Advanced Hydrophobic Catalysis for Commercial Scale Beta-Ionone Manufacturing
Advanced Hydrophobic Catalysis for Commercial Scale Beta-Ionone Manufacturing
The global demand for high-purity beta-ionone, a critical precursor for Vitamin A and E synthesis as well as a valuable fragrance ingredient, necessitates manufacturing processes that balance yield with environmental sustainability. Patent CN113117753B introduces a groundbreaking approach utilizing a novel hydrophobic catalyst to facilitate the indirect acid-catalyzed cyclization of pseudoionone. This technology represents a significant departure from conventional direct acid catalysis, offering a pathway that mitigates the severe heat transfer limitations and wastewater generation typically associated with sulfuric acid methods. By employing a macrocyclic ligand complexed with alkaline earth metals such as Magnesium, Calcium, Strontium, or Barium, the process achieves high conversion rates under remarkably mild conditions.
This innovation addresses the long-standing challenges in ionone production, specifically the difficulty in controlling exothermic reactions and the formation of unwanted isomers. The proprietary catalyst structure allows for precise modulation of the reaction environment, ensuring that the transformation from pseudoionone to beta-ionone proceeds with exceptional selectivity. For industrial stakeholders, this translates to a robust manufacturing protocol that reduces reliance on extreme cooling measures and simplifies downstream purification, thereby enhancing the overall viability of large-scale operations in the fine chemical sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis of beta-ionone typically relies on the direct cyclization of pseudoionone using concentrated sulfuric acid, a method fraught with significant engineering and environmental hurdles. The direct acid-catalyzed reaction is characterized by rapid and intense heat release, coupled with a sharp increase in system viscosity, which creates severe mass transfer and heat transfer bottlenecks. To manage this exothermicity, existing industrial processes often require specialized equipment capable of withstanding harsh corrosive conditions and complex temperature control systems, such as the addition of dry ice, which drastically escalates operational costs. Furthermore, the post-reaction workup involves diluting large quantities of sulfuric acid, resulting in substantial volumes of acidic wastewater with high Chemical Oxygen Demand (COD), posing a heavy burden on waste treatment facilities and regulatory compliance.
The Novel Approach
In stark contrast, the methodology disclosed in CN113117753B utilizes a sophisticated hydrophobic catalyst to enable an indirect reaction pathway that circumvents these thermal and environmental pitfalls. Instead of direct protonation by strong mineral acids, the process first forms a stable sulfonate intermediate through the interaction of pseudoionone with bisulfite, mediated by the hydrophobic catalyst. This intermediate is subsequently cyclized using mild organic acids rather than concentrated sulfuric acid, effectively eliminating the generation of sulfuric acid wastewater. The hydrophobic nature of the catalyst ensures efficient phase separation and recyclability, while the milder reaction conditions (typically between 75°C and 100°C) significantly lower the energy input required for cooling and agitation, presenting a far more sustainable and economically attractive alternative for modern chemical manufacturing.
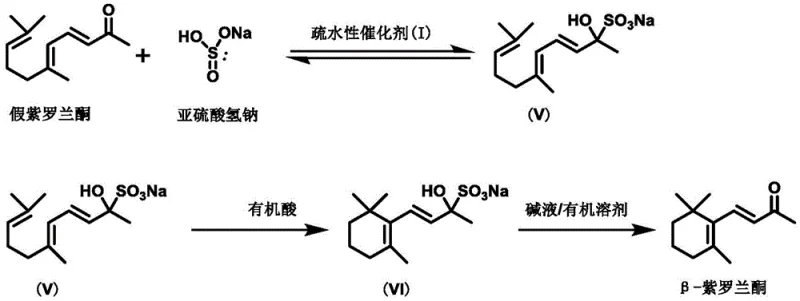
Mechanistic Insights into Hydrophobic Catalyst-Mediated Cyclization
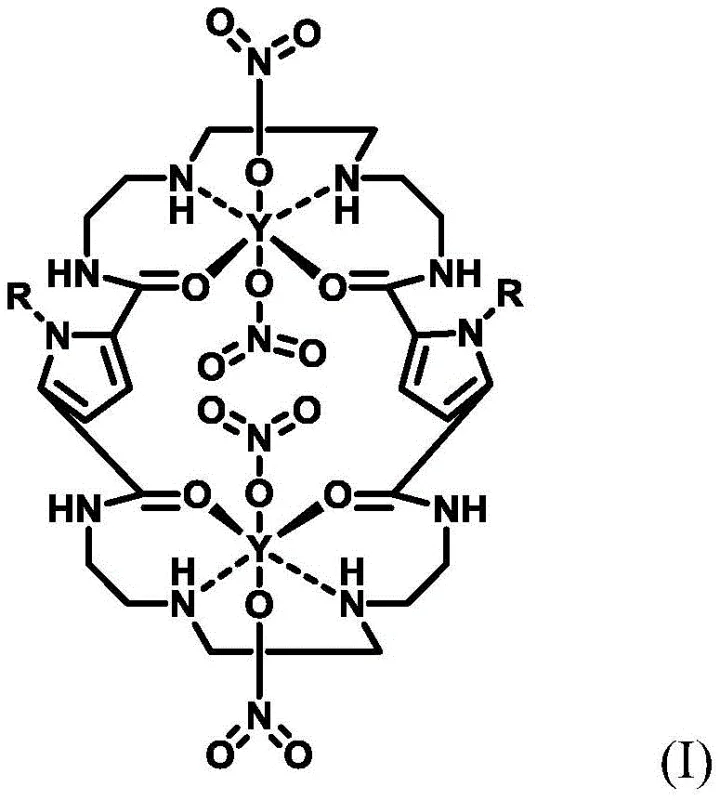
The core of this technological advancement lies in the unique structural design of the hydrophobic catalyst, which features a macrocyclic framework coordinated with metal ions like Mg, Ca, Sr, or Ba. This architecture creates a specific microenvironment that facilitates the nucleophilic attack of the bisulfite ion on the pseudoionone molecule, stabilizing the transition state and preventing premature polymerization or degradation. The catalyst's hydrophobic exterior promotes compatibility with organic solvents while maintaining the necessary activity to drive the formation of the sulfonate adduct (Intermediate V). This step is crucial as it locks the pseudoionone into a configuration that is primed for cyclization, effectively bypassing the chaotic mixture of isomers often seen in uncontrolled acid environments.
