Advanced Chiral Zinc Complex Synthesis for High-Purity Pharmaceutical Intermediates Manufacturing
Advanced Chiral Zinc Complex Synthesis for High-Purity Pharmaceutical Intermediates Manufacturing
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for cost-effective and environmentally benign synthetic routes in the fine chemical industry. Patent CN102633738A introduces a groundbreaking chiral zinc complex, specifically a bis(4-isopropyl-4,5-dihydro-2-oxazolinyl)acetonitrile Zn complex, which represents a pivotal advancement in the design of nitrogenous chiral metal-organic coordination compounds. This innovation addresses the critical demand for reliable pharma intermediates supplier solutions by offering a robust catalytic system that operates effectively in allylic alkylation and Baylis-Hillman reactions. Unlike traditional systems that rely on scarce transition metals, this zinc-based architecture leverages the abundant availability of zinc chloride and simple amino alcohol precursors to construct a highly modular catalytic environment. The technical breakthrough lies not only in the molecular structure but in the streamlined one-pot synthesis methodology that drastically simplifies the manufacturing workflow, thereby enhancing the overall economic viability for large-scale chemical production facilities seeking to optimize their operational expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral catalysts for asymmetric organic transformations has been plagued by prohibitive costs and complex multi-step procedures that hinder commercial scalability. Conventional approaches often necessitate the use of precious metal centers such as palladium, rhodium, or ruthenium, which introduce severe supply chain vulnerabilities and escalate the final cost of the active pharmaceutical ingredient (API). Furthermore, the ligand systems traditionally employed, such as sophisticated phosphines or specialized N-heterocyclic carbenes, frequently require rigorous exclusion of moisture and oxygen, demanding expensive inert atmosphere gloveboxes and specialized reactor configurations that are difficult to maintain in continuous flow manufacturing settings. The purification of these legacy catalysts is equally challenging, often involving tedious chromatographic separations that generate substantial volumes of hazardous organic waste, conflicting with modern green chemistry principles and increasing the environmental compliance burden for chemical manufacturers. These cumulative inefficiencies create a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing, forcing companies to absorb high overheads that ultimately erode profit margins in competitive global markets.
The Novel Approach
In stark contrast to these legacy limitations, the novel approach detailed in the patent utilizes a convergent synthesis strategy that assembles the chiral zinc complex directly from commodity chemicals under relatively mild thermal conditions. The core innovation involves the in situ generation of the bis-oxazoline ligand framework through the reaction of tetracyanoethylene with D-valinol, followed immediately by coordination with anhydrous zinc chloride in a chlorobenzene solvent system. This methodology eliminates the need for pre-synthesized ligands, effectively collapsing multiple synthetic steps into a single reflux operation that can be sustained for 36 to 72 hours to ensure complete conversion. 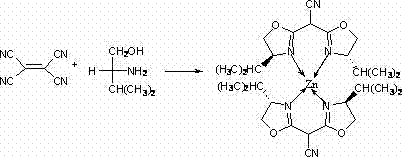 The use of zinc, a non-toxic and earth-abundant metal, fundamentally alters the economic equation by replacing expensive noble metals with a cost-effective alternative that retains sufficient Lewis acidity to drive catalytic cycles. This streamlined process not only reduces the number of unit operations required but also simplifies the downstream purification protocol, allowing for the isolation of the target complex through standard extraction and column chromatography techniques that are easily adaptable to industrial scale-up.
The use of zinc, a non-toxic and earth-abundant metal, fundamentally alters the economic equation by replacing expensive noble metals with a cost-effective alternative that retains sufficient Lewis acidity to drive catalytic cycles. This streamlined process not only reduces the number of unit operations required but also simplifies the downstream purification protocol, allowing for the isolation of the target complex through standard extraction and column chromatography techniques that are easily adaptable to industrial scale-up.
