre range of 60 percent to 70 percent. Additionally, previous attempts to mitigate these issues, such as the introduction of phase-transfer catalysts or specific condensation inhibitors described in other patents like CN 1109462A, have introduced significant economic burdens due to the high cost of these additives and the complexity of their recovery, rendering them less viable for large-scale cost-sensitive manufacturing.
The Novel Approach
The methodology outlined in CN1508113A represents a paradigm shift by optimizing the solvent and catalyst ratios to facilitate a near-homogeneous reaction environment, which dramatically accelerates the reaction kinetics without the need for excessive thermal energy. This allows the condensation of citral and acetone to proceed efficiently at ambient-like temperatures of 18°C to 28°C, shortening the reaction time to just 2 to 4 hours while boosting the molar yield of pseudoionone to nearly 90 percent. In the subsequent cyclization stage, the process ingeniously utilizes the sublimation of dry ice (solid carbon dioxide) to act as an internal cooling and stirring mechanism. This effectively dissipates the intense heat generated during the acid-catalyzed ring closure, preventing the thermal isomerization that typically favors the alpha-form, and ensuring that the final product stream is dominated by the high-value beta-isomer with exceptional purity profiles suitable for downstream pharmaceutical applications.
Mechanistic Insights into Optimized Aldol Condensation and Thermal-Controlled Cyclization
The core chemical advancement lies in the manipulation of phase behavior during the initial aldol condensation. In traditional systems, the immiscibility of the organic citral and the aqueous alkali catalyst creates a mass transfer limitation, requiring high temperatures to overcome the activation energy barrier at the interface. By carefully tuning the volume ratio of acetone to water and the weight ratio of sodium hydroxide, the patent describes a system where the reactants exist in a single, near-homogeneous phase. This maximizes the molecular collision frequency between the enolate of acetone and the aldehyde group of citral, allowing the reaction to proceed rapidly at lower temperatures where side reactions like self-polymerization of citral are kinetically suppressed. This mechanistic optimization not only preserves the integrity of the sensitive citral starting material but also simplifies the workup procedure by reducing the formation of tarry byproducts that complicate purification.
Following the isolation of pseudoionone, the cyclization mechanism is critically dependent on thermal management to control regioselectivity. The acid-catalyzed ring closure of pseudoionone to form ionones is highly exothermic, and without rigorous temperature control, the local heat spikes promote the thermodynamic equilibration towards the alpha-isomer or the formation of polymeric residues. The introduction of dry ice serves a dual mechanistic purpose: firstly, the phase change from solid to gas absorbs a substantial amount of latent heat, acting as an efficient heat sink that maintains the bulk reaction temperature at cryogenic levels (around -18°C to -12°C); secondly, the vigorous evolution of carbon dioxide gas provides mechanical agitation that ensures uniform mixing of the pseudoionone and the sulfuric acid catalyst. This precise thermal regulation kinetically traps the reaction pathway favoring the formation of the beta-ionone structure, achieving a selectivity greater than 99.0 percent, which is a significant improvement over the 3 percent to 5 percent alpha-isomer contamination seen in standard low-temperature processes.
How to Synthesize Beta-Ionone Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for manufacturers seeking to upgrade their production lines for high-purity ionones. The process begins with the preparation of the homogeneous condensation mixture, followed by a controlled addition of citral to manage the exotherm, and concludes with a specialized cyclization workup involving dry ice. This streamlined approach eliminates the need for exotic catalysts while maximizing the throughput of the reactor vessels. For R&D teams looking to implement this technology, the following guide outlines the critical operational parameters derived from the patent examples, emphasizing the importance of temperature monitoring and reagent stoichiometry to replicate the high yields and selectivities reported.
- Perform aldol condensation of citral and acetone using adjusted NaOH/water ratios at 18-28°C to achieve a near-homogeneous phase and high pseudoionone yield.
- Distill the crude pseudoionone under reduced pressure to isolate the intermediate with content greater than 90 percent.