Following the formation of the sulfonate intermediate, the mechanism proceeds through an organic acid-catalyzed cyclization to form Intermediate VI, followed by an alkaline elimination step to yield the final beta-ionone product. The use of organic acids for the cyclization step is particularly advantageous as it provides a controlled proton source that minimizes side reactions such as double bond isomerization or resinification. The catalyst remains stable throughout these transitions, allowing for its recovery and reuse, which is a critical factor in maintaining consistent batch-to-batch quality. This mechanistic precision ensures that the impurity profile of the final product is significantly cleaner, reducing the load on distillation columns and improving the overall yield of the target beta-isomer.
How to Synthesize Beta-Ionone Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology, starting with the preparation of the catalyst itself and proceeding through the three-stage conversion of pseudoionone. The process is designed to be operationally simple, requiring standard reactor setups without the need for exotic high-pressure or cryogenic equipment. The initial step involves reacting pseudoionone with a bisulfite solution in the presence of the catalyst to generate the water-soluble sulfonate intermediate, which naturally separates from the organic phase. Subsequent treatment with organic acid induces the ring closure, and final basification releases the free ketone. Detailed standardized synthesis steps see the guide below.
- React pseudoionone with bisulfite in the presence of the hydrophobic catalyst to form the sulfonate intermediate.
- Treat the intermediate with an organic acid to induce cyclization into the cyclic sulfonate structure.
- Apply alkaline conditions to eliminate the sulfonate group and finalize the beta-ionone structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this hydrophobic catalyst technology offers compelling strategic benefits centered around cost stability and operational reliability. By shifting away from the volatile and hazardous handling of concentrated sulfuric acid and cryogenic cooling agents, manufacturers can significantly reduce their exposure to safety incidents and regulatory fines. The ability to operate at moderate temperatures reduces the strain on utility infrastructure, leading to lower energy consumption per kilogram of product. Moreover, the elimination of sulfuric acid wastewater removes a major cost center associated with neutralization and disposal, directly improving the margin profile of the final beta-ionone product.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive cooling agents like dry ice and reduces the capital expenditure required for specialized corrosion-resistant reactors. By utilizing recyclable catalysts and organic acids, the recurring cost of raw materials is optimized, and the expense associated with treating highly acidic effluent is completely removed, leading to substantial long-term savings in operational expenditure.
- Enhanced Supply Chain Reliability: The mild operating conditions and reduced viscosity of the reaction mixture allow for more consistent batch cycles and shorter turnaround times. This reliability ensures a steady flow of high-purity intermediates, minimizing the risk of production delays caused by equipment fouling or thermal runaway events, thereby securing the supply continuity for downstream vitamin and fragrance customers.
- Scalability and Environmental Compliance: The technology is inherently scalable due to its lower heat release power and simplified workup procedures, making it ideal for expansion from pilot to commercial scale. Furthermore, the green chemistry principles embedded in the process, such as wastewater reduction and catalyst recycling, align perfectly with increasingly stringent global environmental regulations, future-proofing the manufacturing asset against tightening compliance standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydrophobic catalytic system. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, providing a factual basis for evaluating the technology's fit within your existing production portfolio. Understanding these nuances is essential for making informed decisions about process integration and supplier qualification.
Q: How does this hydrophobic catalyst improve upon traditional sulfuric acid methods?
A: Unlike traditional methods that generate large volumes of acidic wastewater and require harsh heat management, this catalyst enables an indirect pathway using organic acids, significantly reducing environmental impact and equipment corrosion risks.
Q: What are the key operational advantages for large-scale production?
A: The process operates under mild temperatures (75-100°C) and avoids the extreme viscosity and heat release associated with direct acid catalysis, allowing for safer scale-up and reduced cooling costs.
Q: Is the catalyst reusable in this synthesis pathway?
A: Yes, the hydrophobic nature of the catalyst allows for effective separation and recycling, which enhances the overall economic efficiency and sustainability of the manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Ionone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN113117753B for the production of high-value fragrance and pharmaceutical intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory concepts are successfully translated into robust industrial realities. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of beta-ionone meets the exacting standards required by the global flavors and vitamins industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis route for your supply chain needs. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this hydrophobic catalyst method can optimize your total cost of ownership. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and competitive supply of high-purity beta-ionone.
