Revolutionizing Cyclopropane Derivative Production: A Scalable, Metal-Free Thermal Route for Global Supply Chains
The landscape of organic synthesis for strained ring systems is undergoing a significant paradigm shift, driven by the urgent need for safer, more scalable, and environmentally benign manufacturing processes. Patent CN114436731B introduces a groundbreaking preparation method for cyclopropane derivatives that fundamentally alters the economic and technical feasibility of producing these high-value intermediates. Cyclopropane motifs are ubiquitous in modern medicinal chemistry, serving as critical bioisosteres that enhance metabolic stability and binding affinity in numerous active pharmaceutical ingredients (APIs). However, their historical synthesis has been plagued by complex catalytic cycles and hazardous reagents. This new technology leverages a thermal denitrification strategy starting from pyrazoline compounds, offering a direct pathway to high-purity cyclopropane derivatives without the burden of transition metal contamination. For R&D directors and procurement strategists alike, this represents a pivotal opportunity to optimize cost structures while securing a more resilient supply chain for essential chemical building blocks.
The strategic importance of this innovation cannot be overstated in the context of global pharmaceutical manufacturing. Traditional routes often rely on precious metals or toxic reagents that create bottlenecks in regulatory approval and waste management. By transitioning to a base-catalyzed thermal process, manufacturers can drastically simplify their operational protocols. The patent details a meticulous temperature-controlled distillation process that ensures maximum conversion of raw materials into the target product, achieving yields that consistently surpass industry standards. This level of efficiency translates directly into reduced raw material consumption and lower overall production costs, making it an attractive option for cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the elimination of heavy metals simplifies the purification train, reducing the lead time for high-purity cyclopropane derivatives to reach the market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the cyclopropane ring has been one of the most challenging tasks in synthetic organic chemistry, often requiring harsh conditions that are ill-suited for large-scale industrial application. Conventional methodologies such as the Simmons-Smith-Furukawa reaction necessitate the use of iodine-zinc reagents, which are not only expensive but also generate substantial amounts of zinc waste that requires costly disposal. Similarly, metal-promoted azo ester intercalation and Corey-Chaykovsky cyclopropanation reactions demand strict anhydrous and anaerobic environments, imposing severe constraints on reactor design and operational safety. The use of diazomethane or diazonium reagents in other traditional pathways introduces extreme safety hazards due to their explosive nature, creating significant liability and insurance costs for manufacturing facilities. Moreover, these legacy processes frequently suffer from yields below 90%, leading to significant wastage of valuable starting materials and complicating the isolation of the final product due to complex impurity profiles.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN114436731B offers a streamlined, metal-free alternative that circumvents these historical bottlenecks through a clever thermal denitrification mechanism. By utilizing pyrazoline compounds as precursors, the process exploits the inherent instability of the nitrogen-nitrogen bond under elevated temperatures to drive the formation of the strained three-membered carbon ring. This approach operates under normal atmospheric conditions, removing the need for specialized high-pressure equipment or inert gas manifolds. The reaction is catalyzed by inexpensive and readily available bases such as potassium hydroxide or sodium hydroxide, which are far more economical than transition metal complexes. The result is a robust synthesis capable of achieving yields greater than 95%, with the added benefit of generating nitrogen gas as the primary byproduct, thereby minimizing environmental impact and waste treatment costs. This simplicity makes it an ideal candidate for the commercial scale-up of complex polymer additives and pharmaceutical intermediates.
Mechanistic Insights into Thermal Denitrification Cyclization
The core of this technological breakthrough lies in the precise control of thermal energy to facilitate the extrusion of nitrogen from the pyrazoline ring system. The reaction mechanism involves the homolytic or heterolytic cleavage of the N-N bond within the pyrazoline structure, generating a reactive intermediate that rapidly collapses to form the thermodynamically stable cyclopropane ring. This transformation is highly sensitive to the presence of moisture, which can deactivate the basic catalyst or promote hydrolysis side reactions. Therefore, the patent emphasizes a rigorous pre-drying step where the pyrazoline starting material is treated with a desiccant to ensure water content is minimized before the reaction commences. This attention to detail in the pre-reaction phase is critical for maintaining catalyst activity throughout the prolonged heating cycle, ensuring that the reaction kinetics remain favorable for the formation of the desired cyclopropane derivative rather than decomposition products.
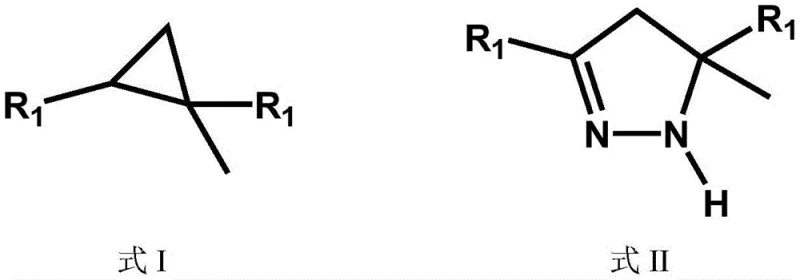
Furthermore, the process employs a sophisticated multi-stage heating profile to manage the reaction equilibrium and product distillation effectively. Initially, the reaction mixture is heated to a moderate temperature range of 180-190°C to initiate the reaction and remove low-boiling impurities. Subsequently, the temperature is carefully ramped to 200-210°C and held for an extended period to allow the bulk of the conversion to occur while distilling off the product as it forms. This continuous removal of the product from the reaction zone helps to drive the equilibrium forward according to Le Chatelier's principle. Finally, a second heating stage at 230-240°C ensures that any residual starting material is fully converted, maximizing the overall yield. This staged approach prevents the rapid evolution of gas that could lead to foaming or entrainment of raw materials, thereby preserving the integrity of the final product and ensuring a clean impurity profile suitable for sensitive pharmaceutical applications.