- Execute acid-catalyzed cyclization of pseudoionone while simultaneously adding dry ice to prevent local overheating and ensure beta-isomer selectivity greater than 99 percent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into enhanced operational efficiency and reduced cost of goods sold (COGS) without compromising on quality standards. The elimination of expensive phase-transfer catalysts and transposition inhibitors, which are difficult to recover and recycle in traditional methods, removes a significant variable cost from the bill of materials. Furthermore, the drastic reduction in reaction time from roughly 6 hours to under 4 hours in the condensation step increases the asset turnover rate of the production facility, allowing for more batches to be produced within the same timeframe, effectively expanding capacity without capital expenditure on new reactors. The robustness of the process against thermal runaway also reduces the risk of batch failures, ensuring a more consistent and reliable supply of this critical intermediate for global clients.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the dependency on high-cost specialty additives such as phase-transfer catalysts and specific transposition preventers that were required in older patent literature. By relying on commodity chemicals like acetone, sodium hydroxide, and dry ice, the raw material costs are minimized, and the simplified downstream processing reduces utility consumption associated with prolonged heating and complex purification steps. The higher overall yield, reaching up to 85 percent, means that less raw citral is wasted per kilogram of finished beta-ionone, directly improving the gross margin profile for manufacturers adopting this route.
- Enhanced Supply Chain Reliability: The shift to milder reaction conditions and the use of readily available reagents enhances the resilience of the supply chain against raw material volatility. Since the process does not rely on niche catalysts that may have long lead times or single-source suppliers, procurement teams can secure materials more easily and maintain continuous production schedules. The shortened cycle times also mean that inventory turnover is faster, allowing the supply chain to respond more agilely to fluctuations in market demand for vitamin A precursors and fine fragrances without building up excessive safety stock.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process offers distinct advantages by simplifying waste streams and reducing energy intensity. The near-homogeneous condensation reduces the formation of heavy tars and polymeric sludge, easing the burden on wastewater treatment facilities and lowering disposal costs. Additionally, the use of dry ice as a cooling agent avoids the need for energy-intensive refrigeration cycles to maintain low temperatures during cyclization, aligning with green chemistry principles and helping facilities meet increasingly stringent environmental regulations regarding energy consumption and emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this high-purity beta-ionone synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on performance metrics and operational requirements for potential licensees or manufacturing partners.
Q: How does the new process improve beta-ionone selectivity compared to traditional methods?
A: Traditional cyclization often suffers from local overheating due to the exothermic nature of the reaction, leading to higher alpha-isomer content. This patented method introduces dry ice during cyclization, which sublimates to absorb heat and provide internal stirring, maintaining low temperatures that favor beta-isomer formation with selectivity exceeding 99.0 percent.
Q: What are the purity specifications achievable with this industrial method?
A: The optimized process yields a final product containing greater than or equal to 96.0 percent beta-ionone, with alpha-ionone content suppressed to less than or equal to 1.0 percent, meeting stringent requirements for vitamin A synthesis and high-end fragrance applications.
Q: Why is the condensation step performed at lower temperatures in this protocol?
A: By adjusting the acetone, water, and alkali ratios to create a near-homogeneous reaction system, the reaction rate is significantly accelerated. This allows the condensation to proceed efficiently at mild temperatures between 18°C and 28°C, preventing thermal degradation of citral and avoiding self-condensation side reactions common at higher temperatures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity beta-ionone plays in the synthesis of Vitamin A and the formulation of premium fragrances. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated thermal control and homogeneous reaction conditions described in advanced patents are meticulously replicated at an industrial scale. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch meets the exacting standards required by the pharmaceutical and fine chemical industries, with beta-isomer content consistently maintained above 96 percent.
We invite global partners to collaborate with us to leverage this advanced manufacturing technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized process can reduce your total landed cost. We encourage you to contact us today to obtain specific COA data from our recent pilot runs and to discuss route feasibility assessments that can accelerate your product development timelines and secure your position in the competitive fine chemicals market.
