Advanced Thiocarbamate Production: Transforming Mining Reagent Manufacturing Efficiency and Purity
The global demand for high-efficiency mineral beneficiation reagents continues to drive innovation in fine chemical synthesis, particularly for thiocarbamate compounds which serve as critical collectors in froth flotation processes. A pivotal advancement in this domain is documented in patent CN1045086C, which outlines a novel preparation method that fundamentally restructures the synthetic pathway to eliminate traditional inefficiencies. This technology addresses the longstanding challenges of yield limitation and environmental burden associated with legacy manufacturing protocols by introducing a homogeneous reaction system. By utilizing the reactant alcohol itself as the solvent medium, the process achieves a seamless integration of nucleophilic substitution, isomerization, and esterification steps within a single reactor vessel. This strategic modification not only enhances the thermodynamic favorability of the isomerization step but also drastically simplifies the downstream processing requirements. For industrial stakeholders, this represents a significant leap forward in process intensification, offering a robust framework for the reliable mining chemical supplier seeking to optimize production metrics while adhering to stricter environmental compliance standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of thiocarbamate compounds has relied heavily on biphasic reaction systems, as exemplified by earlier methodologies such as those disclosed in US patent 4479903. These conventional processes typically involve dissolving sodium thiocyanate in water and employing tetrabutylammonium bromide as a phase transfer catalyst to facilitate the reaction with alkyl halides in an organic phase. This approach introduces several critical bottlenecks that hinder operational efficiency and cost-effectiveness in large-scale manufacturing environments. Firstly, the reliance on aqueous media necessitates complex phase separation steps, which are often time-consuming and result in significant product loss due to the solubility of intermediates in the water phase. Secondly, the presence of water creates a low-boiling azeotrope that limits the reflux temperature to approximately 90°C, which is thermodynamically insufficient to drive the isomerization of thiocyanate to isothiocyanate to completion. Furthermore, the subsequent removal of residual water requires energy-intensive distillation under reduced pressure, during which toxic intermediates may volatilize, posing severe safety and environmental hazards. The accumulation of saline wastewater contaminated with organic byproducts and phase transfer catalysts further exacerbates the ecological footprint, requiring expensive treatment infrastructure that erodes profit margins.
The Novel Approach
In stark contrast to the fragmented and inefficient aqueous protocols, the novel methodology described in the patent data utilizes a homogeneous alcohol-based system that streamlines the entire synthetic sequence into a cohesive, one-pot operation. By selecting an alcohol (ROH) with a carbon chain length of C4-6 as both the reactant and the solvent, the process creates a unified medium that dissolves the thiocyanate salt and facilitates the reaction with the halogenated hydrocarbon without the need for phase transfer agents. This structural change allows the reaction temperature to be elevated significantly, reaching reflux temperatures between 80°C and 140°C, which provides the necessary thermal energy to overcome the activation barrier for the thiocyanate-to-isothiocyanate isomerization. The absence of water eliminates the formation of low-boiling azeotropes, thereby permitting higher operating temperatures that favor the forward reaction kinetics. Moreover, because the alcohol is present in excess throughout the process, the newly formed isothiocyanate is immediately consumed to generate the final thiocarbamate product, effectively pulling the equilibrium towards completion according to Le Chatelier's principle. This integrated approach not only omits the tedious dehydration and separation steps but also ensures that toxic intermediates remain sealed within the reaction system until conversion, thereby enhancing operator safety and reducing the intervention of impurities.
Mechanistic Insights into Alcohol-Mediated Isothiocyanate Formation
The core chemical transformation in this advanced synthesis route involves a sequential cascade of nucleophilic substitution followed by thermal isomerization and final esterification. Initially, the thiocyanate anion attacks the alkyl halide to form an alkyl thiocyanate intermediate, a step that proceeds efficiently in the polar alcoholic medium without the steric hindrance often encountered in biphasic systems. Upon heating, this intermediate undergoes a rearrangement to form the more reactive isothiocyanate species, a process that is kinetically accelerated by the elevated boiling point of the alcohol solvent compared to water. The presence of excess alcohol ensures that as soon as the isothiocyanate is generated, it reacts to form the stable thiocarbamate linkage, preventing the reverse reaction and minimizing side products. This continuous consumption of the reactive intermediate is crucial for maintaining high selectivity and yield, as it prevents the accumulation of unstable species that could otherwise degrade or polymerize under prolonged heating conditions.
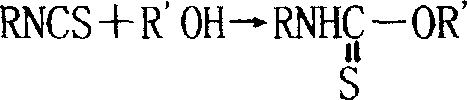
Furthermore, the mechanistic pathway benefits from the potential addition of titanium-based catalysts, such as tetrabutyl titanate derivatives, which can further lower the activation energy for the final esterification step. The catalyst facilitates the coordination of the alcohol oxygen to the electrophilic carbon of the isothiocyanate group, enhancing the nucleophilic attack and ensuring rapid conversion even at moderate temperatures. From an impurity control perspective, the homogeneous nature of the reaction minimizes the formation of emulsions and interfacial byproducts that are common in phase-transfer catalyzed reactions. The absence of water also precludes hydrolysis side reactions that could lead to the formation of thiols or other sulfur-containing impurities, resulting in a crude product with significantly higher purity profiles. This mechanistic elegance translates directly into simplified purification protocols, as the primary byproduct is simply an inorganic salt that can be easily removed by filtration, leaving a filtrate that requires only distillation to recover excess reagents and isolate the high-purity target compound.
