Advanced Synthesis of Cyclopropyl Substituted 2H-Benzopyran Derivatives for Commercial Scale Production
Advanced Synthesis of Cyclopropyl Substituted 2H-Benzopyran Derivatives for Commercial Scale Production
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex small molecules, particularly those targeting inflammatory pathways. Patent CN116396330A introduces a significant advancement in the preparation of cyclopropyl substituted 2H-benzopyran derivatives, specifically the bis-tromethamine salt of a potent CRAC channel inhibitor known as Compound IV. This compound has demonstrated remarkable inhibition effects on calcium release-activated calcium channels and inflammatory cells, showing excellent potential for treating acute membrane adenitis. The disclosed methodology addresses critical bottlenecks in previous synthetic routes, offering a pathway that ensures high purity, consistent crystal form, and enhanced operational safety. For R&D directors and procurement specialists, understanding these technical nuances is vital for securing a reliable supply chain of high-quality active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art routes, such as those described in WO2021057890A1 and WO2022179577A1, presented substantial challenges for large-scale manufacturing. A primary issue was the instability of the intermediate, Compound III. In conventional processes, the post-treatment involved concentration steps that led to obvious degradation of Compound III, significantly increasing impurity levels and complicating purification. Furthermore, the salt formation reaction to produce Compound IV was prone to an "explosion phenomenon," where rapid precipitation made stirring difficult and caused impurity entrapment. This often resulted in a mixed crystal form, necessitating additional, time-consuming crystal transformation steps that further degraded product quality and introduced static electricity issues during drying.
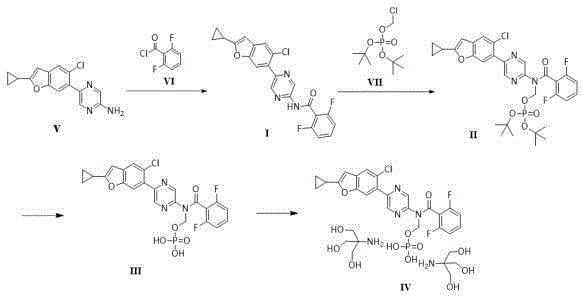
The Novel Approach
The innovative process detailed in CN116396330A fundamentally restructures the synthesis to mitigate these risks. By utilizing a specific acidic hydrolysis system comprising citric acid and disodium hydrogen phosphate, the method achieves efficient conversion under mild conditions. Crucially, the post-treatment employs solvents like 2-methyltetrahydrofuran to allow for direct transition to the next step without concentration, thereby preserving the integrity of Compound III. The salt formation is meticulously controlled by maintaining a fully-dissolved state at elevated temperatures before inducing crystallization with seed crystals. This strategic adjustment ensures the exclusive formation of the target single crystal form, eliminating the need for polymorph conversion and drastically simplifying the isolation process.
Mechanistic Insights into Acidic Hydrolysis and Controlled Crystallization
The core of this technological breakthrough lies in the precise control of chemical equilibrium and phase transitions. The hydrolysis of Compound II is conducted in a buffered acidic environment (pH 2.0-5.0), which facilitates the cleavage of the protecting group while minimizing side reactions. The selection of acetonitrile as the reaction solvent R-1 optimizes solubility and reaction kinetics at temperatures between 30-60°C. Following hydrolysis, the extraction protocol is designed to remove acidic impurities while retaining Compound III in the aqueous phase, which is then back-extracted into an organic phase suitable for the subsequent salt formation. This multi-step extraction strategy effectively purifies the intermediate in situ, reducing the burden on final purification.
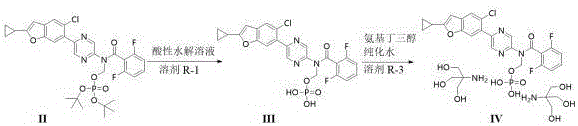
Impurity control is further enhanced during the final salt formation stage. By heating the reaction mixture of Compound III and tromethamine to 40-50°C, the system remains homogeneous, preventing the premature nucleation that leads to mixed crystals or amorphous solids. The introduction of seed crystals of the target crystal form acts as a template, directing the molecular assembly into the desired lattice structure. This seeded crystallization not only guarantees polymorphic purity but also results in a product with superior physical properties, such as reduced static electricity and improved flowability. These characteristics are essential for downstream processing, including tableting or capsule filling, ensuring that the final drug product meets stringent regulatory specifications.
