Advanced Asymmetric Hydrogenation for High-Purity Optically Active Citronellal Production
The global demand for high-purity chiral intermediates in the flavor and fragrance industry continues to drive innovation in catalytic processes. A pivotal advancement in this sector is detailed in patent CN111056932A, which discloses a highly efficient method for preparing optically active citronellal via asymmetric hydrogenation. This technology addresses long-standing challenges in homogeneous catalysis, specifically focusing on the stability and longevity of rhodium-based catalyst systems. By rigorously controlling trace impurities such as peroxides and hydrolytic chlorine in the substrate—neral or geranial—the process achieves unprecedented turnover numbers, making it exceptionally viable for large-scale industrial manufacturing. For R&D directors and procurement specialists seeking a reliable flavor & fragrance intermediate supplier, understanding the mechanistic nuances of this patent is crucial for securing a stable supply chain of high-value chiral aldehydes.
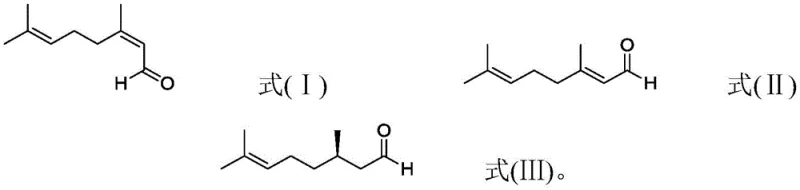
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of optically active R-citronellal has been plagued by inefficiencies inherent in early catalytic systems. Prior art, such as European patent EP0000315, utilized simple 2,3-bis(diphenylphosphino)butane ligands which suffered from mediocre chemo- and stereoselectivity, necessitating the use of large amounts of expensive catalyst to drive the reaction. Furthermore, literature from the Journal of Molecular Catalysis (1982) highlighted that while rhodium carbonyl complexes could achieve high chemo-selectivity, the stereoselectivity remained limited, and hydrogenation efficiency was suboptimal. More recent attempts, such as those disclosed in WO2009068444, introduced mixtures of CO and H2 to improve selectivity but still faced significant hurdles regarding catalyst deactivation. These conventional methods often resulted in short catalyst service lives due to noble metal coupling inactivation and complex operational procedures, rendering them economically unfeasible for cost-sensitive synthetic flavors manufacturing.
The Novel Approach
The methodology presented in CN111056932A represents a paradigm shift by targeting the root cause of catalyst failure: substrate impurity. Instead of merely optimizing reaction parameters, this novel approach mandates a pretreatment step where the substrate (neral and/or geranial) is purified to ensure a peroxide value of ≤500ppm and hydrolytic chlorine of ≤10ppm. This rigorous purification, often achieved through rectification after treatment with reducing agents like ferrous sulfate, fundamentally alters the reaction landscape. By eliminating the accumulated poisoning effect of trace impurities, the catalytic stability is dramatically enhanced. This allows the transition metal catalyst to maintain high activity over extended periods, facilitating a total turnover number that can exceed 50,000 and even reach nearly 1,000,000 in continuous processes, thereby offering a robust solution for the commercial scale-up of complex flavor intermediates.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The core of this technological breakthrough lies in the intricate interplay between the transition metal center and the chiral ligand environment. The catalyst is formed by reacting a soluble rhodium compound, such as Rh(CO)2acac or [Rh(cod)OH]2, with an optically active ligand containing two phosphorus atoms. Preferred ligands include those defined by formulas (IV), (V), and (VI), which feature specific steric and electronic properties essential for inducing chirality. As illustrated in the structural diagrams, these ligands create a chiral pocket around the rhodium atom, directing the hydrogen addition to the pro-chiral double bond of the substrate with high enantioselectivity. The presence of carbon monoxide in the hydrogen feed (100ppm to 10,000ppm) is also critical, as it helps modulate the electronic state of the rhodium center, preventing the formation of inactive rhodium clusters while maintaining sufficient coordination sites for substrate binding.
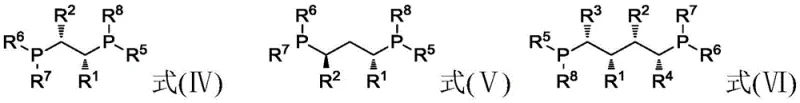
However, the true genius of this patent is its explanation of catalyst deactivation mechanisms, which are vividly depicted in the degradation pathways. In the absence of strict impurity control, trace peroxides in the neral/geranial substrate act as potent oxidizing agents. They readily react with the phosphine ligands, converting them into phosphine oxides (as shown in formula XIII), which lack the necessary electron-donating capability to stabilize the rhodium complex. Simultaneously, hydrolyzable chlorine impurities undergo oxidative addition with the transition metal, stripping the ligand and converting the active species into inactive metal chlorides (formula XIV). By preemptively removing these poisons through the described pretreatment protocol, the integrity of the catalytic cycle is preserved. This ensures that the rhodium complex remains in its active hydride form, capable of executing thousands of hydrogenation cycles without significant loss of optical purity or conversion rate.