How to Synthesize Thiocarbamate Compounds Efficiently
The implementation of this synthesis strategy requires precise control over reaction parameters to maximize the benefits of the homogeneous system. The process begins with the dissolution of the thiocyanate salt in the selected alcohol, followed by the controlled addition of the halogenated hydrocarbon to manage exothermicity. Subsequent heating stages are critical, with specific temperature ramps designed to first promote isomerization and then drive the final coupling reaction to completion. The detailed standardized synthesis steps, including specific molar ratios and timing for catalyst addition, are outlined below to ensure reproducibility and safety in a pilot or production setting.
- Dissolve thiocyanate salt (MSCN) in alcohol (ROH) and add halogenated hydrocarbon (R-X) at 30-80°C to form thiocyanate intermediate.
- Heat the mixture to 80-140°C and reflux for 2-10 hours to isomerize thiocyanate into isothiocyanate.
- Add titanium catalyst and reflux at 110-170°C for 2-12 hours to react isothiocyanate with alcohol, then filter and distill to isolate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers substantial strategic advantages that extend beyond mere technical feasibility. The elimination of complex unit operations such as phase separation, repeated dehydration, and wastewater treatment translates directly into a streamlined manufacturing workflow that reduces both capital expenditure and operational overhead. By removing the dependency on expensive phase transfer catalysts like tetrabutylammonium bromide, the raw material costs are significantly lowered, while the simplified process flow reduces the labor and energy inputs required per kilogram of product. This efficiency gain is particularly relevant in the context of cost reduction in mining reagent manufacturing, where margin pressures often dictate the viability of supply contracts. Furthermore, the robustness of the one-pot process enhances supply chain reliability by minimizing the risk of batch failures associated with complex multi-step separations, ensuring consistent delivery schedules for downstream customers.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the drastic simplification of the production cycle and the elimination of auxiliary chemicals. By obviating the need for phase transfer agents and the associated recovery or disposal costs, the variable cost of goods sold is materially reduced. Additionally, the energy savings derived from avoiding repeated distillation cycles for water removal contribute to a lower carbon footprint and reduced utility bills. The ability to recover and recycle excess alcohol and alkyl halides directly from the reaction mixture further enhances the atom economy of the process, ensuring that raw material utilization is optimized. These cumulative factors result in a highly competitive cost structure that allows for flexible pricing strategies in volatile market conditions without compromising profitability.
- Enhanced Supply Chain Reliability: From a logistics and planning perspective, the shortened cycle time inherent in this continuous or semi-continuous process allows for faster turnaround on customer orders. The reduction in process steps decreases the likelihood of bottlenecks that often plague traditional batch manufacturing, thereby improving the overall throughput capacity of the production facility. Moreover, the use of readily available and stable raw materials, such as common alcohols and alkyl halides, mitigates the risk of supply disruptions that might occur with specialized catalysts or reagents. This stability ensures that the reliable mining chemical supplier can maintain consistent inventory levels and meet urgent procurement demands with greater agility, fostering stronger long-term partnerships with key accounts in the mining and agrochemical sectors.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, as the homogeneous reaction conditions are easily replicated from laboratory benchtop to industrial reactor scales without significant re-engineering. The absence of aqueous effluent streams simplifies regulatory compliance, as there is no toxic wastewater requiring complex treatment before discharge, aligning with increasingly stringent global environmental standards. This 'green chemistry' aspect not only reduces the liability associated with environmental permits but also enhances the brand reputation of the manufacturer among sustainability-conscious clients. The ability to scale up complex thiocarbamate production while maintaining high purity and yield demonstrates a mature technological capability that supports the expansion of product portfolios into high-value specialty chemical markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiocarbamate synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical principles underlying the process for technical decision-makers. Understanding these details is essential for evaluating the feasibility of integrating this method into existing production lines or for specifying quality requirements in procurement contracts.
Q: How does the alcohol-solvent method improve yield compared to traditional aqueous methods?
A: By using alcohol as a homogeneous medium, the reaction temperature for isomerization can be raised to 140°C, significantly shifting the equilibrium towards isothiocyanate formation. Additionally, the immediate consumption of isothiocyanate by the excess alcohol drives the reaction to completion, avoiding the low-temperature limitations of aqueous azeotropes.
Q: What are the environmental advantages of eliminating tetrabutylammonium bromide?
A: Removing the phase transfer catalyst eliminates the generation of toxic wastewater containing organic intermediates and salts. This simplifies waste treatment protocols, reduces the need for expensive water treatment infrastructure, and prevents environmental contamination associated with quaternary ammonium salt discharge.
Q: Is this process suitable for large-scale commercial production of flotation collectors?
A: Yes, the process is highly scalable as it operates in a single reaction vessel without intermediate separation or purification steps. The elimination of complex dehydration and phase separation stages reduces operational complexity and equipment requirements, making it ideal for continuous or batch commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiocarbamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the modern chemical industry. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory techniques like the one described in CN1045086C are successfully translated into robust industrial operations. We are committed to delivering high-purity thiocarbamate compounds that meet stringent purity specifications, supported by our rigorous QC labs which employ advanced analytical methods to verify product identity and impurity profiles. Our capability to adapt and optimize such solvent-free or homogeneous catalytic systems positions us as a strategic partner for companies seeking to enhance their supply chain resilience and reduce manufacturing costs.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits applicable to your specific supply chain context. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your mineral processing operations.