How to Synthesize Cyclopropane Derivative Efficiently
The synthesis of these valuable intermediates requires strict adherence to the thermal profiling and drying protocols outlined in the patent to achieve optimal results. The process begins with the careful preparation of the pyrazoline precursor, followed by a controlled addition of the base catalyst under stirring conditions. The subsequent heating phases must be managed with precision to balance reaction rate with product quality. For detailed operational parameters and specific stoichiometric ratios, please refer to the standardized synthesis guide below.
- Dry the pyrazoline compound by mixing with a drying agent (KOH/NaOH), stirring, and separating the upper liquid phase to remove moisture.
- Add a basic catalyst to the dried pyrazoline and heat the mixture to 180-190°C to initiate distillation of low-boiling impurities.
- Perform staged heating: maintain at 200-210°C until condensation stops, then ramp slowly to 230-240°C to collect the final target product condensate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this thermal denitrification technology offers profound advantages for procurement managers and supply chain directors seeking to optimize their sourcing strategies. The elimination of expensive transition metal catalysts and hazardous diazo reagents results in a drastic simplification of the raw material supply chain, reducing dependency on volatile commodity markets for specialty metals. This shift not only lowers the direct cost of goods sold but also mitigates the risk of supply disruptions caused by geopolitical factors affecting mining and refining industries. Additionally, the simplified waste stream, consisting primarily of benign salts and nitrogen gas, significantly reduces the environmental compliance burden and associated disposal fees, contributing to substantial cost savings in long-term manufacturing operations.
- Cost Reduction in Manufacturing: The replacement of precious metal catalysts with commodity bases like potassium hydroxide represents a fundamental shift in the cost structure of cyclopropane production. By removing the need for expensive ligands and metal scavengers, the process eliminates several unit operations from the downstream processing train. This reduction in complexity translates directly into lower capital expenditure for equipment and reduced operational expenditure for labor and utilities. Furthermore, the high reaction yield minimizes the loss of valuable starting materials, ensuring that every kilogram of input generates maximum output value. These factors combine to create a highly competitive cost position for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: The reliance on readily available, non-hazardous reagents enhances the resilience of the supply chain against external shocks. Unlike diazomethane, which requires on-site generation due to its instability, the reagents used in this process are stable, shelf-stable, and easily transported. This stability allows for larger inventory buffers and more flexible scheduling, reducing the risk of production stoppages due to reagent shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing production throughput and ensuring consistent delivery schedules to downstream customers.
- Scalability and Environmental Compliance: The simplicity of the reaction setup, which utilizes standard heating and distillation equipment, facilitates seamless scale-up from pilot plant to full commercial production. There is no need for specialized high-pressure reactors or complex inert atmosphere systems, lowering the barrier to entry for contract manufacturing organizations. Moreover, the generation of nitrogen gas as the sole gaseous byproduct aligns perfectly with green chemistry principles, minimizing the carbon footprint of the manufacturing process. This environmental compatibility simplifies regulatory approvals and supports corporate sustainability goals, making the final product more attractive to environmentally conscious pharmaceutical and agrochemical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the practical aspects of adoption.
Q: How does this thermal method compare to traditional Simmons-Smith cyclopropanation?
A: Unlike the Simmons-Smith reaction which requires hazardous organozinc reagents and strict anhydrous conditions, this thermal denitrification method operates under atmospheric pressure using simple base catalysts, significantly reducing safety risks and equipment costs.
Q: What is the expected yield and purity profile for this synthesis?
A: Experimental data indicates reaction yields consistently exceeding 95%, with some optimized batches reaching 98%. The process inherently minimizes heavy metal impurities, simplifying downstream purification for pharmaceutical applications.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the method eliminates the need for expensive transition metal catalysts and explosive diazo reagents. The straightforward heating and distillation protocol is highly adaptable to standard industrial reactors, ensuring robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free thermal synthesis route for the global supply of high-performance chemical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of cyclopropane derivative meets the exacting standards required for pharmaceutical and aerospace applications. We are committed to leveraging this advanced technology to provide our partners with a secure, cost-effective, and sustainable source of critical building blocks.
We invite procurement leaders and R&D teams to engage with us to explore how this innovative process can optimize your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic benefits tailored to your volume needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to support your next project. Contact us today to discuss how we can collaborate to bring your cyclopropane-based innovations to market faster and more efficiently.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