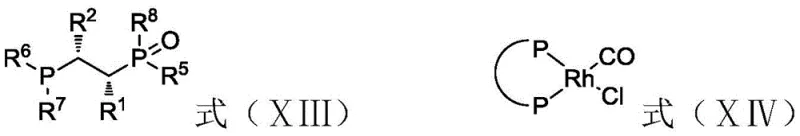
How to Synthesize Optically Active Citronellal Efficiently
The synthesis of optically active citronellal via this patented route involves a streamlined sequence that balances chemical precision with operational simplicity. The process begins with the critical pretreatment of the unsaturated aldehyde substrate to remove oxidative and halogenated contaminants, followed by the in-situ generation of the chiral rhodium catalyst. The reaction is then conducted under controlled hydrogen pressure with trace CO, often without the need for additional solvents, which simplifies product isolation. This approach not only maximizes yield and enantiomeric excess but also aligns with green chemistry principles by reducing waste generation. For technical teams looking to implement this, the detailed standardized synthesis steps are outlined below to ensure reproducibility and safety.
- Pretreat the neral or geranial substrate with reducing agents like ferrous sulfate or ascorbic acid, followed by rectification to ensure peroxide value is ≤500ppm and hydrolytic chlorine is ≤10ppm.
- Prepare the catalyst by reacting a rhodium compound (e.g., Rh(CO)2acac) with a chiral bis-phosphine ligand (e.g., Chiraphos) in a molar ratio of 0.5: 1 to 10:1.
- Conduct the asymmetric hydrogenation reaction under hydrogen pressure (0.1-10 MPa) containing 100-10,000 ppm CO at temperatures between 25-90°C to achieve high conversion and optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the implications of this technology extend far beyond the laboratory bench, offering tangible benefits in cost structure and supply reliability. The primary economic driver is the drastic extension of catalyst life. In traditional homogeneous hydrogenation, the frequent need to replenish expensive rhodium catalysts due to poisoning represents a significant operational expense. By stabilizing the catalyst through substrate purification, this method allows for a single batch of catalyst to process vastly larger quantities of substrate. This directly translates to cost reduction in synthetic flavors manufacturing by minimizing the consumption of precious metals and reducing the frequency of catalyst charging operations. Furthermore, the ability to operate under solvent-free conditions eliminates the costs associated with solvent purchase, recovery, and disposal, further enhancing the overall process economics.
- Cost Reduction in Manufacturing: The elimination of frequent catalyst reloading and the potential for solvent-free operation significantly lowers the variable costs associated with production. By achieving turnover numbers in the hundreds of thousands, the amortized cost of the rhodium catalyst per kilogram of product becomes negligible compared to conventional methods. Additionally, the high conversion rates (>99%) minimize raw material waste, ensuring that the expensive neral/geranial feedstock is efficiently converted into the high-value citronellal product without the need for complex recycling loops.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system against impurity-induced deactivation means that production schedules are less likely to be disrupted by unexpected catalyst failures. The process tolerates a wider range of operating conditions while maintaining high selectivity, providing a buffer against minor fluctuations in utility supplies or feed quality. This stability is crucial for maintaining consistent delivery timelines to downstream customers in the fragrance and pharmaceutical sectors, who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The simplicity of the process, particularly the option to run without solvents, greatly facilitates scale-up from pilot plant to commercial production. Fewer unit operations for solvent handling reduce the capital expenditure required for new facilities. Moreover, the reduction in chemical waste and the avoidance of volatile organic compounds (VOCs) associated with solvents align perfectly with increasingly stringent global environmental regulations, mitigating regulatory risk and enhancing the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric hydrogenation technology. These answers are derived directly from the experimental data and technical disclosures found in the patent documentation, providing clarity on process feasibility and performance metrics. Understanding these details is essential for evaluating the technology's fit within existing manufacturing frameworks.
Q: Why is controlling peroxide value critical in citronellal synthesis?
A: Trace peroxides in the substrate can oxidize the phosphine ligand to phosphine oxide, destroying the catalyst structure and causing rapid deactivation. Keeping peroxide values below 500ppm prevents this poisoning.
Q: What represents the primary advantage of this rhodium-catalyzed method over previous techniques?
A: The primary advantage is the significantly extended catalyst service life and higher turnover number (up to ~943,312), achieved by eliminating trace impurities that typically cause noble metal coupling inactivation.
Q: Can this process be operated without solvents?
A: Yes, the patent explicitly states that the asymmetric hydrogenation can be carried out under solvent-free conditions, which simplifies downstream processing and reduces environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Citronellal Supplier
The technological potential of high-turnover asymmetric hydrogenation is immense, yet translating patent data into commercial reality requires deep process engineering expertise. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this rhodium-catalyzed process are fully realized in practice. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of monitoring trace impurities like peroxides and hydrolytic chlorine to the ppm level, guaranteeing that every batch meets stringent purity specifications required by top-tier global clients.
We invite you to leverage our technical capabilities to optimize your supply chain for optically active intermediates. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing protocols can deliver superior value and reliability for your business.