Mechanistic Insights into Zn-N Coordination and Catalytic Activity
The structural integrity and catalytic efficacy of this novel complex are rooted in the precise coordination geometry established between the zinc center and the nitrogen atoms of the bis-oxazoline ligand system. 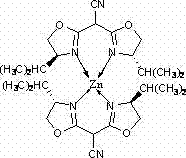 Spectroscopic analysis confirms that the tetracyanoethylene precursor undergoes a decomposition pathway in the presence of zinc chloride to yield a tricyanomethane intermediate, which subsequently condenses with two equivalents of D-valinol to form the rigid bis-oxazoline scaffold. This scaffold then chelates the zinc ion in a 2:1 stoichiometry, releasing two molecules of hydrochloric acid as a byproduct and establishing a stable tetrahedral or distorted tetrahedral coordination environment around the metal center. The resulting chiral pocket created by the isopropyl groups on the oxazoline rings provides the necessary steric bulk to induce enantioselectivity during substrate binding, a critical feature for the synthesis of high-purity OLED material or pharmaceutical intermediates where optical purity is paramount. The robustness of the Zn-N bond ensures that the catalyst maintains its structural fidelity throughout the reaction cycle, preventing leaching of the metal and ensuring consistent performance over extended operational periods.
Spectroscopic analysis confirms that the tetracyanoethylene precursor undergoes a decomposition pathway in the presence of zinc chloride to yield a tricyanomethane intermediate, which subsequently condenses with two equivalents of D-valinol to form the rigid bis-oxazoline scaffold. This scaffold then chelates the zinc ion in a 2:1 stoichiometry, releasing two molecules of hydrochloric acid as a byproduct and establishing a stable tetrahedral or distorted tetrahedral coordination environment around the metal center. The resulting chiral pocket created by the isopropyl groups on the oxazoline rings provides the necessary steric bulk to induce enantioselectivity during substrate binding, a critical feature for the synthesis of high-purity OLED material or pharmaceutical intermediates where optical purity is paramount. The robustness of the Zn-N bond ensures that the catalyst maintains its structural fidelity throughout the reaction cycle, preventing leaching of the metal and ensuring consistent performance over extended operational periods.
Beyond its structural stability, the complex demonstrates versatile catalytic utility in carbon-carbon bond-forming reactions, specifically serving as a potent Lewis acid activator for electrophilic substrates. 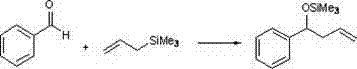 In the context of allylic alkylation of benzaldehyde, the zinc center activates the carbonyl group towards nucleophilic attack by allyl trimethyl silane, facilitating the formation of homoallylic alcohols with moderate to good conversion rates. Similarly, in Baylis-Hillman reactions involving methyl acrylate, the complex promotes the coupling of aldehydes with activated alkenes, showcasing its broad substrate scope and adaptability to different reaction manifolds. The mechanism likely involves the coordination of the Lewis acidic zinc species to the oxygen atom of the aldehyde, increasing its electrophilicity and lowering the activation energy for the nucleophilic addition step. This dual functionality in promoting both alkylation and condensation reactions underscores the value of this catalyst as a multifunctional tool for the commercial scale-up of complex polymer additives and fine chemicals, offering chemists a single catalytic platform capable of addressing diverse synthetic challenges.
In the context of allylic alkylation of benzaldehyde, the zinc center activates the carbonyl group towards nucleophilic attack by allyl trimethyl silane, facilitating the formation of homoallylic alcohols with moderate to good conversion rates. Similarly, in Baylis-Hillman reactions involving methyl acrylate, the complex promotes the coupling of aldehydes with activated alkenes, showcasing its broad substrate scope and adaptability to different reaction manifolds. The mechanism likely involves the coordination of the Lewis acidic zinc species to the oxygen atom of the aldehyde, increasing its electrophilicity and lowering the activation energy for the nucleophilic addition step. This dual functionality in promoting both alkylation and condensation reactions underscores the value of this catalyst as a multifunctional tool for the commercial scale-up of complex polymer additives and fine chemicals, offering chemists a single catalytic platform capable of addressing diverse synthetic challenges.
How to Synthesize Bis(4-isopropyl-4,5-dihydro-2-oxazolinyl)acetonitrile Zn Complex Efficiently
The practical implementation of this synthesis route requires careful attention to anhydrous conditions and thermal management to maximize yield and purity. The process begins with the charging of anhydrous zinc chloride, tetracyanoethylene, and D-valinol into a reactor containing chlorobenzene, followed by heating the mixture to reflux temperatures for an extended period to drive the condensation and coordination equilibria to completion. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Reflux tetracyanoethylene and D-valinol in chlorobenzene with anhydrous ZnCl2 catalyst under anhydrous conditions for 36-72 hours.