How to Synthesize Cyclopropyl Substituted 2H-Benzopyran Derivative Efficiently
The synthesis of this high-value pharmaceutical intermediate requires strict adherence to the optimized parameters regarding temperature, solvent ratios, and seeding timing. The process begins with the hydrolysis of the protected precursor, followed by a specialized workup that avoids thermal stress on the sensitive intermediate. The final step involves a controlled salt formation where the maintenance of a clear solution prior to seeding is critical for success. Operators must ensure that the system is fully dissolved before introducing the seed crystals to guarantee the formation of the correct polymorph. For a detailed breakdown of the standardized operating procedures and specific reagent quantities, please refer to the technical guide below.
- Hydrolyze Compound II in an acidic solution (citric acid/disodium hydrogen phosphate, pH 2.0-5.0) and solvent R-1 at 30-60°C.
- Perform post-treatment using solvent R-2 (e.g., 2-methyltetrahydrofuran) to extract impurities without concentrating the solution to prevent degradation.
- React the resulting Compound III solution directly with tromethamine in purified water and solvent R-3 at 40-50°C, adding seed crystals to induce single-form crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel manufacturing process offers tangible strategic benefits beyond mere technical compliance. The elimination of unstable intermediate handling and the removal of complex crystal transformation steps translate directly into a more resilient supply chain. By simplifying the workflow, manufacturers can reduce the risk of batch failures and ensure consistent delivery schedules, which is paramount for maintaining continuous drug production lines. The robustness of the new method against common scale-up pitfalls, such as agitation issues and solvent retention, further solidifies its value proposition for long-term commercial partnerships.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for concentration steps that previously caused material loss due to degradation. By avoiding the crystal transformation operation, the overall processing time is significantly reduced, leading to lower utility consumption and labor costs. Furthermore, the simplified workup reduces solvent usage and waste generation, contributing to substantial cost savings in raw material procurement and environmental compliance management.
- Enhanced Supply Chain Reliability: The improved stability of the intermediate and the predictable nature of the seeded crystallization process minimize the risk of off-spec batches. This reliability ensures that suppliers can meet tight delivery windows without the delays associated with re-processing or failed quality control tests. The ability to consistently produce a single crystal form reduces the variability in the supply of the active ingredient, allowing downstream formulation teams to plan with greater confidence.
- Scalability and Environmental Compliance: The avoidance of the "explosion phenomenon" during salt formation makes the process inherently safer and easier to scale from pilot plant to commercial production. The reduced generation of degradation impurities lowers the load on wastewater treatment systems, aligning with increasingly strict environmental regulations. Additionally, the improved physical properties of the final powder facilitate easier handling and packaging, reducing logistical friction in the distribution network.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. Understanding these details helps stakeholders evaluate the feasibility of adopting this method for their specific manufacturing needs. The answers are derived directly from the experimental data and comparative examples provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this new method improve the stability of Compound III compared to prior art?
A: The new method avoids the concentration operation after extraction. In previous methods, concentrating the solution caused significant degradation of the unstable Compound III. By using 2-methyltetrahydrofuran and proceeding directly to the next step, degradation is minimized.
Q: What mechanism prevents the formation of mixed crystal forms in Compound IV?
A: The process controls the reaction temperature to keep the system in a fully-dissolved state before adding specific seed crystals. This induces directional crystallization of the target single crystal form, eliminating the risk of mixed crystals found in older methods.
Q: Why is this process considered safer for industrial scale-up?
A: The optimized salt formation conditions prevent the 'explosion phenomenon' (rapid, uncontrolled precipitation) observed in prior art. This ensures smooth stirring, better heat transfer, and avoids the safety risks associated with heterogeneous systems during amplification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropyl Substituted 2H-Benzopyran Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation anti-inflammatory therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market availability is seamless. We are committed to delivering compounds with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify crystal form and impurity profiles. Our capability to implement complex chemistries, such as the acidic hydrolysis and seeded crystallization described in CN116396330A, positions us as a strategic partner for your API development needs.
We invite you to engage with our technical team to discuss how this optimized synthesis can benefit your project timeline and budget. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of this route compared to traditional methods. We encourage potential partners to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