- Remove solvent, dissolve residue in water, extract with chloroform, and purify the organic phase via column chromatography.
- Elute with petroleum ether and methylene dichloride to isolate the white crystalline chiral zinc complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-based catalytic technology offers a compelling value proposition centered on raw material security and operational cost optimization. The reliance on tetracyanoethylene and valinol, which are widely available commodity chemicals, mitigates the risk of supply disruptions often associated with specialized ligands or rare earth metals, thereby ensuring a consistent and reliable supply chain for high-purity pharmaceutical intermediates. Furthermore, the elimination of precious metal catalysts removes the necessity for expensive metal scavenging steps in the downstream processing of the final API, which significantly reduces the consumption of specialized resins and lowers the overall cost of goods sold (COGS). This process intensification allows manufacturers to achieve substantial cost savings without compromising on the quality or stereochemical integrity of the final product, making it an attractive option for generic drug manufacturers operating on thin margins.
- Cost Reduction in Manufacturing: The substitution of noble metals with zinc chloride results in a drastic reduction in catalyst procurement costs, as zinc salts are orders of magnitude cheaper than palladium or rhodium counterparts. Additionally, the simplified one-pot synthesis minimizes solvent usage and energy consumption by reducing the number of isolation and purification stages required, leading to a leaner and more efficient manufacturing process. The avoidance of complex ligand synthesis further contributes to cost efficiency, as the ligand is generated in situ from inexpensive starting materials, effectively bundling the ligand production cost into the main reaction step.
- Enhanced Supply Chain Reliability: By utilizing a catalyst system based on earth-abundant elements and common organic feedstocks, manufacturers can decouple their production schedules from the volatile market dynamics of the precious metals sector. This stability is crucial for long-term contract manufacturing organizations (CMOs) that require guaranteed delivery timelines and consistent pricing structures to satisfy their own downstream clients. The robustness of the chlorobenzene solvent system also implies that the process can be run in standard glass-lined or stainless steel reactors without the need for exotic metallurgy, further enhancing the flexibility and reliability of the production infrastructure.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, as the reaction conditions (reflux in chlorobenzene) are easily transferable from laboratory flasks to multi-ton industrial reactors without significant re-engineering. From an environmental perspective, the use of zinc, a less toxic metal compared to heavy transition metals, simplifies waste stream management and reduces the regulatory burden associated with heavy metal discharge limits. The ability to recover and recycle the chlorobenzene solvent adds another layer of sustainability, aligning the process with increasingly stringent global environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc complex technology in industrial settings. These answers are derived directly from the patent specifications and practical experience in catalytic process development.
Q: What are the primary advantages of this zinc complex over traditional noble metal catalysts?
A: This zinc-based system eliminates the need for expensive palladium or rhodium catalysts, significantly reducing raw material costs while maintaining effective catalytic activity for allylic alkylation and Baylis-Hillman reactions.
Q: Is the synthesis process scalable for industrial production?
A: Yes, the process utilizes common solvents like chlorobenzene and commodity chemicals like tetracyanoethylene, making it highly amenable to scale-up from laboratory to multi-ton commercial production without specialized high-pressure equipment.
Q: What is the enantioselectivity potential of this catalyst?
A: Derived from D-valinol, the complex possesses inherent chirality suitable for asymmetric synthesis, offering a pathway to high optical purity in pharmaceutical intermediates when optimized for specific substrate classes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this chiral zinc complex technology in advancing the synthesis of next-generation pharmaceutical intermediates and fine chemicals. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of catalyst or intermediate produced, giving you the confidence to integrate this technology into your most critical synthetic routes.
We invite you to collaborate with our technical team to explore how this cost-effective catalytic solution can be tailored to your specific process needs. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis, along with specific COA data and route feasibility assessments that demonstrate the tangible economic benefits of adopting this zinc-based catalytic system for your upcoming projects.
